SHORT COMMUNICATION
Cutaneous Adnexal Adenocarcinoma: Genomic Analysis and Successful HER2-targeted Therapy of Metastatic Disease
Yui HIRANO-LOTMAN1#, Yoshihiro ISHIDA1#, Yuichiro ENDO1, Seishi OGAWA2 and Kenji KABASHIMA1
1Department of Dermatology, Kyoto University Graduate School of Medicine, 54 Shogoin Kawahara-cho, Sakyo-ku, Kyoto 606-8507 and 2Pathology and Tumor Biology, Graduate School of Medicine, Kyoto University, Kyoto, Japan. E-mail: yendou-tky@umin.ac.jp
#These authors contributed equally.
Citation: Acta Derm Venereol 2023; 103: adv9399. DOI: https://doi.org/10.2340/actadv.v103.9399.
Copyright: © Published by Medical Journals Sweden, on behalf of the Society for Publication of Acta Dermato-Venereologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: May 30, 2023; Published: Aug 16, 2023
Competing interests and funding: The authors have no conflicts of interest to declare.
INTRODUCTION
Skin adnexal tumour (SAT) is a heterogeneous collection of skin neoplasms that arise from skin appendages. According to the WHO classification, SATs are further classified according to the histological features that are reminiscent of the site of origin. However, some SATs do not display any specific histological features; such tumours are termed cutaneous adnexal adenocarcinoma, not otherwise specified (AC-NOS). Due to its rarity, standardized therapies have not been established (1). To the best of our knowledge, this is the first report of metastatic AC-NOS analysed by whole exome sequencing (WES) and successfully treated with dual anti-HER2 antibodies and docetaxel, using the same regime as standardized in breast cancer (2).
CASE REPORT
A 79-year-old man presented with a dome-shaped nodule on the scalp, left cervical lymphadenopathy, and back pain. The scalp lesion was first noted 3 years previously and had gradually enlarged to the size of 3 cm in diameter. Computed tomography and magnetic resonance imaging revealed a bone metastasis occupying the 12th thoracic vertebra and multiple left cervical lymph node metastases (Fig. 1a).
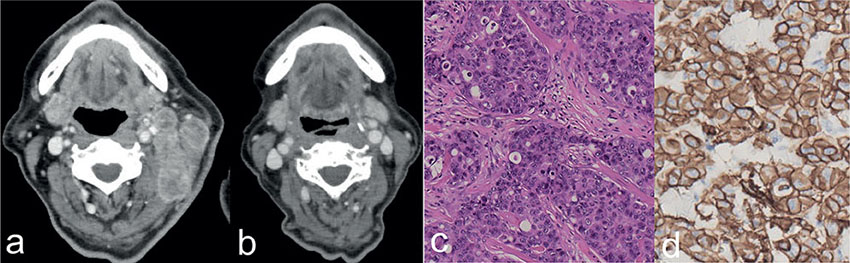
Fig. 1. Clinical and histological findings. (a, b) Computed tomography scans showing the cervical lymph node metastases of apocrine adenocarcinoma. (a) Before the administration of trastuzumab, pertuzumab, docetaxel, and concurrent radiation, and (b) 11 months after administration. (c) Histology of the nodule of the scalp. The lesion was high-grade adenocarcinoma with prominent nuclear pleomorphism and abnormal mitotic figures. Haematoxylin and eosin staining. (d) Immunohistochemical staining for human epidermal growth factor receptor protein 2 (HER2) revealed positive tumour cells. (c, d: x200).
The scalp tumour was resected with a 1-cm margin. Histological examination revealed poorly-differentiated, high-grade adenocarcinoma extending into the subcutaneous tissue (Fig. 1c). The tumour cells were positive for AR, Ki-67 (60%), GCDFP15 and HER2 (Fig. 1d), negative for ER and PR. HER2 gene amplification was also detected by fluorescence in situ hybridization. Because the tumour lacked specific histological features that allow further classification, the tumour was diagnosed as AC-NOS.
To clarify the mutation profile of AC-NOS, WES was performed. The tumour mutation burden was 4.2 mutations/MB (140 single nucleotide variants and 24 indels). A nonsense mutation in TP53 (p.E232X) and loss of heterozygosity at chromosome 17 were detected. These aberrations probably result in biallelic inactivation of TP53. Copy number alteration analysis detected extensive aneuploidy and focal amplification of HER2 (Fig. 2a). Pathogenic mutations were not detected in PIK3CA or RAS genes. The mutational signature was dominated by molecular clock signatures (COSMIC SBS1: 18.2%, SBS5: 61.7%) (Fig. 2b, c).
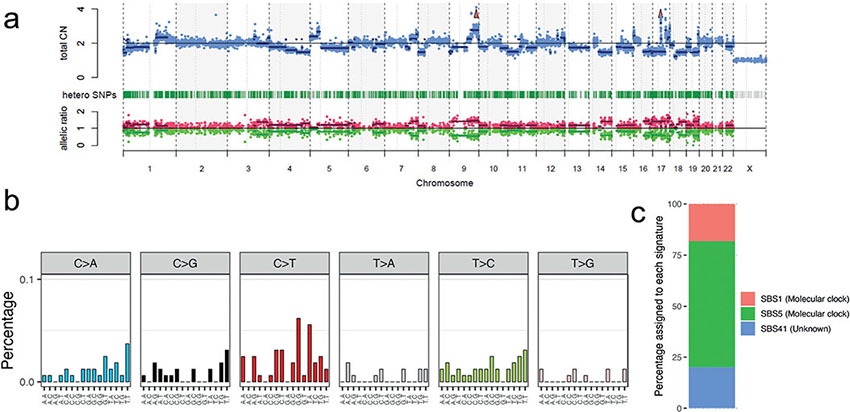
Fig. 2. Exome sequencing results. (a) Copy number aberrations profile inferred by by cnacs software (github.com/OgawaLabTumPath/CNACS). Upper panel: total copy number; lower panel: B allele frequency. (b) Distribution of 96 trinucleotide mutation spectrum. (c) Relative contributions of mutational signatures inferred by deconstructSigs software (github.com/raerose01/deconstructSigs).
The combination of dual HER2 antibodies (pertuzumab and trastuzumab) with docetaxel was administered to the patient. We also administered concurrent radiotherapy to the metastatic sites. A complete response was achieved after 2 cycles (Fig. 1b), but docetaxel was subsequently discontinued due to grade 4 neutro-paenia. Only dual HER2 antibodies were continued for additional 10 cycles as maintenance treatment. The patient remained in complete remission 20 months after treatment cessation.
DISCUSSION
There is no large-scale genomic study that specifically analysed AC-NOS. A recent study analysed a cohort of diverse malignant SATs, including AC-NOS, and found that HER2 overexpression was detected in 2/5 AC-NOS and detected mutations in TP53 (1/5), and CDKN2A (1/5) using targeted sequencing (3). A WES approach found that driver mutations were similar to previous reports. Interestingly, AC-NOS was devoid of ultraviolet (UV) mutational signature, which is characteristic of keratinocyte carcinomas (4, 5). We suspect that the tumour had resided deep in the dermis or subcutaneous layer before it became clinically visible, because UV-B rays, the most mutagenic spectrum of UV, do not penetrate 10–50 mm into the skin (6).
To the best of our knowledge, there is 1 previous case report that describes a patient with AC-NOS who initially achieved clinical resolution while being treated with carboplatin, paclitaxel, and trastuzumab (7). We decided to administer dual anti-HER2 antibodies and docetaxel as the first-line therapy because 1 case of metastatic cutaneous apocrine adenocarcinoma successfully underwent neoadjuvant dual HER2 antibodies and docetaxel (8).
The current case exemplifies the usefulness of intensive genomic and histological analyses in guiding treatments for rare cancers and revealed genomic aberrations of AC-NOS.
ACKNOWLEDGEMENTS
This work was supported by JSPS KAKENHI grant number 21K16210.
All procedures used in this research were approved by the ethics committee of Kyoto University Hospital. The patients in this manuscript have given written informed consent to publication of their case details.
REFERENCES
- De Iuliis F, Amoroso L, Taglieri L, Vendittozzi S, Blasi L, Salerno G, et al. Chemotherapy of rare skin adnexal tumors: a review of literature. Anticancer Res 2014; 34: 5263–5238.
- Swain SM, Miles D, Kim SB, Im YH, Im SA, Semiglazov V, et al. CLEOPATRA study group. Pertuzumab, trastuzumab, and docetaxel for HER2-positive metastatic breast cancer (CLEOPATRA): end-of-study results from a double-blind, randomised, placebo-controlled, phase 3 study. Lancet Oncol 2020; 21: 519–530.
- Cavalieri S, Busico A, Capone I, Conca E, Dallera E, Quattrone P, et al. Identification of potentially druggable molecular alterations in skin adnexal malignancies. J Dermatol 2019; 46: 507–514.
- Chang D, Shain AH. The landscape of driver mutations in cutaneous squamous cell carcinoma. NPJ Genom. Med 2021; 6: 61.
- Bonilla X, Parmentier L, King B, Bezrukov F, Kaya G, Zoete V, et al. Genomic analysis identifies new drivers and progression pathways in skin basal cell carcinoma. Nat Genet 2016; 48: 398–406.
- Meinhardt M, Krebs R, Anders A, Heinrich U, Tronnier H. Wavelength-dependent penetration depths of ultraviolet radiation in human skin. J Biomed Opt 2008; 13: 044030.
- Brown TJ, Sher DJ, Nedzi LA, Hughes RS, Beg MS, Mull J, et al. Cutaneous adnexal adenocarcinoma with exquisite sensitivity to trastuzumab. Head Neck 2017; 39: E69–71.
- Otsuka M, Yamasaki O, Kaji T, Shien T, Iwatsuki K. Metastatic cutaneous apocrine adenocarcinoma treated with a combination of pertuzumab-based targeted therapy and taxane chemotherapy: a case report. JAMA Dermatol 2016; 152: 111–113.