REVIEW ARTICLE
Safety and Efficacy of Topical Calcineurin Inhibitors in the Treatment of Facial and Genital Psoriasis: A Systematic Review
Diva AMIRI, Christopher WILLY SCHWARZ, Lise GETHER and Lone SKOV
Copenhagen Research Group for Inflammatory Skin (CORGIS), Department of Dermatology and Allergy, Herlev and Gentofte Hospital, University of Copenhagen, Hellerup, Denmark
Facial and genital psoriasis impairs quality of life and is challenging to treat because of increased percutaneous penetration and, consequently, increased risk of adverse effects. Topical calcineurin inhibitors are recognized as a valid off-label treatment for these sensitive skin areas, but data on safety and efficacy are limited. This systematic review of the literature included 24 of 3,322 studies (5 randomized controlled trials, 9 open-label studies, 2 case series and 8 case reports). All studies demonstrated positive efficacy; 11 studies found statistically significant reductions in psoriasis severity. Local stinging, burning and itching were the most common short-term adverse effects and were reported in 18 studies. Topical calcineurin inhibitors appear to have an important role in the treatment of facial and genital psoriasis. The drugs are effective and generally well-tolerated with few adverse effects.
Key words: calcineurin inhibitors; pimecrolimus; psoriasis; systematic review; tacrolimus; treatment outcome.
SIGNIFICANCE
Among patients with psoriasis, up to 29% develop facial psoriasis and 63% develop genital psoriasis. Facial and genital psoriasis impairs quality of life and because of the sensitive nature of these skin areas, treatment is challenging. Topical calcineurin inhibitors are recognized as a valid off-label treatment. This study systematically searched the literature to investigate whether this off-label treatment is safe and effective. This systematic review confirms that topical calcineurin inhibitors are safe and effective in treating facial and genital psoriasis.
Citation: Acta Derm Venereol 2023; 103: adv00890. DOI https://doi.org/10.2340/actadv.v103.6525.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Feb 9, 2023; Published: Mar 14, 2023
Corr: Lone Skov, Department of Dermatology and Allergy, Herlev and Gentofte Hospital, Gentofte Hospitalsvej 15, DK-2900 Hellerup, Denmark. E-mail: Lone.Skov.02@regionh.dk
INTRODUCTION
Psoriasis is a common chronic immune-mediated skin disease driven by T-lymphocytes and mediated by pro-inflammatory cytokines, which affects approximately 2–3% of the population in Western countries (1). Psoriasis lesions often present as well-demarcated, erythematous and scaly plaques (2), while genital lesions are thinner and less scaly due to increased friction in the area (3, 4). Approximately 17–29% of adult patients with psoriasis have facial lesions and 33–63% have genital lesions (3, 4), and these patients report significantly decreased quality of life compared with patients with psoriasis located elsewhere (5, 6). Adequate and efficient treatment is important to reduce the negative impact on quality of life. Treatment of these sensitive skin areas, however, is challenging because of increased percutaneous penetration (7, 8), which increases the risk of adverse effects. First-line treatment of psoriasis is topical treatment, with the most prescribed being vitamin D analogues, low-potency corticosteroids and calcineurin inhibitors (9). Although highly efficacious, topical corticosteroids are, especially in facial and genital areas, associated with adverse effects, such as skin atrophy, striae, telangiectasia, perioral dermatitis and acneiform eruptions (10, 11). The use of topical vitamin D analogues is limited in sensitive skin areas since they induce skin irritation (12, 13). In recent years, topical calcineurin inhibitors, especially tacrolimus and pimecrolimus, have been recognized as valid off-label treatments for psoriasis in sensitive skin areas. Topical calcineurin inhibitors are anti-inflammatory drugs that inhibit the activation of T-lymphocytes and the production and secretion of pro-inflammatory cytokines and are approved for the treatment of atopic dermatitis. They are well-tolerated with mild, transient and self-limiting adverse effects (14) and do not cause skin atrophy (15).
The aim of this systematic review was to assess the safety and efficacy of off-label treatment of facial and genital psoriasis with topical calcineurin inhibitors.
MATERIALS AND METHODS
Literature search
This systematic review was performed following Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (16). The study protocol is registered at PROSPERO (ID: CRD42022315216). The 3 databases, PubMed, Web of Science and Embase, were systematically searched from database inception until 18 February 2022. Search terms were “(tacrolimus OR protopic OR fk506 OR calcineurin inhibitor OR calcineurin inhibitors OR pimecrolimus OR elidel OR topical calcineurin inhibitor OR tci) AND (psoriasis)”. The search results were uploaded to Rayyan Qatar Computing Research Institute, a web/mobile application for systematic reviews, to expedite the screening of abstracts and titles (17). The resulting titles were screened by a single author (DA) in order to exclude duplicates. Two authors (CWS and DA) independently screened all titles and abstracts for eligibility, based on the inclusion criteria described below. Relevant full-text articles were retrieved and read. Dis-agreement between the reviewers was resolved through debate and the resulting outcome was agreed upon.
Inclusion and exclusion criteria
All studies in languages other than Danish or English were excluded. All study designs were eligible for inclusion; however, studies had to be original and include patients with facial and/or genital psoriasis treated with topical calcineurin inhibitors. All types of psoriatic lesions in these areas and all age groups were included. Studies investigating combination therapy and studies not including facial and genital psoriasis were excluded.
Data extraction
The following data were retrieved from each publication when applicable: author information, title, publication year, study design, country, number of patients, sex, mean or median age, measure of psoriasis severity, type of psoriasis, psoriasis site, topical calcineurin inhibitor type and strength, treatment area, frequency of treatment, treatment duration, follow-up, evaluation week of response, response in relation to relevant psoriasis severity measures and adverse events.
Quality assessment
To assess the quality of the studies, this study used an adapted version of the Newcastle-Ottawa scale for randomized controlled trials and open-label studies. Each study was evaluated based on 8 items in 3 categories: study selection, comparability and ascertainment of either the exposure or outcome of interest (18). Quality assessment was performed by 2 authors (CWS and DA) and any disagreements were resolved through debate. A Newcastle-Ottawa score of ≥ 7 points was considered high quality (Table SI).
RESULTS
The initial literature search resulted in 3,322 non-duplicate publications. The PRISMA flow diagram in Fig. 1 illustrates the search history. The search resulted in the inclusion of 24 publications with a total of 568 patients. The studies originated from Asia (n = 7) (19–25), Europe (n = 9) (26–34), the Middle East (n = 2) (35, 36) and North America (n = 6) (37–42). Of these 24 publications, 17 included adults aged ≥ 18 years (19, 21–28, 30–34, 37–40) and 6 included paediatric patients aged 6 months to 16 years (20, 29, 35, 36, 41, 42). Among the 24 publications, 16 studied tacrolimus 0.1% (19, 21–24, 28–30, 32–34, 37, 39–42), 1 studied tacrolimus 0.03% (25) and 7 studied pimecrolimus 1% (20, 26, 27, 31, 35, 36, 38). The publications consisted of 5 randomized controlled trials (25, 26, 30, 38, 40), 9 open-label studies (23, 24, 27, 28, 31, 33, 37, 39, 41), 2 case series (32, 42), and 8 case reports (19–22, 29, 34–36).
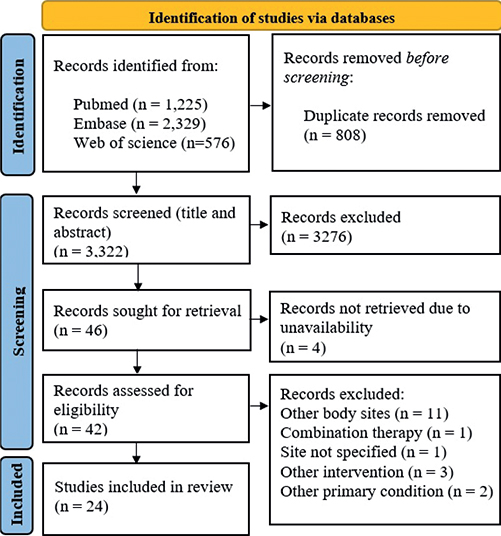
Fig. 1. Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 flow diagram.
Efficacy of tacrolimus
In total, 16 studies assessed the efficacy of treatment with tacrolimus 0.1%/0.03% (Table I). Three randomized double-blind controlled studies assessed the efficacy of twice-daily application of tacrolimus. Liao et al. (25) found that significantly more patients with moderate facial and genital psoriasis were “clear” or “almost clear” after 6 weeks of treatment (60% of patients, n = 25) compared with calcitriol (33% of patients, n = 24, p = 0.04), measured by Physician’s Global Assessment. Likewise, Lebwohl et al. (40) found tacrolimus to be superior to the vehicle in moderate facial and genital psoriasis after 8 weeks of treatment. This was measured by Physician’s Global Assessment, with 67% of patients in the tacrolimus group (n = 112) being “clear” or “excellent”, compared with 37% in the vehicle group (n = 55, p = 0.002). Kleyn et al. (30) found tacrolimus to be equally effective as a mid-potency topical corticosteroid. Tacrolimus and clobetasone butyrate both decreased the Total Affected Area after 6 weeks of treatment (22% decrease in tacrolimus group and 18% decrease in clobetasone butyrate group) with no significant treatment difference.
| Author | Study design | Patients n | Age, years Mean (± SD) | Sex (M/F), n | Psoriasis type | Psoriasis site(s) | Psoriasis severity | Intervention(s) | Outcome measure | Result |
| Kleyn et al. (30) | RCT | 28 | 42 | M: 11 F: 17 |
– | Face, flexures, genital | – | 1: Tacrolimus 0.1% 2: Clobetasone butyrate 0.05% |
Decrease in TAA from baseline | From 32% to 10% in tacrolimus group and from 32% to 14% in the clobetasone butyrate group at week 6 |
| Lebwohl et al. (40) | RCT | 167 | 1: 48.0 (± 15.7) 2: 48.0 (± 15.6) |
M: 98 F: 69 |
Plaque psoriasis | Face, intertriginous |
Moderate psoriasis | 1: Tacrolimus 0.1% 2: Vehicle |
PGAb of clear or excellent improvement | 67% in the tacrolimus group and 37% in the vehicle group at week 8 (p = 0.002) |
| Liao et al(26) | RCT | 49 | 39.6 ± 12.8) | M: 36 F: 13 |
Plaque psoriasis | Face, genitofemoral | Moderate psoriasis | 1: Tacrolimus 0.03% 2: Calcitriol 0.0003% |
PGAa of 4 or 5 | 60% in the tacrolimus group and 33% in the calcitriol group at week 6 (p = 0.04) |
| Bissonnette et al. (37) | Open-label | 12 | 42 | M: 12 | Plaque psoriasis | Penis, scrotum | Severe psoriasis | Tacrolimus 0.1% | Decrease in M-PASIc from baseline | From 15.8 at baseline to 1.2 at week 8 (p < 0.001) |
| Brune et al. (41) | Open-label | 11 | 10.5 | M: 6 F: 5 |
– | Face, intertriginous |
Mild to moderate psoriasis | Tacrolimus 0.1% | Overall severity scoref | From 1.63 at baseline to 0.71 at day 180 (p < 0.0001) |
| Ezquerra et al. (33) | Open-label | 15 | 53 | M: 8 F: 7 |
– | Face, intertriginous, genital |
Severe psoriasis | Tacrolimus 0.1% | Individual symptom scoree | From 6.88 at baseline to 0.37 at day 60 (p < 0.001) |
| Freeman et al.(40) | Open-label | 21 | 48 | M: 15 F: 6 |
– | Face, intertriginous |
Severe psoriasis | Tacrolimus 0.1% | Individual symptom scoree | From 6.7 at baseline to 0.3 at day 57 (p < 0.0001) |
| Rallis et al. (29) | Open-label | 10 | 32.2 | M: 9 F: 1 |
– | Face, glans penis, scrotum | Moderate psoriasis | Tacrolimus 0.1% | Overall severity scored | From 7.8 at baseline to 0 at week 3 |
| Yamamoto et al. (23) | Open-label | 21 | 51.1 | M: 15 F: 6 |
Psoriasis vulgaris | Face | Moderate psoriasis | Tacrolimus 0.1% | Decrease in clinical score (erythema, infiltration, desquamation) from baselineg | Erythema from 1.76 to 0.62 at week 4 (p < 0.0001), infiltration from 1.33 to 0.43 at week 4 (p < 0.0005), and desquamation from 0.95 to 0.24 at week 4 (p < 0.001) |
| Yamamoto et al. (24) | Open-label | 11 | 49.3 | M: 6 F: 5 |
Psoriasis vulgaris | Face | – | Tacrolimus 0.1% | Global evaluation of response | 45.5% with complete response, 45.5% with partial response, and 9% resistant at week 4 |
| Clayton et al.(32) | Case series |
4 | 30.3 | M: 1 F: 3 |
Plaque psoriasis | Face | – | Tacrolimus 0.1% | Resolution | 75% with complete clearance, 25% with considerable improvement at 2 months |
| Karajovanov et al. (29) | Case report | 1 | 11 | M: 1 | – | Axillae, genital, auricular |
– | Tacrolimus 0.1% | Resolution | Resolution of the lesions at week 4 |
| Kroft et al. (34) | Case report | 1 | 51 | M: 1 | Plaque psoriasis | Face | – | Tacrolimus 0.1% | Resolution | Almost complete clearance at week 8 |
| Steele et al. (43) | Case series Retrospective study |
13 | 6.6 | M: 9 F: 4 |
– | Penis, scrotum, buttocks, perianal, intertriginous, vulva | – | Tacrolimus 0.1% | Resolution | 92% with complete clearance, 8% with no improvement at week 2 |
| Yamamoto et al. (22) | Case report | 2 | 39.5 | M: 2 | – | Face, lips | Severe psoriasis | Tacrolimus 0.1% | Physician assessment | Completely disappeared and good improvement at week 2 |
| Yao et al. (22) | Case report | 1 | 37 | M: 1 | Psoriasis vulgaris | Glans penis | – | Tacrolimus 0.1% | Resolution | Resolution of the lesion at week 3 |
| aPGA: –1 = Worse than baseline, 0 = No change, 1 = 25% from baseline minimal improvement, 2 = 50% from baseline, 3 = 75% from baseline marked improvement, 4 = 90% from baseline almost clear and 5 = 100% from baseline clear. bPGA: 100% = clear, 90–99% = Excellent improvement, 60–89% = Good improvement, 30–59% = Fair improvement, 1–29% = Slight improvement, 0 = No change. cM-PASI: Scale 0–4 (none, mild, moderate, severe, very severe), (erythema + induration + desquamation) · Genital area. 0=no involvement; 1=1–9%, 2=10–29%, 3 = 30–49%, 4 = 50–69%, 5 = 70–90% and 6 = 90–100% involvement. dSeverity score: Scale 0–3 (none, mild, moderate, severe) in erythema, scaling, infiltration and lesional extent. 0=clear, 1–4 = mild, 5–8 = moderate, 9–12 = severe. eIndividual symptom score: Scale 0–3 (absent, mild, moderate, severe) in erythema, infiltration, and desquamation. fSeverity score: Scale 0–3 (none, mild, moderate, severe). gClinical score (Erythema, desquamation, infiltration) 0 = absent, 1 = slight, 2 = moderate, 3 = striking 4 = exceptionally striking. | ||||||||||
| M-PASI: Modified Psoriasis Area and Severity Index; PGA: Physician’s Global Assessment; TAA: Total Affected Area in %; M: male; F: female; RCT: randomized controlled trial; SD: standard deviation. | ||||||||||
Seven open-label studies found improvement in psoriasis with tacrolimus treatment (Table I). Five of these studies reported significant improvement in psoriasis after twice-daily applications of tacrolimus 0.1% compared with baseline (23, 33, 37, 39, 41). Bissonnette et al. (37) found a significant reduction in the severity of genital psoriasis after 8 weeks of treatment measured by Modified Psoriasis Area and Severity Index (n = 12, p < 0.001). Freeman et al. (33) found a significant reduction in Individual Symptom Score after 57 days of treatment (n = 21, p < 0.0001), and Ezquerra et al. reported similar results after 60 days (n = 15, p < 0.001). Both studies assessed severe facial and genital psoriasis. Brune et al. (41) found a significant reduction in the Overall Severity Score of mild-to-moderate psoriasis after 180 days of treatment (n = 8, p < 0.0001). Yamamoto et al. (23) found a significant reduction in all clinical parameters for moderate facial psoriasis after 4 weeks of treatment (n = 19, erythema p < 0.0001, infiltration p < 0.0005, desquamation p < 0.001).
The 4 case reports and the 2 case series report favourable results of twice-daily application of tacrolimus 0.1% (Table I). All studies found complete or almost complete resolution of both facial and genital psoriasis within 2–8 weeks of treatment (21, 22, 29, 32, 34, 42).
Safety of tacrolimus
Thirteen studies assessed the safety of treatment with tacrolimus 0.1%/0.03% (Table SII). Nine of these studies reported transient stinging, burning and itching as adverse effects (23, 25, 30, 33, 37, 39–42). Kleyn et al. (30) and Brune et al. (41) both reported 1 patient withdrawal due to unbearable itching. Liao et al. (25) found that 92% of both groups had excellent tolerance and fewer patients had perilesional erythema after 6 weeks of tacrolimus treatment (16% of patients, n = 25) compared with calcitriol treatment (58% of patients, n = 24, p = 0.004). Lebwohl et al. (40) found no significant difference in burning/stinging, hyperaesthesia or itching between tacrolimus and the vehicle. One case report found deep facial dermatophytosis with tinea corporis following a 4-week treatment with tacrolimus 0.1% of facial psoriasis (19).
Efficacy of pimecrolimus
In total, 7 studies assessed the efficacy of treatment with pimecrolimus 1% (Table II). Two randomized double-blinded controlled studies assessed the efficacy of twice-daily application of pimecrolimus. Gribetz et al. (38) found pimecrolimus to be superior to the vehicle measured by Investigator’s Global Assessment, with 71% of patients in the pimecrolimus group (n = 28) being “clear” or “almost clear” of moderate genital psoriasis after 8 weeks of treatment compared with 21% in the vehicle group (n = 29, p < 0.0001). Kreuter et al. (26) did not find a significant advantage of pimecrolimus compared with the vehicle or calcipotriol 0.005% in the treatment of severe genital psoriasis measured by Modified Psoriasis Area and Severity. The study also found that betamethasone 0.1% was significantly more effective than both pimecrolimus 1% and the vehicle (n = 60, p < 0.01).
| Author | Study design | Patients n | Age, years Mean (±SD) | Sex (M/F) | Psoriasis type | Psoriasis site(s) | Psoriasis severity | Intervention(s) | Outcome measure | Result |
| Gribetz et al. (38) | RCT | 57 | 1: 47.8 (± 15.17) 2: 47.8 (± 14.49) |
M: 29 F: 28 |
Inverse psoriasis Plaque psoriasis |
Inguinal, gluteal cleft, inframammary, axillae |
Moderate psoriasis | 1: Pimecrolimus 1% 2: Vehicle |
IGAa of almost clear or clear | 71% in the pimecrolimus group and 21% in the vehicle group at week 8 (p < 0.0001) |
| Kreuter et al. (27) | RCT | 80 | 53.2 (± 14.5) | M: 49 F: 31 |
Inverse psoriasis | Inguinal folds, gluteal cleft, genital, axillae |
Severe psoriasis | 1: Pimecrolimus 1% 2: Calcipotriol 0.005% 3: Betamethasone 0.1% 4: Vehicle |
Decrease in M-PASI from baseline | From 19.2 to 11.5 in the pimecrolimus group (p=0.001) at day 28. From 25.3 to 9.7 (p < .001) in the calcipotriol group) at day 28. From 22.1 to 2.9 (p < 0.001) in the betamethasone group at day 28. From 18.2 to 13.8 (p = 0.008) in the vehicle group at day 28. |
| Frigerio et al. (31) | Open-label | 40 | 48.3 ± 16.5) | M: 27 F: 13 |
Psoriasis vulgaris | Facial | Moderate psoriasis | Pimecrolimus 1% | PGAc of complete resolution or excellent results. | 72.5% at week 8. |
| Jacobi et al. (28) | Open-label | 20 | Mean age not stated. Range: 19–65 |
M: 13 F: 7 |
Plaque psoriasis | Facial | Moderate psoriasis | Pimecrolimus 1% | Improvement in TSSb from baseline | From 4.2 at baseline to 1.1 at week 8 (p < 0.005) |
| Amichai (36) | Case report | 1 | 10 | M: 1 | – | Glans penis | – | Pimecrolimus 1% | Resolution | Resolution at week 3. |
| Canpolat et al. (20) | Case report | 1 | 6 months | F: 1 | Plaque psoriasis | Anogenital, scalp, trunk, extremities | – | Pimecrolimus 1% | Resolution | Resolution after 1 month. |
| Mansouri et al. (36) | Case report | 1 | 10 | F: 1 | Plaque psoriasis | Face, anogenital | – | Pimecrolimus 1% | Resolution | Resolution of the lesion at day 20. |
| aIGA: 5-point scale. 0 (clear) = no signs of inverse psoriasis except for residual discoloration, 1 (almost clear) = perceptible erythema, no induration, no scaling, 2 (mild disease) = mild erythema, no induration, mild or no scaling, 3 (moderate disease) = moderate erythema, mild induration, mild or no scaling, and 4 (severe disease) = severe erythema, moderate/severe induration and scaling. bTSS: Sum score of erythema and induration, 5-point scale (0–4) 0 = normal skin, 4 = severe erythema, and induration. cPGA: Not explained. > 75% = excellent results. | ||||||||||
| IGA: Investigator’s Global Assessment; M-PASI: Modified Psoriasis Area and Severity Index; PGA: Physician’s Global Assessment of change; TSS: Total Symptom Score. | ||||||||||
Two open-label studies reported improvement of moderate facial psoriasis with twice-daily application of pimecrolimus 1% (Table II) (27, 31). Jacobi et al. found a significant reduction in Total Symptom Score of moderate psoriasis after 8 weeks of treatment (n = 20, p < 0.005) (27).
Three case reports found complete resolution of both facial and genital psoriasis with twice-daily application of pimecrolimus 1% within 4 weeks of treatment (20, 35, 36).
Safety of pimecrolimus
Five studies assessed the safety of pimecrolimus 1% (Table SIII) and 4 reported transient stinging, burning, itching and paraesthesia as adverse effects (26, 27, 31, 38). Frigerio et al. reported that 1 of 40 patients withdrew from the study due to itching and burning (31).
DISCUSSION
This systematic review investigated the short-term safety and efficacy of topical calcineurin inhibitors in the treatment of facial and genital psoriasis in 568 patients across 24 studies. Topical tacrolimus and pimecrolimus appear to have an important role in the treatment of facial and genital psoriasis. The drugs are effective and generally well-tolerated with few local short-term adverse effects. Although 5 of the 24 studies were randomized controlled studies, it should be noted that most publications consisted of small open-label studies, case reports and case series.
Topical corticosteroids are first-line treatment for most patients with localized psoriasis, but their use in the treatment of facial and genital psoriasis is limited due to the increased susceptibility to adverse effects, such as skin atrophy, striae, telangiectasia and acneiform eruptions. The risk of these adverse effects increases with potency, duration, frequency, and application site (11). Several studies have found variation in percutaneous absorption of topical corticosteroids, depending on application site, and 2 studies have found that percutaneous absorption of hydrocortisone on the forehead, scrotum and vulva is greater than on the forearm (7, 43). In this systematic review, 2 of the included studies compared topical corticosteroids with topical calcineurin inhibitors, and found that topical corticosteroids are equally or more effective than topical calcineurin inhibitors and have fewer adverse effects (26, 30). However, treatment durations were short, with a maximum of 6 weeks, which limits the exposure and thereby decreases the risk of adverse effects from topical corticosteroids. The common adverse effects reported, relating to treatment with topical calcineurin inhibitors for facial and genital psoriasis, are transient and mild and often decrease with ongoing use, which is consistent with data from clinical trials in patients with atopic dermatitis (14).
Facial involvement in psoriasis is typically a marker of severe disease with a longer disease duration (44). Severe facial and genital psoriasis are increasingly treated with systemic treatments because of the negative impact on quality of life. In agreement, a new classification of psoriasis severity from the International Psoriasis Council suggests separating patients with psoriasis into patients eligible for topical treatment and patients in need of systemic treatment including those with facial or genital psoriasis (45). Patients with mild facial and genital psoriasis may benefit from topical calcineurin inhibitors, or alternatively it may be used as a maintenance treatment. Patients with facial and genital psoriasis have limited options for topical treatments, since other commonly prescribed treatments, such as topical keratolytics, vitamin D analogues and coal-tar preparations either are not cosmetically acceptable in these areas or can induce severe skin irritation. These factors can affect medication adherence, which is important for adequate treatment results (46).
The efficacy and safety of pimecrolimus in the treatment of facial and genital psoriasis have been evaluated in fewer studies than tacrolimus. Pimecrolimus permeates slower through the skin than tacrolimus, thus having a later onset of action (47), and studies in patients with atopic dermatitis have found topical tacrolimus more effective than pimecrolimus (48). Although pimecrolimus appears to be effective, there is insufficient data to fully determine its role in the treatment of facial and genital psoriasis. Pimecrolimus is more lipophilic than tacrolimus and therefore has greater retention within the skin, reducing the risk of systemic adverse effects (49). These safety advantages and positive results of pimecrolimus warrant studies assessing treatment in facial and genital psoriasis and comparative studies with tacrolimus.
Paediatric studies reported a decrease in clinical severity and a few cases of common adverse effects, such as burning and itching, which is consistent with data from studies in paediatric patients with atopic dermatitis (50). Tacrolimus and pimecrolimus are extensively tested in both adult and paediatric patients with atopic dermatitis. Several randomized controlled trials have shown long-term safety and low systemic absorption of the drugs when applied topically (51–53). Patients with atopic dermatitis have an impaired skin barrier and, consequently, increased percutaneous absorption (54). The proven long-term safety of topical calcineurin inhibitors in paediatric patients with atopic dermatitis and the results from this systematic review support the safety of topical calcineurin inhibitors in the treatment of paediatric facial and genital psoriasis.
The current study has some limitations. Most of the included studies do not differentiate between inverse psoriasis and genital psoriasis, thus varying the descriptions and characterizations of genital psoriasis. Different measures of psoriasis severity are used to assess the treatment efficacy, which complicates comparison of the included studies. Safety assessment varies and few of the included studies do not report adverse effects. Most of the study durations do not exceed 8 weeks and therefore only report short-term adverse effects. The reported adverse effects only include local and not systemic effects. The increased percutaneous absorption through facial and genital skin can lead to a higher systemic concentration; however, none of the included studies measured serum concentration of tacrolimus or pimecrolimus. The systemic concentration may be negligible, but important in order to define safety.
Although there is an increasing number of studies that have assessed the safety and efficacy of topical calcineurin inhibitors in treatment of facial and genital psoriasis, long-term randomized controlled trials or clinical trials with larger sample sizes are warranted. Ten studies were case reports or case series, and therefore a substantial risk of publication bias is present. Case reports of improved facial and genital psoriasis after treatment with topical calcineurin inhibitors are more likely to be published than cases with treatment failure.
Furthermore, a consistent scoring system for the severity of facial and genital psoriasis will ease the comparison of studies and consequently aid in the determination of the most efficient treatment options for these patients.
In conclusion, topical calcineurin inhibitors appear to be effective and safe treatment options for facial and genital psoriasis with few local adverse effects. The drugs should be considered a plausible treatment for facial and genital psoriasis, or, alternatively, used in combination with topical corticosteroids or as a maintenance treatment.
ACKNOWLEDGEMENTS
Conflicts of interest disclosures; LS has received research funding from Novartis, Bristol-Myers Squibb, AbbVie, Janssen Pharmaceuticals, the Danish National Psoriasis Foundation, the LEO Foundation, and the Kgl Hofbundtmager Aage Bang Foundation, and honoraria as consultant and/or speaker for AbbVie, Eli Lilly, Novartis, Pfizer, and LEO Pharma, Janssen Cilag, UCB, Almirall, Bristol-Myers Squibb, and Sanofi. She has served as an investigator for AbbVie, Pfizer, Sanofi, Janssen Cilag, Boehringer Ingelheim, AstraZeneca, Eli Lilly, Novartis, Regeneron, Galderma and LEO Pharma. The remaining authors have no conflicts of interest to declare.
REFERENCES
- Parisi R, Iskandar IYK, Kontopantelis E, Augustin M, Griffiths CEM, Ashcroft DM. National, regional, and worldwide epidemiology of psoriasis: systematic analysis and modelling study. BMJ 2020; 369: m1590.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, Barker JNWN. Psoriasis. Lancet (London, England) 2021; 397: 1301–1315.
- Pincus SH. Vulvar dermatoses and pruritus vulvae. Dermatol Clin 1992; 10: 297–308.
- Shim TN, Ali I, Muneer A, Bunker CB. Benign male genital dermatoses. BMJ 2016; 354: i4337.
- Meeuwis KAP, de Hullu JA, van de Nieuwenhof HP, Evers AWM, Massuger LFAG, van de Kerkhof PCM, et al. Quality of life and sexual health in patients with genital psoriasis. Br J Dermatol 2011; 164: 1247–1255.
- Alpsoy E, Polat M, FettahlıoGlu-Karaman B, Karadag AS, Kartal-Durmazlar P, YalCın B, et al. Internalized stigma in psoriasis: a multicenter study. J Dermatol 2017; 44: 885–891.
- Feldmann RJ, Maibach HI. Regional variation in percutaneous penetration of 14C cortisol in man. J Invest Dermatol 1967; 48: 181–183.
- Britz MB, Maibach HI, Anjo DM. Human percutaneous penetration of hydrocortisone: the vulva. Arch Dermatol Res 1980; 267: 313–316.
- Mason AR, Mason J, Cork M, Dooley G, Hancock H. Topical treatments for chronic plaque psoriasis. Cochrane database Syst Rev 2013: CD005028.
- Lebwohl MG, Tan MH, Meador SL, Singer G. Limited application of fluticasone propionate ointment, 0.005% in patients with psoriasis of the face and intertriginous areas. J Am Acad Dermatol 2001; 44: 77–82.
- Hengge UR, Ruzicka T, Schwartz RA, Cork MJ. Adverse effects of topical glucocorticosteroids. J Am Acad Dermatol 2006; 54: 1–8.
- Kienbaum S, Lehmann P, Ruzicka T. Topical calcipotriol in the treatment of intertriginous psoriasis. Br J Dermatol 1996; 135: 647–650.
- Kragballe K. Treatment of psoriasis with calcipotriol and other vitamin D analogues. J Am Acad Dermatol 1992; 27: 1001–1008.
- Alomar A, Berth-Jones J, Bos JD, Giannetti A, Reitamo S, Ruzicka T, et al. The role of topical calcineurin inhibitors in atopic dermatitis. Br J Dermatol 2004; 151: 3–27.
- Reitamo S, Rissanen J, Remitz A, Granlund H, Erkko P, Elg P, et al. Tacrolimus ointment does not affect collagen synthesis: results of a single-center randomized trial. J Invest Dermatol 1998; 111: 396–398.
- Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ 2009; 339: b2535.
- Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev 2016; 5: 210.
- Wells G, Shea B, O’Connell D, Peterson J. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa, Ottawa Hosp Res Inst 2000 [cited 2022 Jun 1]. Available from: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
- Yamamoto T, Nishioka K. Deep dermatophytosis during topical tacrolimus therapy for psoriasis. Acta Derm Venereol 2003; 83: 291–292.
- Canpolat F, Cemil BC, Tatlican S, Eskioglu F, Oktay M, Alper M. Pimecrolimus 1% cream is effective in the treatment of psoriasis in an infant. Eur J Dermatol 2009; 19: 168–169.
- Yao XJ, Zhang TD. Psoriasis localized to the glans penis in a 37-year-old man. CMAJ 2018; 190: E747.
- Yamamoto T, Nishioka K. Successful treatment with topical tacrolimus for oral psoriasis. J Eur Acad Dermatol Venereol 2006; 20: 1137–1138.
- Yamamoto T, Nishioka K. Topical tacrolimus: an effective therapy for facial psoriasis. Eur J Dermatol 2003; 13: 471–473.
- Yamamoto T, Nishioka K. Topical tacrolimus is effective for facial lesions of psoriasis. Acta Derm Venereol 2000; 80: 451.
- Liao YH, Chiu HC, Tseng YS, Tsai TF. Comparison of cutaneous tolerance and efficacy of calcitriol 3 microg g(-1) ointment and tacrolimus 0.3 mg g(-1) ointment in chronic plaque psoriasis involving facial or genitofemoral areas: a double-blind, randomized controlled trial. Br J Dermatol 2007; 157: 1005–1012.
- Kreuter A, Sommer A, Hyun J, Bräutigam M, Brockmeyer NH, Altmeyer P, et al. 1% pimecrolimus, 0.005% calcipotriol, and 0.1% betamethasone in the treatment of intertriginous psoriasis: a double-blind, randomized controlled study. Arch Dermatol 2006; 142: 1138–1143.
- Jacobi A, Braeutigam M, Mahler V, Schultz E, Hertl M. Pimecrolimus 1% cream in the treatment of facial psoriasis: a 16-week open-label study. Dermatology 2008; 216: 133–136.
- Rallis E, Nasiopoulou A, Kouskoukis C, Roussaki-Schulze A, Koumantaki E, Karpouzis A, et al. Successful treatment of genital and facial psoriasis with tacrolimus ointment 0.1%. Drugs Exp Clin Res 2005; 31: 141–145.
- Karajovanov ID, Najdova A, Nikolovska S, Damevska K, Duma S. Psoriasis inversa in child-case report. J Dermatol Nurses Assoc 2020; 12.
- Kleyn CE, Woodcock D, Sharpe GR. The efficacy of 0.1% tacrolimus ointment compared with clobetasone butyrate 0.05% ointment in patients with facial, flexural or genital psoriasis. Br J Dermatol 2005; 153: 33.
- Frigerio E, Taglioni M, Franchi C, Garutti C, Spadino S, Altomare A, et al. The use of pimecrolimus cream 1% in the treatment of facial psoriasis. G Ital di Dermatologia e Venereol 2006; 141: 415–419.
- Clayton T, Harrison PV, Nicholls R, Delap M. Topical tacrolimus for facial psoriasis. Br J Dermatol 2003; 149: 419–420.
- Martín Ezquerra G, Sánchez Regaña M, Herrera Acosta E, Umbert Millet P. Topical tacrolimus for the treatment of psoriasis on the face, genitalia, intertriginous areas and corporal plaques. J Drugs Dermatol 2006; 5: 334–336.
- Kroft EB, Erceg A, Maimets K, Vissers W, van der Valk PGM, van de Kerkhof PCM. Tacrolimus ointment for the treatment of severe facial plaque psoriasis. J Eur Acad Dermatol Venereol 2005; 19: 249–251.
- Mansouri P, Farshi S. Pimecrolimus 1 percent cream in the treatment of psoriasis in a child. Dermatol Online J 2006; 12: 7.
- Amichai B. Psoriasis of the glans penis in a child successfully treated with Elidel (pimecrolimus) cream. J Eur Acad Dermatol Venereol 2004; 18: 742–743.
- Bissonnette R, Nigen S, Bolduc C. Efficacy and tolerability of topical tacrolimus ointment for the treatment of male genital psoriasis. J Cutan Med Surg 2008; 12: 230–234.
- Gribetz C, Ling M, Lebwohl M, Pariser D, Draelos Z, Gottlieb AB, et al. Pimecrolimus cream 1% in the treatment of intertriginous psoriasis: a double-blind, randomized study. J Am Acad Dermatol 2004; 51: 731–738.
- Freeman AK, Linowski GJ, Brady C, Lind L, Vanveldhuisen P, Singer G, et al. Tacrolimus ointment for the treatment of psoriasis on the face and intertriginous areas. J Am Acad Dermatol 2003; 48: 564–568.
- Lebwohl M, Freeman AK, Chapman MS, Feldman SR, Hartle JE, Henning A. Tacrolimus ointment is effective for facial and intertriginous psoriasis. J Am Acad Dermatol 2004; 51: 723–730.
- Brune A, Miller DW, Lin P, Cotrim-Russi D, Paller AS. Tacrolimus ointment is effective for psoriasis on the face and intertriginous areas in pediatric patients. Pediatr Dermatol 2007; 24: 76–80.
- Steele JA, Choi C, Kwong PC. Topical tacrolimus in the treatment of inverse psoriasis in children. J Am Acad Dermatol 2005; 53: 713–716.
- Oriba HA, Bucks DA, Maibach HI. Percutaneous absorption of hydrocortisone and testosterone on the vulva and forearm: effect of the menopause and site. Br J Dermatol 1996; 134: 229–233.
- Young Park J, Hyun Rim J, Beom Choe Y, Il Youn J. Facial psoriasis: comparison of patients with and without facial involvement. J Am Acad Dermatol 2004; 50: 582–584.
- Strober B, Ryan C, van de Kerkhof P, van der Walt J, Kimball AB, Barker J, et al. Recategorization of psoriasis severity: Delphi consensus from the International Psoriasis Council. J Am Acad Dermatol 2020; 82: 117–122.
- Carroll CL, Feldman SR, Camacho FT, Balkrishnan R. Better medication adherence results in greater improvement in severity of psoriasis. Br J Dermatol 2004; 151: 895–897.
- Billich A, Aschauer H, Aszódi A, Stuetz A. Percutaneous absorption of drugs used in atopic eczema: pimecrolimus permeates less through skin than corticosteroids and tacrolimus. Int J Pharm 2004; 269: 29–35.
- Paller AS, Lebwohl M, Fleischer ABJ, Antaya R, Langley RG, Kirsner RS, et al. Tacrolimus ointment is more effective than pimecrolimus cream with a similar safety profile in the treatment of atopic dermatitis: results from 3 randomized, comparative studies. J Am Acad Dermatol 2005; 52: 810–822.
- Van Leent EJM, Ebelin ME, Burtin P, Dorobek B, Spuls PI, Bos JD. Low systemic exposure after repeated topical application of pimecrolimus (Elidel), SD Z ASM 981) in patients with atopic dermatitis. Dermatology 2002; 204: 63–68.
- Siegfried EC, Jaworski JC, Kaiser JD, Hebert AA. Systematic review of published trials: long-term safety of topical corticosteroids and topical calcineurin inhibitors in pediatric patients with atopic dermatitis. BMC Pediatr 2016; 16: 75.
- Sigurgeirsson B, Boznanski A, Todd G, Vertruyen A, Schuttelaar MLA, Zhu X, et al. Safety and efficacy of pimecrolimus in atopic dermatitis: a 5-year randomized trial. Pediatrics 2015; 135: 597–606.
- Soter NA, Fleischer AB, Webster GF, Monroe E, Lawrence I. Tacrolimus ointment for the treatment of atopic dermatitis in adult patients: Part II, safety. J Am Acad Dermatol 2001; 44: S39–S46.
- Hanifin JM, Ling MR, Langley R, Breneman D, Rafal E. Tacrolimus ointment for the treatment of atopic dermatitis in adult patients: part I, efficacy. J Am Acad Dermatol 2001; 44: S28–S38.
- Halling-Overgaard AS, Kezic S, Jakasa I, Engebretsen KA, Maibach H, Thyssen JP. Skin absorption through atopic dermatitis skin: a systematic review. Br J Dermatol 2017; 177: 84–106.