ORIGINAL REPORT
Sun Protection Behaviours and Photosensitivity in Japanese Children: Toyama Adjunct Study of Japan Environment and Children’s Study (JECS)
Megumi MIZAWA1* , Kenta MATSUMURA2,3
, Kenta MATSUMURA2,3 , Teruhiko MAKINO1
, Teruhiko MAKINO1 , Akiko TSUCHIDA2,3, Hidekuni INADERA2,3
, Akiko TSUCHIDA2,3, Hidekuni INADERA2,3 and Tadamichi SHIMIZU1
and Tadamichi SHIMIZU1
1Department of Dermatology, Faculty of Medicine, Academic Assembly, University of Toyama, Toyama, Japan, 2Department of Public Health, Faculty of Medicine, Academic Assembly, University of Toyama, Toyama, Japan, and 3Toyama UC (unit center) for JECS, University of Toyama, Toyama, Japan
Corr: Megumi Mizawa, Department of Dermatology, Faculty of Medicine, Academic Assembly, University of Toyama, 2630 Sugitani, Toyama, Japan. *Email: megumiza@med.u-toyama.ac.jp
Key words: Pediatrics; sun protection; photosensitivity disorders; photosensitivity; sunscreen; Photodermatology; Erythropoietic protoporphyria.
Citation: Acta Derm Venereol 2026; 106: adv-2026-0397. DOI: https://doi.org/10.2340/actadv.v106.adv-2026-0397.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/).
Submitted: Feb 3, 2026. Accepted after revision: Apr 8, 2026.
Published: Apr 28, 2026.
Competing interests and funding: The authors have no conflicts of interest to declare.
The JECS was funded by the Ministry of Environment, Japan. The findings and conclusions of this article are solely the responsibility of the authors and do not represent the official views of the Ministry of Environment, Japan.
Data are unsuitable for public deposition owing to ethical restrictions and the legal framework of Japan. The Act on the Protection of Personal Information (Act No. 57 of May 30, 2003, September 9, 2015) prohibits publicly depositing data containing personal information. The Ethical Guidelines for Medical and Health Research Involving Human Subjects enforced by the Japanese Ministry of Education, Culture, Sports, Science and Technology and the Ministry of Health, Labour and Welfare also restrict the open sharing of epidemiological data. All enquiries about access to the dataset jecs-qa-20210401 should be sent to: jecs-en@nies.go.jp. The person responsible for handling inquiries sent to this e-mail address is Dr Shoji F. Nakayama, JECS Program Office, National Institute for Environmental Studies. For other inquiries regarding data access, please contact the corresponding author.
This study was conducted in accordance with the principles of the Declaration of Helsinki. The study protocol was approved by the Institutional Review Board on Epidemiological Studies of the Japanese Ministry of the Environment and the Ethics Committee of the University of Toyama (approval number: R2019015). All the participants provided their written informed consent. This study was an observational study without any interventions; therefore, separate trial registration was not required. The Japan Environment and Children’s Study (JECS), from which this study was derived, is registered in the UMIN Clinical Trials Registry (UMIN000030786).
Children have up to 3 times the annual sun exposure of adults. This cross-sectional study evaluated sun protection behaviour, related factors and the incidence of photosensitivity in Japanese children. A self-administered questionnaire administered to mothers of 2nd-grade children as part of the Toyama Adjunct Study of the Japan Environment and Children’s Study was used and analysed (n=1,691). The results showed that 67.5 % of children always or sometimes used sunscreen when going out for a long time, while 73.2 % used at least one method of sun protection when swimming in an outdoor pool. Behaviours related to sun exposure were related to the sex of the child, Fitzpatrick skin phototype, skin symptoms after sun exposure, annual household income, father’s highest educational level and maternal age. The incidence of photosensitivity disorders was 0.5 %. Diagnoses included polymorphous light eruption in 4 (50%) and solar urticaria in 2 (25%). However, 5.9 % of children had skin symptoms after sun exposure but were undiagnosed. In general, photosensitivity is rarely encountered in paediatric patients; in this study, we determined its prevalence in children in Japan. Six factors appeared to be relevant to behaviours related to sun exposure. Further investigation is required to confirm these findings.
Significance
Children are exposed to much more sunlight than adults, yet little is known about how they protect their skin or how often they develop light-related skin problems. Using data from a large national child cohort in Japan, this study shows how common sun-protective behaviours are in young children and identifies family and social factors that influence these behaviours. It also reveals that some children develop skin symptoms after sun exposure without receiving a diagnosis. These findings can help parents, schools and healthcare providers improve early education, awareness and support for safe sun exposure in childhood.
Protection of the skin against sunlight is increasingly recognized as a major public health issue, as the risks of ultraviolet (UV) exposure have become increasingly apparent. It is well documented that UV exposure can cause sunburn, immunosuppression, premature aging and skin cancer. Children and adolescents may be vulnerable to increased UV exposure and have a greater risk of subsequent sun-related pathologies. It was reported that 25–50 % of the cumulative UV exposure by 60 years of age occurs during childhood because children have up to 3 times the annual sun exposure of adults (1). According to the protocol published by the World Health Organization (WHO), photoprotection is achieved by avoiding the sun, staying in the shade, wearing clothes that block sunlight, hats and sunglasses, and using sunscreen (2, 3).
Photosensitivity can be defined as an abnormal or adverse skin reaction to UV or visible light exposure. The incidence of photosensitivity disorders in the paediatric population is much lower than that in adults (4, 5). However, there is no information regarding attitudes towards sun exposure or photosensitivity in Japanese children.
The Japan Environment and Children’s Study (JECS) is a nationwide government-funded birth cohort study that evaluated the impact of certain environmental factors on child health and development. In this study, an additional survey was conducted with children and mothers who had visited the JECS survey’s 2nd-grade school-age test to examine sun protection behaviour, its related factors and the incidence of photosensitivity in Japanese children. This is the first study to assess behaviours related to sun exposure and photosensitivity among Japanese children in the 2nd grade of elementary school (age 7–8 years).
MATERIALS AND METHODS
Study design and population
This study was conducted in the Toyama Prefecture as part of an adjunct survey accompanying a 2nd-grade elementary school-age examination within the JECS. The JECS protocol has been previously described in detail elsewhere (6, 7). Briefly, the JECS is a nationwide government-funded birth cohort study aimed at evaluating the impact of environmental factors on children’s health and development. This study involved 15 Japanese regional unit centres (UCs), including the Toyama UC. Pregnant participants were recruited between January 2011 and March 2014, with 5,384 live births recorded in the Toyama UC. The cohort is currently being followed up longitudinally.
The 2nd-grade elementary school-age examination is a face-to-face assessment conducted when children enter the second grade of elementary school. During this assessment, Toyama UC administered an original questionnaire survey as part of a school-age adjunct study. The participants in this study included 1,862 mother–child pairs who participated in the adjunct survey. After excluding 132 cases due to multiple participation (including multiple registrations or multiple births) and 39 cases due to missing data (except for annual household income), 1,691 mother–child pairs were included in the final analysis.
The study protocol was approved by the Japanese Ministry of the Environment’s Institutional Review Board on Epidemiological Studies and Ethics Committees of the University of Toyama (R2019015). All the participants provided their written informed consent.
Self-administered questionnaires
The self-administered questionnaires used in this study are presented in Table I. The questionnaires were completed by mothers. Briefly, the questions were as follows: (1) How does your child’s skin respond to sunlight? (2) Do you use sunscreen when going out for a long time? (3) Do you use sun protection when swimming in an outdoor pool? (4) After exposure to the sun, does the skin in the area of exposure become prickly, itchy, blistered or red and swollen? (5) At what age did the onset of skin symptoms after sun exposure occur? (6) In what months do skin symptoms most commonly appear after exposure to sunlight? (7) Have you ever been diagnosed with a photosensitivity disorder by your healthcare provider? (8) Have you ever been diagnosed with any of the following diseases and medical conditions (solar urticaria, PLE, EPP or porphyria, hydroa vacciniforme, oculocutaneous albinism, xeroderma pigmentosum, or others)? The item on sunscreen use when going out for a long time did not specify a fixed duration in the questionnaire. In addition, skin symptoms after sun exposure were assessed as a binary variable (yes/no) and were not graded for severity.
Table I. Self-administered questionnaire
| Question | Answer |
|---|---|
| How does your child’s skin respond to the sunlight? | Usually, burns and tans minimally, sometimes mild burn and tans uniformly, or burns minimally and always tans well. |
| Do you use sunscreen when going out for a long time? | Always, sometimes or never. |
| Do you use any sun protection when swimming in an outdoor pool? | Sunscreen, rash guard, T-shirt or never. |
| After exposure to the sun, does the skin in the area of exposure become prickly, itchy, blistered, or red and swollen? | Yes or no. |
| At what age did the onset of skin symptoms after sun exposure occur? | From 0–8 years old. |
| In what months do skin symptoms most commonly appear after exposure to sunlight? | From January to December. |
| Have you ever been diagnosed with photosensitivity disorder by your healthcare providers? | Yes or no. |
| Have you ever been diagnosed with any of the following diseases and medical conditions? | Solar urticaria, polymorphous light eruption, erythropoietic protoporphyria or porphyria, hydroa vacciniforme, oculocutaneous albinism, xeroderma pigmentosum or others. |
Evaluation of covariates
The covariates considered in this study were as follows: Fitzpatrick skin phototype (II, III or IV); skin symptoms after sun exposure (yes or no); annual household income (<4 million, 4–6 million, or >6 million JPY); mother’s highest educational level (1, junior high or high school; 2, technical junior college, technical/vocational college or associate degree; or 3, bachelor or postgraduate degree); father’s highest educational level (1, junior high or high school; 2, technical junior college, technical/vocational college, or associate degree; or 3, bachelor or postgraduate degree); maternal age during pregnancy (<25, 25–29, 30–34, or ≥35 years); previous deliveries (nulliparous or multiparous); sex of the child (male or female). Fitzpatrick skin phototype and skin symptoms after sun exposure were obtained from the self-administered questionnaire completed by mothers, whereas the remaining covariates were sourced from the dataset jecs-qa-20210401.
Statistical analysis
Data are expressed as the mean±SD or frequency (percentage), unless stated otherwise. To estimate the association between photosensitivity, socioeconomic and demographic factors and sun protection behaviour, we performed a multivariable logistic regression analysis to calculate ORs and 95 % CIs, with adjustment for all of the covariates mentioned in the Evaluation of covariates section. A complete case analysis was conducted, with missing data on annual household income included in the model as dummy coded variables labelled as “missing.” Two-sided p-values of <0.05 were considered to indicate statistical significance. All statistical analyses were performed using SAS (ver. 9.4; SAS Institute Inc., Cary, NC, USA).
To minimize potential sources of bias, standardized questionnaires and predefined inclusion and exclusion criteria were used, and potential confounders were adjusted for in the multivariable analyses.
RESULTS
A total of 1,862 mother–child pairs participated in the adjunct survey; after excluding 171 participants because of duplicate participation or missing data, 1,691 mother–child pairs were included in the final analysis. The self-administered questionnaires used in this study are presented in Table I. In the child population, 49.1 % of the children were male and 46.2 % were first-born children (Table II). The most common maternal age was 30–34 years (37.5%). The characteristics of the children and parents are summarized in Table II. The most common Fitzpatrick skin phototype was III (60.4%) (Table III). Among the enrolled children, 6.4 % developed any skin symptoms (e.g. prickly/tingling, itchy, blistered, erythematous or swollen skin) after sun exposure. The responses indicated that 6.7 % of the children always used sunscreen when they went out for a long time, 60.8 % sometimes used sunscreen when they went out for a long time. At outdoor pools, 38.7 % used sunscreen, 59.1 % used a rash guard and 6.8 % used a T-shirt; overall, 73.2 % used at least one sun-protective measure.
Table II. Participant characteristics (N=1,691)
| Variable | n | (%) | |
|---|---|---|---|
| Sex of the child | Male | 830 | (49.1) |
| Female | 861 | (50.9) | |
| Annual household income (JPY) | <4 million | 351 | (20.8) |
| 4–<6 million | 568 | (33.6) | |
| ≥6 million | 665 | (39.3) | |
| Missing | 107 | (6.3) | |
| Mother’s highest educational level | ≤12 | 371 | (21.9) |
| (years) | >12–<16 | 769 | (45.5) |
| ≥16 | 551 | (32.6) | |
| Father’s highest educational level | ≤12 | 560 | (33.1) |
| (years) | >12–<16 | 357 | (21.1) |
| ≥16 | 774 | (45.8) | |
| Maternal age (years) | <25 | 79 | (4.7) |
| 25–29 | 447 | (26.4) | |
| 30–34 | 631 | (37.3) | |
| ≥35 | 534 | (31.6) | |
| Previous deliveries | 0 | 782 | (46.2) |
| 1 | 659 | (39.0) | |
| >2 | 250 | (14.8) |
Table III. Response to sun exposure and sun protection behaviours (N=1,691)
| Variable | n | (%) | |
|---|---|---|---|
| Fitzpatrick skin phototype | II | 315 | (18.6) |
| III | 1,022 | (60.4) | |
| IV | 354 | (20.9) | |
| Skin symptoms after sun exposure | No | 1,583 | (93.6) |
| Yes | 108 | (6.4) | |
| Use of sunscreen when going out for a long time | Always | 114 | (6.7) |
| Sometimes | 1,028 | (60.8) | |
| Never | 549 | (32.5) | |
| Sun protection when swimming in an outdoor pool | Sunscreen | 655 | (38.7) |
| Rash guard | 1,000 | (59.1) | |
| T-shirt | 115 | (6.8) | |
| Never | 522 | (32.6) |
The use of sunscreen when going out for a long time was significantly related to Fitzpatrick skin phototypes II and III (adjusted odds ratio [OR]: 1.80, 95 % confidence interval [CI]: 1.28–2.54; adjusted OR: 1.46, 95% CI: 1.12–1.89), annual household income (adjusted OR: 1.37, 95% CI: 1.02–1.85), father’s highest educational level (adjusted OR: 1.52, 95% CI: 1.17–1.98) and female sex (adjusted OR: 2.72, 95% CI: 2.19–3.37) (Table IV).
Table IV. Use of sunscreen when going out for a long time (N=1,691)
| Yes (n=1,142) n (%) | No (n=549) n (%) | Crude model OR (95% CI) | Adjusted model* OR (95% CI) | ||
|---|---|---|---|---|---|
| Fitzpatrick skin phototype | II | 228 (20.0) | 87 (15.9) | 1.71 (1.24–2.38) | 1.80 (1.28–2.54) |
| III | 700 (61.3) | 322 (58.7) | 1.42 (1.11–1.83) | 1.46 (1.12–1.89) | |
| IV | 214 (18.7) | 140 (25.5) | ---- | ---- | |
| Skin symptoms after sun exposure | No | 1,063 (93.1) | 520 (94.7) | ---- | ---- |
| Yes | 79 (6.9) | 29 (5.3) | 1.33 (0.86–2.07) | 1.32 (0.83–2.08) | |
| Annual household income (JPY) | <4 million | 220 (19.3) | 131 (23.9) | ---- | ---- |
| 4–<6 million | 377 (33.0) | 191 (34.8) | 1.18 (0.89–1.55) | 1.16 (0.87–1.57) | |
| ≥6 million | 473 (41.4) | 192 (35.0) | 1.47 (1.12–1.93) | 1.37 (1.02–1.85) | |
| Missing value | 72 (6.3) | 35 (6.4) | 1.23 (0.78–1.94) | 1.08 (0.67–1.76) | |
| Mother’s highest educational level | ≤12 | 246 (21.5) | 125 (22.8) | ---- | ---- |
| (years) | >12–<16 | 520 (45.5) | 249 (45.4) | 1.06 (0.82–1.38) | 0.91 (0.68–1.20) |
| ≥16 | 376 (32.9) | 175 (31.9) | 1.09 (0.83–1.45) | 0.79 (0.57–1.10) | |
| Father’s highest educational level | ≤12 | 361 (31.6) | 199 (36.3) | ---- | ---- |
| (years) | >12–<16 | 226 (19.8) | 131 (23.9) | 0.95 (0.72–1.25) | 1.04 (0.77–1.39) |
| ≥16 | 555 (48.6) | 219 (39.9) | 1.40 (1.11–1.76) | 1.52 (1.17–1.98) | |
| Maternal age (years) | <25 | 51 (4.5) | 28 (5.1) | ---- | ---- |
| 25–29 | 283 (24.8) | 164 (29.9) | 0.95 (0.58–1.56) | 0.90 (0.53–1.53) | |
| 30–34 | 440 (38.5) | 191 (34.8) | 1.27 (0.77–2.07) | 1.12 (0.66–1.91) | |
| ≥35 | 368 (32.2) | 166 (30.2) | 1.22 (0.74–2.00) | 1.08 (0.63–1.86) | |
| Previous deliveries | 0 | 517 (45.3) | 265 (48.3) | ---- | ---- |
| 1 | 454 (39.8) | 205 (37.3) | 1.14 (0.91–1.42) | 1.11 (0.88–1.41) | |
| >2 | 171 (15.0) | 79 (14.4) | 1.11 (0.82–1.51) | 1.01 (0.73–1.41) | |
| Sex of the child | Male | 473 (41.4) | 357 (65.0) | ---- | ---- |
| Female | 669 (58.6) | 192 (35.0) | 2.63 (2.13–3.25) | 2.72 (2.19–3.37) | |
|
––– represents the reference. Bold indicates statistical significance (p < 0.05). * All ORs and 95%CIs were adjusted for all factors shown in Table III. |
|||||
The use of sun protection when swimming in an outdoor pool was significantly related to Fitzpatrick skin phototypes II and III (adjusted OR: 1.49, 95% CI: 1.05–2.12; adjusted OR: 1.47, 95% CI: 1.13–1.92), skin symptoms after sun exposure (adjusted OR: 2.75, 95% CI: 1.51–5.01), maternal age of 25–29 years (adjusted OR: 1.82, 95% CI: 1.07–3.10) and female sex (adjusted OR: 1.88, 95% CI: 1.51–2.35) (Table V). The use of sunscreen when swimming in an outdoor pool was significantly related to skin symptoms after sun exposure (adjusted OR: 2.14, 95% CI: 1.42–3.23) and female sex (adjusted OR: 2.45, 95% CI: 1.99–3.00) (Table SI). Fitzpatrick skin phototype II seemed to be related to the use of sunscreen when swimming in an outdoor pool in a crude analysis; however, no significance was recognized in the adjusted analysis. The use of a rash guard when swimming in an outdoor pool was significantly related to Fitzpatrick skin phototypes II and III (adjusted OR: 1.54, 95% CI: 1.12–2.11; adjusted OR: 1.43, 95% CI: 1.12–1.83), maternal age >25 years (adjusted OR: 2.75, 95% CI: 1.65–4.58; adjusted OR: 2.39, 95% CI: 1.44–3.98; adjusted OR: 2.34, 95% CI: 1.40–3.92) and female sex (adjusted OR: 1.37, 95% CI: 1.13–1.67) (Table SII). No factor was related to the use of a T-shirt when swimming in an outdoor pool (Table SIII).
Table V. Prevalence of sun protection when swimming in an outdoor pool (N=1,691)
| Yes (n=1,238) n (%) | No (n=453) n (%) | Crude model OR (95% CI) | Adjusted model* OR (95% CI) | ||
|---|---|---|---|---|---|
| Fitzpatrick skin phototype | II | 238 (19.2) | 77 (17.0) | 1.55 (1.10–2.17) | 1.49 (1.05–2.12) |
| III | 764 (61.7) | 258 (57.0) | 1.48 (1.14–1.93) | 1.47 (1.13–1.92) | |
| IV | 236 (19.1) | 118 (26.1) | ---- | ---- | |
| Skin symptoms after sun exposure | No | 1,143 (92.3) | 440 (97.1) | ---- | ---- |
| Yes | 95 (7.7) | 13 (2.9) | 2.81 (1.56–5.07) | 2.75 (1.51–5.01) | |
| Annual household income (JPY) | <4 million | 250 (20.2) | 101 (22.3) | ---- | ---- |
| 4–<6 million | 418 (33.8) | 150 (33.1) | 1.13 (0.84–1.52) | 1.12 (0.82–1.52) | |
| ≥6 million | 494 (39.9) | 171 (37.8) | 1.17 (0.87–1.56) | 1.14 (0.83–1.55) | |
| Missing value | 76 (6.1) | 31 (6.8) | 0.99 (0.62–1.60) | 0.96 (0.59–1.58) | |
| Mother’s highest educational level (years) | ≤12 | 262 (21.2) | 109 (24.1) | ---- | ---- |
| >12–<16 | 571 (46.1) | 198 (43.7) | 1.20 (0.91–1.58) | 1.07 (0.80–1.44) | |
| ≥16 | 405 (32.7) | 146 (32.2) | 1.15 (0.86–1.55) | 0.95 (0.68–1.34) | |
| Father’s highest educational level (years) | ≤12 | 404 (32.6) | 156 (34.4) | ---- | ---- |
| >12–<16 | 254 (20.5) | 103 (22.7) | 0.95 (0.71–1.28) | 0.99 (0.73–1.34) | |
| ≥16 | 580 (46.9) | 194 (42.8) | 1.15 (0.90–1.48) | 1.18 (0.90–1.55) | |
| Maternal age (years) | <25 | 51 (4.1) | 28 (6.2) | ---- | ---- |
| 25–29 | 343 (27.7) | 104 (23.0) | 1.81 (1.09–3.02) | 1.82 (1.07–3.10) | |
| 30–34 | 461 (37.2) | 170 (37.5) | 1.49 (0.91–2.44) | 1.50 (0.88–2.54) | |
| ≥35 | 383 (30.9) | 151 (33.3) | 1.39 (0.85–2.29) | 1.42 (0.83–2.42) | |
| Previous deliveries | 0 | 584 (47.2) | 198 (43.7) | ---- | ---- |
| 1 | 476 (38.5) | 183 (40.4) | 0.88 (0.70–1.12) | 0.89 (0.70–1.14) | |
| >2 | 178 (14.4) | 72 (15.9) | 0.84 (0.61–1.15) | 0.84 (0.60–1.18) | |
| Sex of the child | Male | 558 (45.1) | 272 (60.0) | ---- | ---- |
| Female | 680 (54.9) | 181 (40.0) | 1.83 (1.47–2.28) | 1.88 (1.51–2.35) | |
|
––– represents the reference. Bold indicates statistical significance (p < 0.05). * All ORs and 95%CIs were adjusted for all factors shown in Table IV. |
|||||
Based on the questionnaire, 108 children (6.4%) experienced skin symptoms after sun exposure, and the age at the onset of skin symptoms was 5.04±1.81 years (Fig. 1a). Skin symptoms after sun exposure mainly developed from April to October (Fig. 1b). Eight (0.5%) of the 108 children had been diagnosed with photosensitivity disorders by their healthcare providers. The age at the diagnosis of photosensitivity disorder was 3.00±2.08 years (Fig. 1c). Of the 8 patients, 1 had ongoing symptoms, 3 had no current symptoms, and 4 did not respond to the question. Four children (50%) were diagnosed with polymorphous light eruption (PLE) and 2 (25%) were diagnosed with solar urticaria (Fig. 1d). The others (5.9%) were undiagnosed, despite developing skin symptoms after sun exposure.
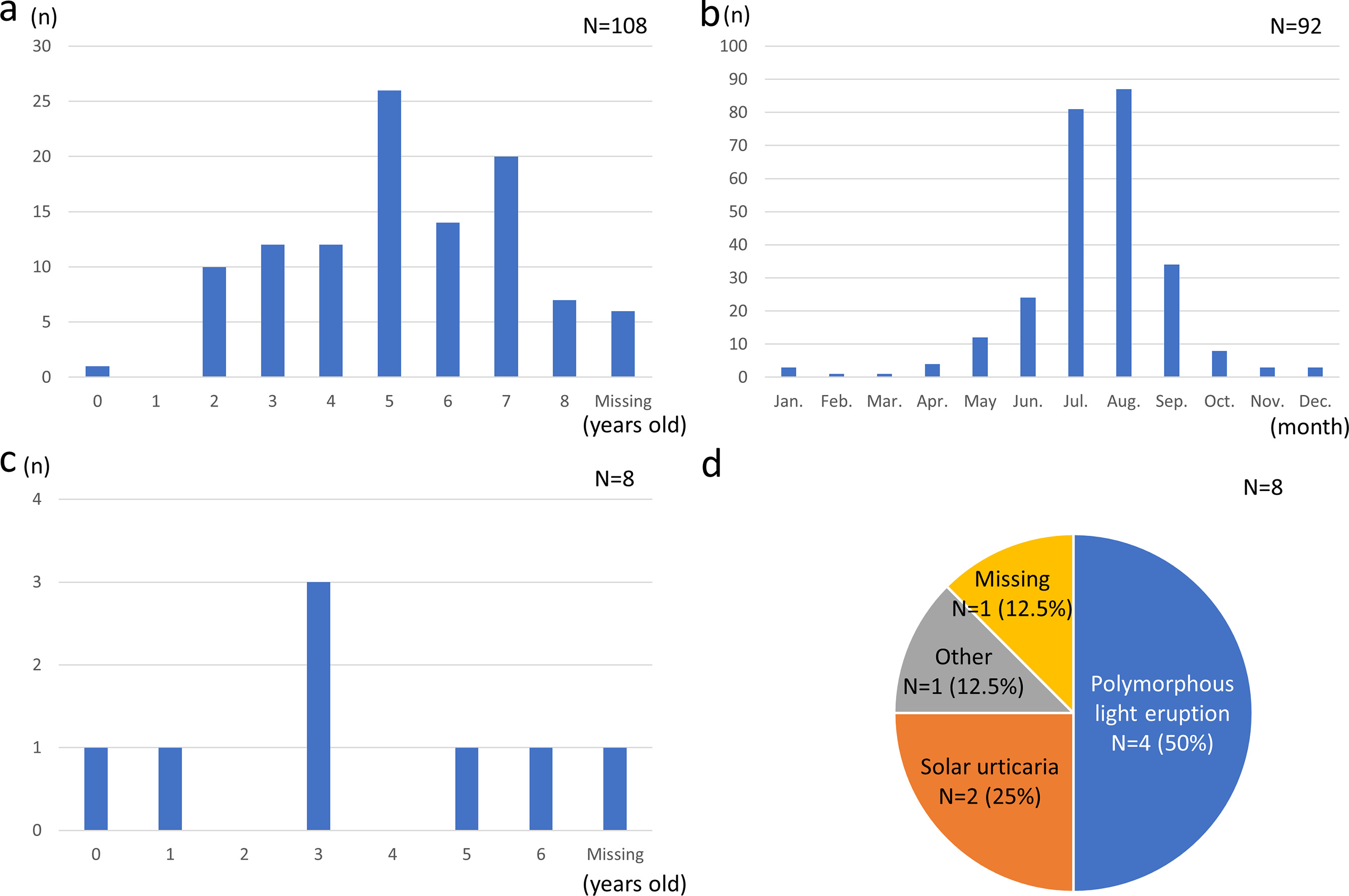
Fig. 1. (A) The age at the onset of skin symptoms after sun exposure. (B) The month in which skin symptoms appear after sun exposure. (C) The age at the diagnosis of photosensitivity. (D) Diagnosis of photosensitivity disorder made by healthcare providers.
DISCUSSION
We found that 67.5 % of children always or sometimes used sunscreen when going out for a long time, while 73.2 % used at least one sun-protective measure when swimming in an outdoor pool. Sun protection behaviours were associated with sex, Fitzpatrick skin phototypes, skin symptoms after sun exposure, annual household income, father’s highest educational level and maternal age. Photosensitivity disorders were diagnosed in 0.5 % of children, whereas 5.9 % had skin symptoms after sun exposure but remained undiagnosed.
Behaviours related to sun exposure
Sun protection behaviours are influenced by age, sex, Fitzpatrick skin phototype, culture, socioeconomic status and geography (8, 9). In the present study, 67.5 % of the children always or sometimes used sunscreen when going out for a long time. A previous Japanese study reported sunscreen use in 45.7 % of adults (8). A previous study suggested that, relative to Europeans and North Americans, Japanese individuals may be more cautious about sun-induced hazards and may consider lighter skin more attractive (8). Iranian studies reported that approximately 80 % of children used some form of sun protection, most commonly long-sleeved clothing or hats (9, 10). Because Japanese children do not usually wear long sleeves in summer, we excluded this item when assessing sun protection during prolonged outdoor exposure. Rash guards were the most common method of sun protection, and the use of sunscreen was the second most common method when swimming in an outdoor pool. This may partly reflect the ready availability and convenience of rash guards in Japan, as rash guards provide continuous body coverage and, unlike sunscreen, do not require reapplication. However, we did not assess whether children used rash guards and sunscreen in combination during longer periods of sun exposure, nor did we collect information on the frequency of sunscreen reapplication.
The present study showed that the Fitzpatrick skin phototype and skin symptoms after sun exposure influenced sun protection behaviours. A previous report also showed differences in sunscreen utilization, with Fitzpatrick skin phototypes I-III, which tend to sunburn rather than tan, using more sunscreen than Fitzpatrick skin phototypes IV-VI, which tend to tan rather than sunburn (11, 12). For children developing skin symptoms after sun exposure, it is reasonable to perform strict sun protection.
Female sex was significantly associated with sunscreen use when going out for a long time and with sun protection when swimming in an outdoor pool. A previous Japanese study also found greater sunscreen use among females than among males (8). Approximately 54 % of respondents said they stayed in the shade to protect themselves from the sun. This behaviour was more prevalent among females (67.05%) and fair-skinned individuals (56.13%) (8). In addition, there was a significant difference between males and females regarding the appeal of tanned skin: 54.95 % of males perceived a tan as an aesthetic asset versus only 34.67 % (p<0.001) of females (8). Similarly, an Iranian study reported higher sun-protection knowledge, attitudes and behaviours among females (13), consistent with our findings. Girls or their parents may therefore be more likely to take precautions against sun exposure. A sociocultural preference for lighter skin in Japan may also partly influence these behaviours; however, our study did not directly assess parental attitudes toward tanning or lighter skin, and therefore, the magnitude of this effect could not be determined.
This study suggested that annual household income and father’s highest educational level were significantly associated with the use of sunscreen when going out for a long time. It has been reported that sun protection behaviours are more frequent among urban children, children from wealthy families and children whose mothers had higher educational levels or who were employed (9). According to a previous study, students whose parents held university degrees exhibited higher knowledge and practice scores regarding sun-protective measures (14). Higher levels of education and income may be associated with health awareness among parents. Although public educational resources regarding UV protection are available in Japan, their influence was not assessed in the present study. In addition, the affordability of sunscreens and rash guards was not directly assessed.
Furthermore, sun protection and the use of a rash guard when swimming in an outdoor pool were related to maternal age 25–29 years and maternal age >25 years, respectively. Individuals aged >40 years tend to wear a cap and sunglasses and stay in the shade more than those aged <40 years. Females and individuals aged >40 years also expressed greater concern about the risk of skin ageing (8). A significant correlation has been reported between mothers’ sunscreen use and their children’s sun-protection behaviours (9). Although parental sun-protection behaviour may have influenced children’s behaviours in the present study, this factor was not directly assessed in our questionnaire. These findings suggest that maternal age may be associated with children’s sun-protection behaviours.
Photosensitivity in Japanese children
In this study, 6.4 % of children developed skin symptoms after sun exposure, and the age at the onset of skin symptoms was 5.04±1.81 years. Skin symptoms after sun exposure were observed primarily from April to October. This finding was not unexpected, because this period corresponds to spring through autumn in Japan, when UV exposure and outdoor activity are generally increased. Although Toyama, the prefecture in which this study was performed, is an area with relatively low sunshine hours throughout the year, there are times when the monthly sunshine hours exceed 140 h during the period from April to October (Fig. S1). Thus, the seasonal concentration of symptoms is consistent with increased daylight hours and more time spent outdoors during these months.
Photosensitivity disorders are much less common in children than in adults (15). PLE is the most common paediatric photosensitivity disorder, followed by erythropoietic protoporphyria (EPP) (5), and it was also the most common diagnosis in our study. Previous studies from Scotland, Finland and Hungary have shown that PLE often begins before adolescence (5, 16, 17, 18).
EPP is the second most common paediatric photosensitivity disorder (5). No children were diagnosed with EPP in this study. Although 108 children (6.4%) developed any skin symptoms after sun exposure, 100 children (5.9%) were undiagnosed. We previously reported EPP with a mild phenotype in the Japanese population as incomplete EPP (19). Patients with incomplete EPP have mild photosensitivity associated with a slight increase in their PP concentration, the formation of a small number of fluorocytes and no FECH gene mutations. Homozygous IVS3-48C polymorphism may cause a mild EPP phenotype via a slight increase in protoporphyrin levels. Japanese EPP shows a characteristic phenotype, with a late onset and mild symptoms relative to Caucasian EPP (20, 21). The median age at the onset of EPP in Japanese patients was 6 years (20). In the present study, the age at the onset of skin symptoms after sun exposure was 5.04±1.81 years, which is close to the age at the onset of EPP in the Japanese population. The frequency of the homozygous IVS3-48C polymorphism in the Japanese population is more than 10 times higher than that in individuals from European countries (22). Skin symptoms after sun exposure may represent non-pathological sunburn; however, there may have been patients with incomplete EPP among the 100 children who remained undiagnosed.
Strengths and limitations of this study
The strengths of the present study are that it is the first to investigate sun protection behaviour, its related factors and the incidence of photosensitivity among Japanese children in the second grade of elementary school. In addition, the study was conducted within a well-characterized birth cohort. However, the present study had some limitations. First, this was an observational study, and unmeasured residual factors may have confounded the results. Second, because the study relied on a self-administered questionnaire completed by mothers, reporting bias and misclassification were possible. In particular, the questionnaire item regarding “going out for a long time” was not defined by a fixed duration, and skin symptoms after sun exposure were assessed as a yes/no item without severity grading; therefore, subtle erythema may have been underreported. Third, Fitzpatrick skin phototype was based on questionnaire responses rather than investigator assessment. Fourth, we did not collect information on environmental or behavioural factors such as shade availability, local rules for outdoor swimming pools, sunscreen reapplication, combined use of rash guards and sunscreen, or parental attitudes and behaviours regarding sun exposure. Finally, because the study was conducted in one regional city, the participants may not be considered representative of children in Japan.
Conclusion
Six factors were associated with sun-protection behaviours. Photosensitivity disorders were diagnosed in 0.5 % of children, whereas 5.9 % had skin symptoms after sun exposure but remained undiagnosed. Some children may have undiagnosed incomplete EPP. Further investigation is required to confirm these findings.
ACKNOWLEDGEMENTS
We are grateful to all the JECS participants and all individuals involved in data collection. The JECS was funded by the Ministry of Environment, Japan. The findings and conclusions of this article are solely the responsibility of the authors and do not represent the official views of the Ministry of Environment, Japan.
REFERENCES
- Cestari T, Buster K. Photoprotection in specific populations: children and people of color. J Am Acad Dermatol 2017; 76: S110–S121. https://doi.org/10.1016/j.jaad.2016.09.039
- Burnett ME, Hu JY, Wang SQ. Sunscreens: obtaining adequate photoprotection. Dermatol Ther 2012; 25: 244–251. https://doi.org/10.1111/j.1529-8019.2012.01503.x
- Thoonen K, Osch L van, Vries H de, Jongen S, Schneider F. Are environmental interventions targeting skin cancer prevention among children and adolescents effective? A systematic review. Int J Environ Res Public Health 2020; 17: 529. https://doi.org/10.3390/ijerph17020529
- ten Berge O, Sigurdsson V, Bruijnzeel-Koomen CAFM, van Weelden H, Pasmans SGMA. Photosensitivity testing in children. J Am Acad Dermatol 2010; 63: 1019–1025. https://doi.org/10.1016/j.jaad.2009.12.032
- Horkay I, Emri G, Varga V, Simics E, Remenyik E. Photosensitivity skin disorders in childhood. Photodermatol Photoimmunol Photomed 2008; 24: 56–60. https://doi.org/10.1111/j.1600-0781.2008.00341.x
- Kawamoto T, Nitta H, Murata K, Toda E, Tsukamoto N, Hasegawa M, et al. Rationale and study design of the Japan Environment and Children’s Study (JECS). BMC Public Health 2014; 14: 25. https://doi.org/10.1186/1471-2458-14-25
- Michikawa T, Nitta H, Nakayama SF, Yamazaki S, Isobe T, Tamura K, et al. Baseline profile of participants in the Japan Environment and Children’s Study (JECS). J Epidemiol 2018; 28: 99–104. https://doi.org/10.2188/jea.JE20170018
- Morita A, Lim HW, Passeron T, Goh CL, Kang HY, Ly F, et al. Attitudes and behaviors regarding sun exposure in Japan compared to Europe and North America. J Dermatol 2024; 51: 1004–1009. https://doi.org/10.1111/1346-8138.17217
- Nouri N, Iravani P, Abtahi-Naeini B. Sun protection behaviors among children aged 6-18 years old, the role of socioeconomic factors: A cross-sectional study. Health Sci Rep 2023; 6: e1727. https://doi.org/10.1002/hsr2.1727
- Moshki M, Karimy M, Asl HR, Mojadam M, Araban M. Predictors of sun protection behavior in high school students of Ahvaz: a cross-sectional study. Int J Adolesc Med Health 2020; 32: 20180006. https://doi.org/10.1515/ijamh-2018-0006
- Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol 1988; 124: 869–871. https://doi.org/10.1001/archderm.124.6.869
- Ullman LE, Nasir-Moin M, Hoffman V, Ghadersohi S, Swartzman I, de Weever M, et al. Sunscreen use and affordability attitudes based on ethnicity, socioeconomic status, and Fitzpatrick skin type. Arch Dermatol Res 2024; 316: 266. https://doi.org/10.1007/s00403-024-02997-z
- Janjani H, Nedjat S, Yunesian M, Nasseri S, Haghighat Doost F, Nabizadeh R. Sun exposure and health safety practices of high school students in an urban population of Iran. BMC Public Health 2019; 19: 1736. https://doi.org/10.1186/s12889-019-8100-7
- Taheri F, Salehpour M, Taheri AR. Survey of knowledge, attitude, and practice of female high school students regarding sun protection in Birjand, Iran. Mod Care J 2022; 19: e122395. https://doi.org/10.5812/modernc-122395
- Chantorn R, Lim HW, Shwayder TA. Photosensitivity disorders in children: part I. J Am Acad Dermatol 2012; 67: 1093. https://doi.org/10.1016/j.jaad.2012.07.033
- Frain-Bell W. Idiopathic photodermatoses. In: Frain-Bell W, editor. Cutaneous photobiology. Oxford: Oxford University Press; 1985: p. 51–55.
- Ferguson J. Polymorphic light eruption and actinic prurigo. Curr Probl Dermatol 1990; 2: 127–147. https://doi.org/10.1016/S1040-0486(10)80001-8
- Jansén CT. Photosensitivity in childhood. Acta Derm Venereol Suppl 1981; 95: 54–57.
- Mizawa M, Makino T, Nakano H, Sawamura D, Shimizu T. Incomplete erythropoietic protoporphyria caused by a splice site modulator homozygous IVS3-48C polymorphism in the ferrochelatase gene. Br J Dermatol 2016; 174: 172–175. https://doi.org/10.1111/bjd.14078
- Mizawa M, Makino T, Nakano H, Sawamura D, Shimizu T. Erythropoietic protoporphyria in a Japanese population. Acta Derm Venereol 2019; 99: 634–639. https://doi.org/10.2340/00015555-3184
- Mizawa M, Hara H, Makino T, Shimizu T. Clinical, genetic, and epidemiological findings of erythropoietic protoporphyria in Japan. Photomed Photobiol 2023; 43, 44: 7–16.
- Nakano H, Nakano A, Toyomaki Y, Ohashi S, Harada K, Moritsugu R, et al. Novel ferrochelatase mutations in Japanese patients with erythropoietic protoporphyria: high frequency of the splice site modulator IVS3-48C polymorphism in the Japanese population. J Invest Dermatol 2006; 126: 2717–2719. https://doi.org/10.1038/sj.jid.5700456