RESEARCH LETTER
Reconsidering Efficacy and Safety: A Critical Appraisal of Repurposing Anti-inflammatory Targeted Drugs in Darier disease
Estelle BURLE1† , Juliette MAZEREEUW-HAUTIER1,2† and Maella SEVERINO-FREIRE1,2*
, Juliette MAZEREEUW-HAUTIER1,2† and Maella SEVERINO-FREIRE1,2*
1Department of Dermatology and Allergology, Toulouse University Hospital, 31400 Toulouse, France, and 2Reference Centre for Rare Skin Diseases, Dermatology Department, CHU Toulouse, Toulouse, France. *Emails: severino-freire.m@chu-toulouse.fr; burle.e@chu-toulouse.fr
†These authors contributed equally to this work as first authors.
Citation: Acta Derm Venereol 2026; 106: adv-2026-0322. DOI: https://doi.org/10.2340/actadv.v106.adv-2026-0322.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Jan 6, 2026. Accepted after revision: Apr 1, 2026.
Published: Apr 22, 2026.
Competing interests and funding: This retrospective study was conducted in accordance with the Declaration of Helsinki (1964, last revised in 2013) and approved by the relevant ethical committee (CHU Toulouse). Data were collected and processed in compliance with French regulations governing research on previously collected medical data (MR-004).
JM-H and MS-F have served as investigators and experts over the past five years for AbbVie and Lilly. EB have no conflicts of interest to declare.
To the Editor,
Darier disease (DD) is a rare autosomal dominant genodermatosis caused by mutations in ATP2A2, which encodes the sarco/endoplasmic reticulum calcium ATPase 2 (SERCA2) (1). Patients develop inflammatory, keratotic papules, often erosive and exudative, mainly in seborrheic areas and skin folds, with variable nail, mucosal, neuropsychiatric and ocular involvement (2).
Heat, UV exposure and sweating exacerbate lesions, while chronic symptoms such as pain, pruritus and infections markedly impair quality of life (QoL). Therapeutic options, whether topical or systemic, are mainly symptomatic, not very effective and often associated with side-effects. The main systemic therapies used are acitretin and alitretinoin.
Recent studies have identified a Th17-driven immune profile in DD skin lesions, suggesting potential for repurposing targeted anti-inflammatory agents (3). Isolated case reports have shown positive outcomes with biologics or JAK inhibitors (JAKi) (4, 5).
We report our experience with a series of 8 patients (7 females, 1 male) with moderate-to-severe, long-standing, histologically confirmed DD treated with JAK-STAT inhibitors or biologics for ≥6 months or until discontinuation due to adverse events. The mean age was 47.9±14.3 years (range 30-67), and the mean body surface area involvement was 36.9 %±14.6 (range 20-70). Treatments included IL-17 inhibitors (ixekizumab, n=2 – including one patient with cutaneous transcriptomic evidence of IL-17 overexpression – and secukinumab, n=2), IL-23 inhibitor (guselkumab, n=1), JAKi (baricitinib, n=1; upadacitinib, n=1) and TYK2 inhibitor (deucravacitinib, n=1).
All treatments were administered at the dosages approved in their respective marketing authorizations for psoriasis. Concomitant therapies included oral acitretin (n=3) and topical agents for all but one (mainly corticosteroids and silver sulfadiazine). Clinical efficacy was evaluated using the Investigator Global Assessment-Change (IGA-C) score (from +1 = worsening to -3=remission); scores 0/+1 indicated nonresponse, and -1 to -3 indicated response (-2/-3=good response).
Two patients discontinued treatment after 1 or 3 months, due to adverse events (severe herpes infection requiring a 3-day hospitalization on upadacitinib; dizziness with hypotension on ixekizumab). Only one patient responded (IGA-C -1 at 6 months; NRS pruritus 4 at 6 months vs 7 at inclusion) but relapsed after discontinuation, with partial improvement upon retreatment. Two remained stable (IGA-C 0), including the IL-17-overexpressing patient, and three worsened with increasing body surface area or severity (Fig. 1).
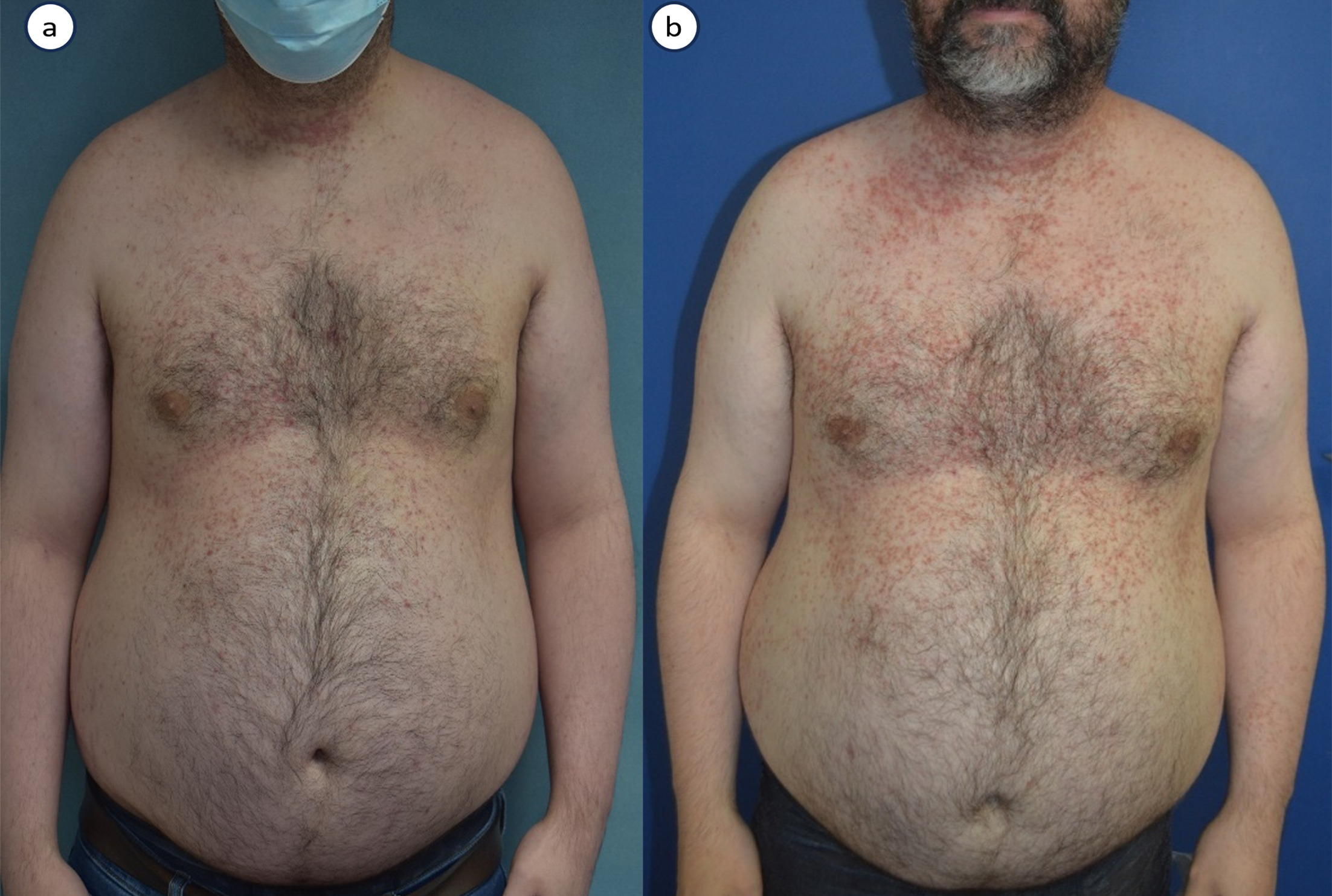
Fig. 1. Thirty-six-year-old male with Darier disease treated with secukinumab 300 mg every 4 weeks. At baseline (a), he presented with predominantly inflammatory keratotic papules on the lateral sides of the neck and mid-trunk. After 6 months (b) his condition worsened, with more diffuse and inflammatory lesions.
These findings are in contrast with previously published reports.
Ettinger et al. reported 3 patients treated with IL-23 or IL-17 inhibitors (selected on dominant cytokines in affected skin), who exhibited a clinical improvement within 3 to 4 months, sustainable at 1 year, and accompanied by normalization of cutaneous inflammatory cytokine gene expression (3).
Giorgio CM et al. reported near-complete remission at six months in three of four patients treated with brodalumab. The remaining patient, who had a more severe condition, experienced only mild relief from pruritus (6).
More recently, near-complete responses to JAKi have been reported: abrocitinib induced remission in a patient with concomitant atopic dermatitis, maintained at 4 months (5), while baricitinib achieved comparable outcomes in another patient in the absence of associated atopic dermatitis (4).
Our series, the first on Darier disease treated with biologics or JAK-STAT inhibitors, did not confirm a clear clinical benefit of these anti-inflammatory therapies. This discrepancy may be explained by publication bias, highlighting the importance of reporting negative results. Moreover, our findings draw attention to potential safety concerns, particularly severe herpes infections, in a population already predisposed to infections.
No patients received dupilumab, which has recently been reported to improve pruritus and QoL in a case of DD with perioral cutaneous cobblestoning (7). Recently, dysregulation of the MAPK/ERK pathway has emerged as a key pathogenic mechanism, with trametinib demonstrating significant clinical improvement in a refractory case resistant to conventional treatments and combination therapy with baricitinib and secukinumab (8).
In conclusion, managing DD remains challenging. Robust evaluation of repurposed anti-inflammatory drugs, ideally through clinical trials, is essential to establish their efficacy and safety.
REFERENCES
- Sakuntabhai A, Ruiz-Perez V, Carter S, Jacobsen N, Burge S, Monk S, et al. Mutations in ATP2A2, encoding a Ca2+ pump, cause Darier disease. Nat Genet 1999; 21: 271–277. https://doi.org/10.1038/6784
- Burge SM, Wilkinson JD. Darier-White disease: a review of the clinical features in 163 patients. J Am Acad Dermatol 1992; 27: 40–50. https://doi.org/10.1016/0190-9622(92)70154-8
- Ettinger M, Kimeswenger S, Deli I, Traxler J, Altrichter S, Noack P, et al. Darier disease: current insights and challenges in pathogenesis and management. J Eur Acad Dermatol Venereol 2025; 39: 942–951. https://doi.org/10.1111/jdv.20448
- Busto Leis JM, Negre GS, Mayor Ibarguren AP, Pinto PH. Response of darier disease following treatment with baricitinib. JAMA Dermatol 2022; 158: 699–701. https://doi.org/10.1001/jamadermatol.2022.1021
- Bianco M, Di Giulio S, Ibba L, Narcisi A, Costanzo A, Gargiulo L. Clearance of Darier disease lesions in a patient with concomitant atopic dermatitis treated with abrocitinib. J Eur Acad Dermatol Venereol 2025; 39: e417–e419. https://doi.org/10.1111/jdv.20399
- Giorgio CM, Di Brizzi EV, Licata G, Fiorentino C, Argenziano G, Balato A. Brodalumab for darier disease: a case series highlighting il-17 inhibition as a potential treatment. Int J Dermatol 2025; 64: 1720–1722. https://doi.org/10.1111/ijd.17751
- Alzahrani N, Grossman-Kranseler J, Swali R, Fiumara K, Zancanaro P, Tyring S, et al. Hailey-Hailey disease treated with dupilumab: a case series. Br J Dermatol 2021; 185: 680–682. https://doi.org/10.1111/bjd.20475
- Soto-García D, Dávila-Seijo P, González-Moure C, Pampín A, Zulaica A, Allegue F, et al. Trametinib as a promising therapy for Darier disease: case report. Br J Dermatol 2025; 193: 339–341. https://doi.org/10.1093/bjd/ljaf160