ORIGINAL REPORT
Prognostic Factors for Long-term Aesthetic Outcome of Infantile Haemangioma Treated with Beta-blockers
Mireille M. HERMANS1, Suzanne G. M. A. PASMANS1#, Marlies DE GRAAF2#, Aviël RAGAMIN1, Elodie J. MENDELS1, Johannes M. P. J. BREUR3, Hester R. LANGEVELD4, Martine F. RAPHAEL2,5, Peter C. J. DE LAAT6, Saskia N. DE WILDT7, André B. RIETMAN8, Corstiaan C. BREUGEM9,10* and Renske SCHAPPIN1,11*
1Erasmus MC Sophia Children’s Hospital, University Medical Center Rotterdam, Department of Dermatology – Center of Pediatric Dermatology, Center of Rare Skin Diseases, Vascular Anomaly Center Erasmus MC Rotterdam, member of the ERN-SKIN-Mosaic group and ERN-VASCERN-VASCA group, 2Department of Dermatology, Wilhelmina Children’s Hospital, University Medical Center Utrecht, UMC Utrecht Center for Vascular Anomalies, 3Department of Pediatric Cardiology, Wilhelmina Children’s Hospital, University Medical Center Utrecht, Utrecht, 4Erasmus MC Sophia Children’s Hospital, University Medical Center Rotterdam, Department of Intensive Care and Pediatric Surgery, Center of Rare Skin Diseases, Vascular Anomaly Center Erasmus MC Rotterdam, member of the ERN-SKIN-Mosaic group and ERN-VASCERN-VASCA group, 5Department Emma Children’s Hospital, Amsterdam UMC Location University of Amsterdam, Amsterdam, 6Erasmus MC Sophia Children’s Hospital, University Medical Center Rotterdam, Department of Pediatrics (-Hemato-oncology), Center of Rare Skin Diseases, Vascular Anomaly Center Erasmus MC Rotterdam, member of the ERN-SKIN-Mosaic group and ERN-VASCERN-VASCA group, Rotterdam, 7Department of Pharmacology and Toxicology, Radboud Institute for Health Sciences, Radboud University Medical Center, Nijmegen, 8Erasmus MC Sophia Children’s Hospital, University Medical Center Rotterdam, Department of Child and Adolescent Psychology/Psychiatry, Rotterdam, 9Department of Plastic Surgery, Wilhelmina Children’s Hospital, University Medical Center Utrecht, UMC Utrecht Center for Vascular Anomalies, Utrecht, 10Department of Reconstructive and Hand Surgery, Amsterdam UMC, University of Amsterdam, Amsterdam, and 11Department of Surgery, Wilhelmina Children’s Hospital, University Medical Center Utrecht, Utrecht, the Netherlands. #Contributed equally as co-second authors. *Contributed equally as co-last authors.
Parents of infants treated with beta-blockers for infantile haemangioma are often concerned about the long-term aesthetic outcome. This cross-sectional study assessed the influence on the long-term aesthetic outcome of characteristics of the infantile haemangioma, the beta-blocker treatment, and the infant. The study included 103 children aged 6–12 years, treated with beta-blockers (propranolol or atenolol) for infantile haemangioma during infancy (age at treatment initiation ≤1 year) for ≥6 months. Dermatologists and parents scored the Patient Observer Scar Assessment Scale, and the child scored a visual analogue scale. Dermatologists identified whether telangiectasia, fibrofatty tissue, and atrophic scar tissue were present. The long-term aesthetic outcome of infantile haemangioma was judged more negatively by dermatologists and parents in case of a superficial component, ulceration, older age at treatment initiation, higher cumulative dose, and/or shorter follow-up time. According to children, infantile haemangioma located on the head had better aesthetic outcome than infantile haemangioma located elsewhere. Close monitoring, particularly of infantile haemangioma with a superficial component, is essential for early initiation of treatment, and to prevent or treat ulceration. These outcome data can support parental counselling and guide treatment strategy.
Key words: vascular tissue neoplasm; adrenergic beta-antagonist; infant; aesthetics; cicatrix; telangiectasis.
SIGNIFICANCE
Parents of infants receiving beta-blockers for infantile haemangioma commonly ask their physician about the aesthetics of infantile haemangioma at school age. This study evaluated which characteristics during beta-blocker treatment are associated with long-term (≥6 years) aesthetic outcome. Negative aesthetics were found for children with infantile haemangioma with a superficial component, ulcerated infantile haemangioma, older age at treatment initiation, higher cumulative dose, and shorter follow-up time. As this study considered the perspectives of dermatologists, parents, and children in multivariable analyses, the results readily apply to clinical practice. Using this information, physicians can inform parents and assess whether additional treatment (e.g. surgery or laser treatment) is needed.
Citation: Acta Derm Venereol 2023; 103: adv5286. DOI: https://doi.org/10.2340/actadv.v103.5286.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Dec 16, 2023; Published: Aug 30, 2023
Corr: Suzanne G. M. A. Pasmans, Erasmus MC Sophia Children’s Hospital, University Medical Center Rotterdam, Department of Dermatology – Center of Pediatric Dermatology, Doctor Molewaterplein 40, NL-3015 GD Rotterdam, The Netherlands. E-mail: s.pasmans@erasmusmc.nl
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Infantile haemangiomas (IH), the most common benign tumours of childhood, occur in approximately 2.0–4.5% of infants (1–3). IH typically show rapid growth during the first months of infancy (proliferative phase), before transitioning into spontaneous and slow involution (involution phase (4–6). Although the involution phase can continue throughout childhood, most involution occurs before the age of 3.5 years (6).
A substantial proportion of IH are treated systemically to prevent or treat complications, such as ulceration, functional impairment, or aesthetic disfigurement (7). Since 2008, oral beta-blockers have been the first-choice treatment worldwide for these complicated IH (8–10). Despite effective beta-blocker treatment and spontaneous involution, IH may still leave long-standing sequelae, such as telangiectasia, fibrofatty tissue, and atrophic scar tissue (11). Parents are often concerned about the psychosocial consequences of visible sequelae at school age, potential stigmatization and bullying (12). These concerns may guide treatment strategies, such as additional surgical excision or laser treatment of the residual lesion. In order to optimise parent counselling and guide treatment strategies, it is important to know which factors during infancy are associated with long-term aesthetic outcome.
Recently, we have demonstrated that the type of beta-blocker treatment (propranolol or atenolol) is not related to long-term aesthetic outcome (13). Previous studies have shown that aesthetic outcome is influenced by IH characteristics (4, 11, 14–18). IH with a superficial component, ulcerated IH, and IH located on the scalp, forehead, cheeks and parotid area are at risk of more long-term sequelae and poor aesthetic outcome (4, 11, 14–18). Segmental IH are more likely to cause complications and require treatment compared with focal or indeterminate IH, but the long-term aesthetic outcome has not yet been studied (5, 7, 16, 17). These studies either involved untreated IH or did not consider the aspect of variations in treatment-related variables (e.g. treatment dose, age at treatment initiation) or demographic variables (e.g. skin type, sex, socioeconomic status). Furthermore, aesthetic outcome was mainly assessed by physicians, and the opinions of the child or parents were not taken into account.
The aim of this study was to assess the association of characteristics of the IH (location, type, pattern, size, ulceration), the beta-blocker treatment (beta-blocker type, cumulative dose, age at treatment initiation, follow-up time), and the child (sex, skin type, maternal educational level) with the long-term aesthetic outcome of IH after beta-blocker treatment, as judged by the dermatologist, parents and the child.
MATERIALS AND METHODS
Design and setting
This study was part of a 2-centre cross-sectional study conducted at the vascular anomaly centres of Erasmus MC, University Medical Center Rotterdam (Erasmus MC, Rotterdam, The Netherlands) and the University Medical Center Utrecht (UMCU, Utrecht, The Netherlands). The study method has been described previously in more detail (13, 19). The study was exempt from the Dutch Medical Research Involving Human Subjects Act according to the Institutional Review Boards of Erasmus MC (MEC-2019-0268) and the UMCU (19-115/C). All parent(s)/guardian(s) provided written informed consent.
Participants
Patient records of children born between 2008 and 2014 (age ≥ 6 years upon participation in the study) and treated for IH with beta-blockers at Erasmus MC or UMCU were screened for participation. Eligible children had been treated for IH with either oral propranolol (dose ≥ 2 mg/kg/day) or oral atenolol (dose ≥ 1 mg/kg/day); had a treatment duration of ≥ 6 months; and were ≤ 1 year old at initiation of beta-blocker treatment. Those children were invited to participate. Children who had received treatment for IH subsequent to the beta-blocker treatment (e.g. laser, surgery, cryotherapy, or oral or intralesional corticosteroids), were excluded from the study. In addition, complete subcutaneous IH, not eligible for clinical scoring, were excluded from the analyses.
Children were actively recruited between April and December 2019, with the last child assessed in March 2020. All participating children underwent a dermatological examination by a paediatric dermatologist (MdG, SP). During this examination, the paediatric dermatologist, 1 of the parents, and the child independently rated the current state of the residual lesion (Figs 1 and 2). A professional photographer took 3 standardised photographs of each residual lesion (1 frontal and 2 sagittal (both sides) views, all with a ruler). Photographs were retrieved from the medical records of the IH prior to beta-blocker treatment, together with information about the clinical characteristics of the patient and of the IH, and details of the beta-blocker treatment.
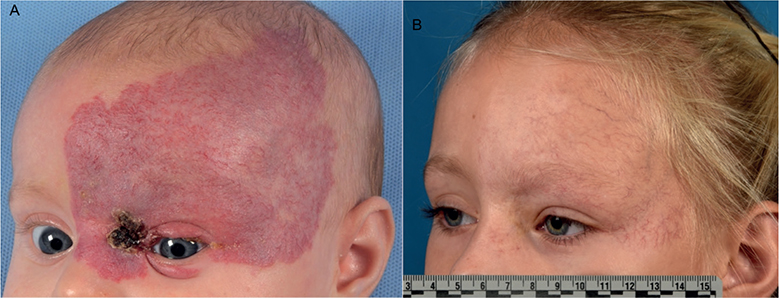
Fig. 1. Photographs of the infantile haemangioma before beta-blocker treatment initiation and at follow-up. (A) Female with a large (>50 mm) ulcerated superficial and segmental infantile haemangioma located on the head, before initiation of beta-blocker treatment (propranolol) at the age of 3 months. (B) The residual lesion was scored during the follow-up visit, when the child was 6 years old. The paediatric dermatologist gave an Observer Scar Assessment Scale score of 5, parents gave a Patient Scar Assessment Scale score of 4, and the child gave a visual analogue scale score of 2. The paediatric dermatologist also determined that telangiectasia were present, and that fibrofatty tissue and atrophic scar tissue were absent. Both parents gave written permission to publish these photographs.
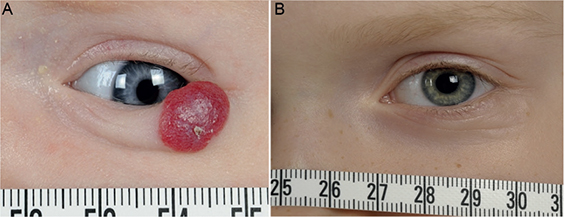
Fig. 2. Photographs of the infantile haemangioma before beta-blocker treatment initiation and at follow-up. (A) A female with a small (< 30 mm) superficial and focal infantile haemangioma with a tendency to ulceration located on the lower eyelid, before initiation of beta-blocker treatment (atenolol) at the age of 2 months. (B) The residual lesion was scored during the follow-up visit, when the child was 7 years old. The paediatric dermatologist gave an Observer Scar Assessment Scale (OSAS) score of 1, parents gave a Patient Scar Assessment Scale (PSAS) score of 2, and the child gave a visual analogue scale (VAS) score of 0. The paediatric dermatologist also determined that telangiectasia, fibrofatty tissue, and atrophic scar tissue were absent.
Measurements
A paediatric dermatologist (MdG, SP) and 1 of the parents evaluated the residual lesion at follow-up using the Patient and Observer Scar Assessment Scale (POSAS) (20). The POSAS consists of 2 components, of which the Observer Scar Assessment Scale (OSAS) was evaluated by the paediatric dermatologist and the Patient Scar Assessment Scale (PSAS) was evaluated by the parent. Both components include an evaluation of the overall appearance of the residual lesion, scored on a 10-point rating scale, in which 1 corresponds to “normal skin” appearance and 10 corresponds to the “worst imaginable” residual lesion. The POSAS has sufficient internal consistency and reliability to evaluate scar appearance (20). The child was asked to rate the current appearance of the IH on a visual analogue scale (VAS) ruler, on which different smiley faces corresponded to the outcomes 0 (excellent) to 10 (poor) (21). Studies have shown that the validity of a VAS with smiley faces is adequate in children at this age (22). The paediatric dermatologist also recorded whether telangiectasia, fibrofatty tissue, and atrophic scar tissue were present (1) or absent (0). The child’s skin type was categorised according to the Fitzpatrick classification (23).
Clinical characteristics (location, type, pattern, size, and ulceration) were determined by treating physicians (CB, EM, MdG, SP), based on photographs of the IH prior to beta-blocker treatment and using the classification system of the International Society for the Study of Vascular Anomalies (ISSVA) and definitions provided in clinical practice guidelines (24–26). Follow-up time was defined as the time between the cessation of beta-blocker treatment and participation in the study. Maternal educational level was used for its association with socioeconomic status, healthcare access, and treatment compliance, and was categorised according to the International Standard Classification of Education (ISCED) (27, 28).
Data analysis
In case of multiple IH, the most problematic tumour (i.e. the IH that was the indication to initiate beta-blocker treatment) was included in the analyses. Prior to analysis, missing data were imputed using multivariate imputation by chained equations (MICE). Three separate multivariable linear regression analyses were computed to investigate prognostic factors for 3 measures of aesthetic outcome (i.e. PSAS, OSAS, VAS child). Three separate multivariable logistic regression analyses were computed to assess prognostic factors for telangiectasia, fibrofatty tissue, and atrophic scar tissue. For all analyses, the initial model included all candidate prognostic factors: characteristics of the IH (i.e. location, type, pattern, size, and ulceration), of the beta-blocker treatment (i.e. beta-blocker type, cumulative dose, age at treatment initiation, and follow-up time) included in a second model, based on a 2-sided p < 0.20 to minimise optimism and selection bias. Data were analysed using R Studio version 4.1.1. (29).
RESULTS
Participant characteristics
In total, 103 children were included in the analyses (66% of 157 eligible children). For an elaborate description, including a recruitment flowchart, see Hermans et al. (13, 19). Children were mostly female (81%; Table I) and participated at a median age of 7.5 years (interquartile range (IQR) 6.9–8.6 years). The majority of children had IH sized < 30 mm (60%), and IH were most commonly located on the head (79%). The median dermatologist-rated OSAS score was 2 (IQR 1–3), the median parent-rated PSAS score was 2 (IQR 1–4), and the median child-rated VAS score was 2 (IQR 0–3). Telangiectasia was present in 67 children (65%), fibrofatty tissue in 44 children (43%), and atrophic scar tissue in 31 children (30%).
Prognostic factors for aesthetic outcome as rated by dermatologists, parents, and children
Table II shows the regression coefficients of the prognostic factors with p < 0.20 of dermatologists’, parents’ and children’s scores. Six variables contrived the best prognostic model for the dermatologist-reported OSAS scores. Deep IH (as opposed to mixed or superficial IH) and IH of males (as opposed to those of females) had better OSAS scores. Longer follow-up time was also associated with better OSAS scores. Ulcerated IH (as opposed to IH without ulceration), older age at treatment initiation, and a higher cumulative dose were related to worse OSAS scores. The adjusted R-squared showed that 42% of variance in OSAS scores was explained by these 6 variables.
Comparable to the dermatologist-reported OSAS scores, prognostic factors for parent-reported PSAS scores were deep IH, the presence of ulceration, age at treatment initiation, cumulative dose, and follow-up time. In addition, IH sized 30–50 mm had worse PSAS scores than smaller or larger IH. In total, 29% of variance in PSAS scores was explained by these 6 factors. The aforementioned prognostic factors of dermatologist-reported OSAS and parent-reported PSAS scores were not significantly associated with child-reported VAS scores. Instead, child-reported VAS scores were related only to the location of the IH. Hence, IH located in the head and face region had lower (i.e. better) VAS scores than IH located elsewhere. This variable explained 10% of the variance in VAS scores.
Prognostic factors for sequelae
Telangiectasia were associated with the following 5 prognostic factors (Table III). Deep IH (as opposed to mixed or superficial IH), focal IH (as opposed to segmental or indeterminate IH), and intermediate maternal educational level (as opposed to high or low maternal educational level) were less likely to have telangiectasia. On the other hand, ulcerated IH and IH requiring higher cumulative dose were more likely to have telangiectasia in the long-term.
Fibrofatty tissue was associated with 4 variables. Deep IH (as opposed to mixed or superficial IH) and ulcerated IH (as opposed to IH without ulceration) were less likely to have fibrofatty tissue. Males were less likely to have fibrofatty tissue than females. Higher cumulative dose was related to higher odds of having fibrofatty tissue.
Atrophic scar tissue was less common in children with longer follow-up time. Males were more likely than females to have atrophic scar tissue. Also, IH sized 30–50 mm (as opposed to smaller or larger IH) and ulcerated IH (as opposed to IH without ulceration) were more likely to have atrophic scar tissue.
DISCUSSION
In 103 children who had been treated with beta-blockers (propranolol or atenolol) for IH during infancy (median age at treatment initiation 3.5 months), this study identified prognostic factors for the long-term aesthetic outcome using characteristics of the IH, of the beta-blocker treatment, and of the child. Aesthetic outcome was evaluated by dermatologists, parents, and children. In addition, this study identified prognostic factors for developing telangiectasia, fibrofatty tissue, and atrophic scar tissue on the long term.
The significant prognostic factors for dermatologist- and parent-rated aesthetic outcome largely matched. IH with a superficial component (i.e. superficial or mixed IH), ulcerated IH, and children with older age at treatment initiation, higher cumulative dose, or shorter follow-up time had worse aesthetic outcome. Some of these variables were also associated with sequela type: IH with a superficial component and children with higher cumulative dose were at risk of the development of telangiectasia and fibrofatty tissue. As expected, ulcerated IH were more likely to have telangiectasia and atrophic scar tissue compared with IH without ulceration, but were less likely to have fibrofatty tissue. Longer follow-up time was associated with higher risk of atrophic scar tissue. These results, based on multivariable analyses of IH treated with either propranolol or atenolol, are substantiated by results of previous studies using univariable analyses of untreated IH and IH treated with propranolol (4, 11, 16, 30–32). A higher cumulative dose of the beta-blocker treatment probably reflects more severe IH and is therefore associated with poor outcome. The results underline the importance of close monitoring of IH with a superficial component to establish early treatment initiation and to prevent or treat complications, such as ulceration. Furthermore, the association between longer follow-up time and better aesthetic outcome emphasises that the spontaneous involution phase continues through-out childhood after beta-blocker treatment, which may be reassuring to parents.
When controlled for variables such as ulceration and IH type, the child’s sex and the size of the IH were related to aesthetic outcome. Females had lower aesthetic outcome as rated by the dermatologist and were more likely than males to exhibit fibrofatty tissue. It is possible that the healing mechanism of IH is sex-dependent, given the sex differences in the prevalence of severe IH, and sex differences of the skin, particularly in wound healing (33–35). IH sized 30–50 mm had worse parent-rated aesthetic outcome and were more likely to have atrophic scar tissue compared with smaller (< 30 mm) or larger (> 50 mm) IH. The literature suggests that medical conditions with intermediate visibility may lead to worse psychological adjustment than medical conditions with low or high visibility (36). Perhaps the initial expectations about the long-term aesthetic outcome may not always match the actual long-term aesthetic outcome, leading to negative appraisal. Specifically, parents of infants with either small or large IH may expect either few or many sequelae, respectively, while parents of infants with medium-sized IH may be less certain about the long-term aesthetic outcome. This hypothesis should be tested in larger sample sises. Nevertheless, the results emphasize the importance of creating appropriate expectations about the long-term aesthetic outcome.
Location was not associated with dermatologist- or parent-rated aesthetic outcome, but was the only prognostic factor for child-rated aesthetic outcome. Children gave better scores to IH located in the head and face region than IH located elsewhere. Previous research into school-aged children who had received varying treatments for IH, showed that IH on the scalp, forehead, cheeks, and parotid area had more volumetric regression and less severe sequelae compared with IH located elsewhere (14). In addition, we hypothesise that children may be habituated to a residual lesion located on the head, since they see these IH in the mirror every day, while residual lesions located elsewhere on the body may be visible to them less often.
The significant prognostic factors explained 42% of variance in dermatologist-rated aesthetic outcome, 29% of variance in parent-rated aesthetic outcome, and 10% of variance in child-rated aesthetic outcome. The unexplained variance could be accounted for in several ways. First, other mechanisms may contribute to the long-term aesthetic outcome of IH, such as a genetic predisposition. Secondly, additional subgrouping (e.g. using developmental units (37) to estimate location, instead of global categories) may further clarify how characteristics during infancy contribute to aesthetic outcome on the long-term, although this requires a very large sample size to prevent power problems. Thirdly, older children (e.g. adolescents) may be more concerned with aesthetic outcome, both as a result of ongoing cognitive development that enables self-reflection and the increased importance of body image in adolescence (38, 39). Including adolescents in future research, could therefore lead to more variation in reports.
Study strengths and limitations
Strengths of this study include the consideration of multiple perspectives (i.e. dermatologists, parents, and children) and multivariable analyses to evaluate aesthetic outcome, in contrast to previous research that primarily considered univariable analyses of physician-rated aesthetic outcome. As patient-centred care involves integrating multiple characteristics and the assessment of dermatologists, parents and children, the results closely reflect clinical practice and are readily applicable. This study is limited by the lack of validated outcome measures to score the long-term aesthetic outcome of IH. Furthermore, this study examined a large number of prognostic factors and outcomes in a relatively small and homogeneous sample size, meaning overfitting and false-positive results are plausible. For instance, the finding that average maternal educational level (compared with high or low maternal educational level) was associated with fewer telangiectasia, may have been coincidental. Exact prognoses based on the models here should be tested in larger patient groups. Nevertheless, the fact that the current study confirms previous findings in untreat-ed IH or IH treated with propranolol advocates for the generalizability of the results.
Conclusion
A superficial component, ulceration and older age at treatment initiation are negatively associated with long-term aesthetic outcome of IH treated with either propranolol or atenolol. Furthermore, as expected, ulcerated IH and a higher cumulative dose of beta-blocker treatment are associated with a poorer aesthetic outcome in terms of sequelae. In order to optimise the long-term aesthetic outcome, close monitoring of proliferating IH, especially IH with a superficial component, is required. This may facilitate early treatment initiation to prevent or treat ulceration, to optimise parent counselling and to guide treatment strategies after beta-blocker treatment.
ACKNOWLEDGEMENTS
The authors thank Emma Jonge Poerink for her contribution to facilitating the data collection for this research project.
Trial registration
Netherlands Trial Register, NL7703 https://www.trialregister.nl/trial/7703.
REFERENCES
- Anderson KR, Schoch JJ, Lohse CM, Hand JL, Davis DM, Tollefson MM. Increasing incidence of infantile hemangiomas (IH) over the past 35 years: correlation with decreasing gestational age at birth and birth weight. J Am Acad Dermatol 2016; 74: 120–126.
- Dickison P, Christou E, Wargon O. A prospective study of infantile hemangiomas with a focus on incidence and risk factors. Pediatr Dermatol 2011; 28: 663–669.
- Munden A, Butschek R, Tom WL, Marshall JS, Poeltler DM, Krohne SE, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol 2014; 170: 907–913.
- Bauland CG, Lüning TH, Smit JM, Zeebregts CJ, Spauwen PH. Untreated hemangiomas: growth pattern and residual lesions. Plast Reconstr Surg 2011; 127: 1643–1648.
- Chang LC, Haggstrom AN, Drolet BA, Baselga E, Chamlin SL, Garzon MC, et al. Growth characteristics of infantile hemangiomas: implications for management. Pediatrics 2008; 122: 360–367.
- Couto RA, Maclellan RA, Zurakowski D, Greene AK. Infantile hemangioma: clinical assessment of the involuting phase and implications for management. Plast Reconstr Surg 2012; 130: 619–624.
- Haggstrom AN, Drolet BA, Baselga E, Chamlin SL, Garzon MC, Horii KA, et al. Prospective study of infantile hemangiomas: clinical characteristics predicting complications and treatment. Pediatrics 2006; 118: 882–887.
- Léauté-Labrèze C, Dumas de la Roque E, Hubiche T, Boralevi F, Thambo JB, Taïeb A. Propranolol for severe hemangiomas of infancy. N Engl J Med 2008; 358: 2649-2651.
- Ji Y, Chen S, Yang K, Zhang X, Zhou J, Li L, et al. Efficacy and safety of propranolol vs atenolol in infants with problematic infantile hemangiomas: a randomized clinical trial. JAMA Otolaryngol Head Neck Surg 2021; 147: 599–607.
- Liu Z, Wu C, Song D, Wang L, Li J, Wang C, et al. Atenolol vs. propranolol for the treatment of infantile haemangiomas: a systematic review and meta-analysis. Exp Ther Med 2020; 20: 1644–1652.
- Yu Z, Cai R, Chang L, Qiu Y, Chen X, Chen Q, et al. Clinical and radiological outcomes of infantile hemangioma treated with oral propranolol: a long-term follow-up study. J Dermatol 2019; 46: 376–382.
- Dieterich-Miller CA, Cohen BA, Liggett J. Behavioral adjustment and self-concept of young children with hemangiomas. Pediatr Dermatol 1992; 9: 241–245.
- Hermans MM, Breugem CC, Schappin R, Jonge Poerink E, Mendels EJ, Ragamin A, et al. Aesthetic outcome of propranolol vs atenolol treatment of children with infantile haemangioma. Acta Derm Venereol 2022; 102: adv00788.
- Chang SJ, Yu W, Gu Y, Han Y, Shang Y, Chang L, et al. Location of infantile hemangioma is a predictor of volumetric sequelae after involution. J Dermatol 2019; 46: 371–375.
- Chang SJ, Qiao C, Chang L, Gao W, Jin Y, Ma G, et al. A 7-year follow-up study on untreated deep or mixed facial infantile hemangioma in East-Asian patients: when propranolol was not yet an option. J Dermatol 2019; 46: 962–966.
- Baselga E, Roe E, Coulie J, Munoz FZ, Boon LM, McCuaig C, et al. Risk factors for degree and type of sequelae after involution of untreated hemangiomas of infancy. JAMA Dermatol 2016; 152: 1239–1243.
- Goldenberg DC, Hiraki PY, Marques TM, Koga A, Gemperli R. Surgical treatment of facial infantile hemangiomas: an analysis based on tumor characteristics and outcomes. Plast Reconstr Surg 2016; 137: 1221–1231.
- Yuzuriha S, Nagai F, Noguchi M. How to manage disfiguring scars in involuted infantile hemangioma. Adv Wound Care (New Rochelle) 2019; 8: 221–229.
- Hermans MM, Rietman AB, Schappin R, de Laat PCJ, Mendels EJ, Breur JMPJ, et al. Long-term neurocognitive functioning of children treated with propranolol or atenolol for infantile hemangioma. Eur J Pediatr 2023; 182: 757–767.
- van de Kar AL, Corion LU, Smeulders MJ, Draaijers LJ, van der Horst CM, van Zuijlen PP. Reliable and feasible evaluation of linear scars by the Patient and Observer Scar Assessment Scale. Plast Reconstr Surg 2005; 116: 514–522.
- UMC Utrecht Wilhelmina Kinderziekenhuis. Faces pain scale. [cited 2021 Jul 14] Available from: https://www.hetwkz-kind.nl/obzf/narcose-na-de-ingreep-pijn/pijnliniaaljpg/.
- Tomlinson D, von Baeyer CL, Stinson JN, Sung L. A systematic review of faces scales for the self-report of pain intensity in children. Pediatrics 2010; 126: e1168–1198.
- Fitzpatrick TB. The validity and practicality of sun-reactive skin types I through VI. Arch Dermatol 1988; 124: 869–871.
- Nederlandse Vereniging voor Dermatologie en Venereologie (NVDV). Infantiele Hemangiomen Richtlijn 2021. [cited 2022 Nov 11] Available from: https://www.nvpc.nl/uploads/stand/212NVDV%20Richtlijn%20Infantiele%20Hemangiomen%202021.pdf.
- Krowchuk DP, Frieden IJ, Mancini AJ, Darrow DH, Blei F, Greene AK, et al. Clinical practice guideline for the management of infantile hemangiomas. Pediatrics 2019; 143: e20183475.
- International Society for the Study of Vascular Anomalies (ISSVA). ISSVA Classification of Vascular Anomalies ©2018. [cited 2022 Nov 11] Available from: https://www.issva.org/classification.
- Organisation for Economic Co-operation and Development (OECD), European Union, UNESCO Institute for Statistics. ISCED 2011 Operational Manual: Guidelines for Classifying National Education Programmes and Related Qualifications. [cited 2022 Nov 11] Available from http://dx.doi.org/10.1787/9789264228368-en.
- Mathes T, Jaschinski T, Pieper D. Adherence influencing factors – a systematic review of systematic reviews. Arch Public Health 2014; 72: 37.
- RStudio Team. RStudio: integrated development for R. Boston, MA: RStudio, 2020.
- Sagi L, Zvulunov A, Lapidoth M, Ben Amitai D. Efficacy and safety of propranolol for the treatment of infantile hemangioma: a presentation of ninety-nine cases. Dermatology 2014; 228: 136–144.
- Yildirimcakar D, Demirsoy U, Azizoglu M, Corapcioglu F. Evaluation of clinical properties and treatment responses of infantile hemangioma. J Drugs Dermatol 2020; 19: 1156–1165.
- Al-Mayoof AF, Joda AE, Almushhadany OE. Propranolol therapy in infantile hemangioma: correlation of age and duration of treatment to the outcomes. Ann Pediatr Surg 2019; 15: 8.
- Hoornweg MJ, Smeulders MJC, Ubbink DT, van der Horst CMAM. The prevalence and risk factors of infantile haemangiomas: a case-control study in the Dutch population. Paediatr Perinat Epidemiol 2012; 26: 156–162.
- Dao H, Kazin RA. Gender differences in skin: a review of the literature. Gend Med 2007; 4: 308–328.
- Engeland CG, Bosch JA, Cacioppo JT, Marucha PT. Mucosal wound healing: the roles of age and sex. Arch Surg 2006; 141: 1193–1197.
- Moss TP. The relationships between objective and subjective ratings of disfigurement severity, and psychological adjustment. Body Image 2005; 2: 151–159.
- Haggstrom AN, Lammer EJ, Schneider RA, Marcucio R, Frieden IJ. Patterns of infantile hemangiomas: new clues to hemangioma pathogenesis and embryonic facial development. Pediatrics 2006; 117: 698–703.
- Riley AW. Evidence that school-age children can self-report on their health. Ambul Pediatr 2004; 4: 371–376.
- Smolak L. Appearance in childhood and adolescence. The Oxford handbook of the psychology of appearance. New York, NY: Oxford University Press; 2012: p. 123–141.