SHORT COMMUNICATION
Near-ultraviolet Light Dermoscopy for Identification of Pigmented Skin Tumours
Akane MINAGAWA, Maureen T. MELING, Hiroshi KOGA and Ryuhei OKUYAMA
Department of Dermatology, Shinshu University School of Medicine, 3-1-1 Asahi, Matsumoto, Nagano 390-8621, Japan. E-mail: akn@shinshu-u.ac.jp
Citation: Acta Derm Venereol 2023; 103: adv00876. DOI: https://doi.org/10.2340/actadv.v103.5302.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Jan 11, 2023; Published: Feb 28, 2023
INTRODUCTION
Recently, 405-nm near-ultraviolet light dermoscopy (nUVD) was reported as useful for identifying demarcation of cutaneous malignant melanoma (MM) (1). However, the utility of nUVD for identifying other dermoscopic structures and patterns has not been established. This study evaluated the performance of nUVD for dermoscopic evaluation of 4 types of pigmented skin tumours: MM, melanocytic naevus (MN), basal cell carcinoma (BCC) and seborrhoeic keratosis (SK).
MATERIALS AND METHODS
Patients who consulted the Department of Dermatology at Shinshu University Hospital, Matsumoto, Nagano, Japan, between July 2019 and December 2020 were included in this study. Inclusion criteria were: a diagnosis of MM, MN, BCC or SK as confirmed by dermoscopic findings alone and/or histopathological biopsy evaluation, together with manifestation on the face, trunk or extremities. Lesions on the scalp, nails, palms, or volar areas were excluded, as were cases of a dual diagnosis in 1 lesion. This study was approved by the ethics board of Shinshu University School of Medicine (number 5409). Requirement for patient consent was waived by the ethics board because the study was retrospective and non-interventional.
Digitized images were simultaneously captured under a set magnification for each lesion by polarized dermoscopy (PD), non-polarized dermoscopy (NPD) and nUVD using a DZ-D100 camera (Casio Computer Co., Ltd, Tokyo, Japan) in the customized mode. The DZ-D100 captures sequential PD, NPD and nUVD images with a single button push to obtain images in the exact same location.
In the initial step, 3 dermoscopy readers (MT, AM and HK) evaluated each image obtained by PD, which is the most frequently used method in clinical practice for the identification of structures and patterns, as described in the consensus of the International Society of Dermoscopy (2). Next, the PD images were compared side by side with the corresponding NPD and nUVD images. The test images are shown in Appendix S1. For large lesions, all accompanying images (not shown in Appendix S1) were also evaluated. The identification levels for each dermoscopic finding were judged as: (+) the structure or pattern was easier to identify with the NPD- or nUVD-captured image than with the PD-acquired image alone, (–) the structure or pattern was less identifiable on NPD or nUVD compared with PD alone, and (=) adding the NPD- or nUVD-captured image provided no remarkable advantage in detecting the structure or pattern in comparison with PD alone.
Each reader evaluated the images independently in order to minimize possible observer bias. In the case of disagreement, a consensus was reached by discussion among the readers. The frequencies of (+), (–), and (=) for each finding were calculated and compared among PD, nPD and nUVD. Dermo-scopic structures and patterns found in fewer than 2 lesions were omitted from further comparisons and analyses.
RESULTS
The clinical features of the 207 pigmented skin lesions are shown in Table SI. Representative dermoscopic images of the 4 types of lesion are shown in Fig. 1 and Appendix S2.
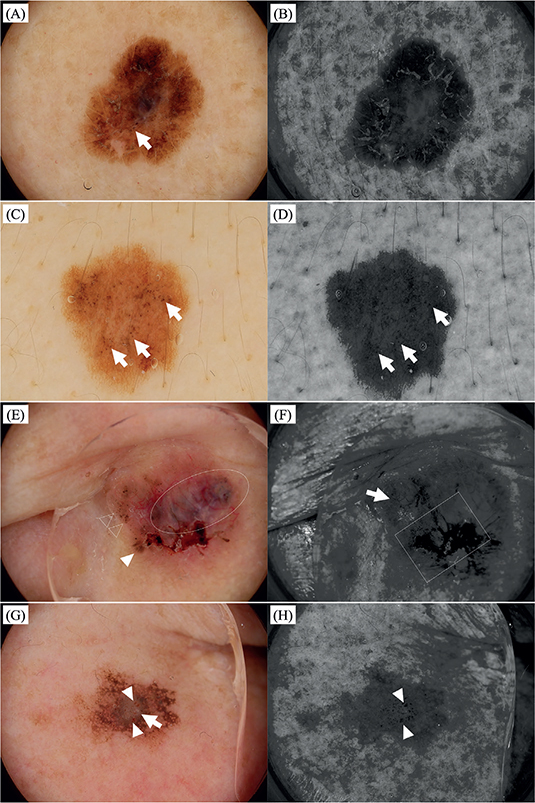
Fig. 1. Representative dermoscopic images of pigmented skin tumours. (A and B) Malignant melanoma on the arm (size: 10*9 mm). (A) Lesion consists of atypical pigment networks and irregularly distributed dots/globules (arrows). (B) Demarcation of the lesion is more clearly identifiable, while pigment networks and dots/globules are less prominent compared with (A). (C and D) Melanocytic nevus on the back (size: 9*9 mm). (C) Lesion consists of typical pigment networks and dots/globules (arrows). (D) Demarcation of the lesion is more clearly identifiable. Pigment networks and some dots/globules (arrows) are equally recognizable as in (C). (E and F) Basal cell carcinoma on the nose (size: 13*11 mm). (E) Blue globules (oval line), maple leaf-like structures (white arrowhead), dots/globules (clear arrowheads), arborizing vessels, and crusts are visible. (F) Crusts (square line) are enhanced, while blue globules, maple leaf-like structures, and dots/globules are scarcely visible. The arborizing vessels on the left side (arrow) are equally observable as in (E), while those on the right side are not detected. The demarcation of the lesion is less apparent. (G and H) Seborrheic keratosis on the cheek (size: 8*7 mm). (G) Pseudo-networks, milia-like cysts (arrow), and multiple comedo-like openings (arrowheads) are seen. (H) Comedo-like openings more clearly distinguishable (arrowheads), although pseudo-networks and milia-like cysts are less visible. (A, C, E, and G; polarized dermoscopy: B, D, F, and H; near-ultraviolet dermoscopy).
The frequencies of dermoscopic findings for PD, NPD and nUVD are summarized in Table SII. Ulcers and crusts in BCC (22/25, 88.0% and 18/25, 72.0%, respectively), as well as comedo-like openings and cerebriform patterns in SK (25/32, 78.1% and 20/28, 71.4%, respectively) were greatly highlighted on nUVD. In addition, all ulcers in MM and SK, cerebriform patterns in MN, milia-like cysts in BCC, and crusts in SK were highlighted on nUVD, although the number of cases was small (<10). Overall, ulcers (26/29, 89.7%) were the most frequently highlighted dermoscopic finding on nUVD, followed by comedo-like openings (25/32, 78.1%), crusts (24/31, 77,4%) and cerebriform patterns (22/30, 73.3%). In contrast, pseudo-networks in MM and SK (7/7, 100% and 8/8, 100%, respectively), and dots/globules in BCC (15/15, 100%) were most frequently non-identifiable on nUVD, followed next by blue globules and blue-grey ovoid nests in BCC (31/32, 96.9% and 24/27, 88.9%, respectively). Visualization on nUVD varied for some dermoscopic findings; pigment networks and dots/globules in MM (10/12, 83.4% and 6/8, 75.0%, respectively) tended to not be highlighted on nUVD, while those in MN (20/24, 83.3% and 14/26, 53.9%, respectively) were equally visualized on nUVD compared with PD. The demarcations of MM (26/27, 96.3%) and MN (34/44, 77.3%) were often highlighted on nUVD, while those of BCC (13/75, 17.3%) and SK (16/61, 26.2%) were not.
DISCUSSION
The current study examined the performance of nUVD for evaluating pigmented skin tumours. Ulcers were the most frequently detected dermoscopic finding in nUVD-assisted visualization, although the precise mechanism for this finding was uncertain. Regarding its ability to detect skin ulcers, nUVD could increase diagnostic accuracy for BCC. Such an advantage of nUVD may also be helpful for examination of MM, as ulcers are a strong predictor of prognosis in patients with MM (3, 4).
The demarcation of melanocytic tumours (MM and MN) was the second most frequently highlighted feature on nUVD, as reported previously (1). In contrast, nUVD was less helpful for determining BCC and SK demarcations. As melanin granules in the superficial skin are more sensitive to ultraviolet (UV) light (5), the granules in that segment are likely to be larger and more homogeneously distributed in melanocytic tumours, although histopathological specimen examination is required for more accurate estimation.
Interestingly, the visualization of dots/globules on nUVD varied among the benign and malignant diseases. As the melanin granules in malignant neoplasms tend to be irregularly distributed across a wider range from the superficial to deep layers, melanin granules in the deeper regions of MM and BCC are presumably less visible by nUVD. One reason is that nUVD light does not penetrate deeply into the skin. Blue globules and blue-grey ovoid nests were likewise less detectable on nUVD, since these findings corresponded to the melanin granules located in the deep areas of BCC.
Crusts and comedo-like openings were other frequently highlighted findings on nUVD. These dermoscopic features include large amounts of keratin (6). Porphyrins present in keratin are the most likely candidate for nUVD detection, since 400-nm wavelength reactivity is typical of the Soret band of porphyrins (7). On the other hand, pseudo-networks were most frequently non-identifiable by nUVD. As melanin granules and keratin in the follicles are components of pseudo-networks and both are highlighted on nUVD, distinguishing them may be difficult by nUVD alone.
The reasons for less, equal, or more enhancement of the remaining dermoscopic findings on nUVD are not completely understood. In parts of cerebriform patterns, the simple unevenness of the surface was presumed to be emphasized by the black-and-white image. Given the high absorbances of both oxidized and reduced haemoglobins at approximately 405 nm wavelength (8), vascular structures were expected to be highlighted by nUVD; however, the current results did not always follow this principle. The visualization of dermoscopic findings on nUVD also seemed to be influenced by the features around the targeted findings, and so further investigation is required.
Limitations
The heterogeneity and non-excision of some lesions, in addition to the small number of samples, might have limited the strength of the results of this study, as they were retrospectively obtained at a single study centre in Japan. Another limitation was that the identification levels of dermoscopic findings were determined based on each reader’s subjective judgement.
Conclusion
Use of nUVD is advantageous for detecting ulcers, comedo-like openings, crusts, and cerebriform patterns compared with conventional dermoscopy alone. The highlighted demarcation of melanocytic tumours is another potential merit of nUVD. Different visualization of pigment networks and dots/globules on nUVD may provide helpful information for malignancy determination in pigmented skin tumours. However, large multicentre studies of cases with varied skin types are needed to validate these results.
ACKNOWLEDGEMENTS
The Department of Dermatology at Shinshu University conducts joint research with Casio Computer Co., Ltd. The Department of Dermatology at Shinshu University received payment from Casio Computer Co., Ltd., for providing dermoscopy images and annotations for their website (https://dz-image.casio.jp). Dr Koga received a speaker’s fee from Casio Computer Co., Ltd. Drs Koga and Minagawa received a consultant fee from Casio Computer Co., Ltd.
REFERENCES
- Sano T, Minagawa A, Suzuki R, Koga H, Okuyama R. Dermoscopy with near-ultraviolet light highlights the demarcation of melanin distribution in cutaneous melanoma. J Am Acad Dermatol 2021; 84: e23–e24.
- Kittler H, Marghoob AA, Argenziano G et al. Standardization of terminology in dermoscopy/dermatoscopy: results of the third consensus conference of the International Society of Dermoscopy. J Am Acad Dermatol 2016; 74: 1093–1106.
- Gershenwald JE, Scolyer RA. Melanoma Staging: American Joint Committee on Cancer (AJCC) 8th Edition and Beyond. Ann Surg Oncol 2018; 25: 2105–2110.
- Garbe C, Amaral T, Peris K et al. European consensus-based interdisciplinary guideline for melanoma. Part 1: Diagnostics – Update 2019. Eur J Cancer 2020; 126: 141–158.
- Klatte JL, van der Beek N, Kemperman PMJH. 100 years of Wood’s lamp revised. J Eur Acad Dermatol Venereol 2015; 29: 842–847.
- Minagawa A. Dermoscopy–pathology relationship in seborrheic keratosis. J Dermatol 2017; 44: 518–524.
- Morvová M Jr, Jeczko P, Šikurová L. Gender differences in the fluorescence of human skin in young healthy adults. Skin Res Technol 2018; 24: 599–605.
- Nitzan M, Nitzan I, Arieli Y. The various oximetric techniques used for the evaluation of blood oxygenation. Sensors (Basel) 2020; 20: 4844.