ORIGINAL ARTICLE
Gestational diabetes alters subgingival pathobiont composition
Fatih Cömerta, Funda Yalçına, Nursen Topçuoğlub, Oya Kaya-Şimşeka, Oya Demircic, Silvi Domnoria,d and Ulku Basera
aDepartment of Periodontology, Faculty of Dentistry, Istanbul University, Istanbul, Türkiye; bDepartment of Oral Microbiology, Faculty of Dentistry, Istanbul University, Istanbul, Türkiye; cDepartment of Perinatology, Zeynep Kamil Women and Children’s Diseases Training and Research Hospital, Health Science University, Istanbul, Türkiye; dDepartment of Periodontology, Faculty of Dentistry, Institute of Graduate Studies in Health Sciences, Istanbul University, Istanbul, Türkiye
ABSTRACT
Objective: This cross-sectional study assesses the relationship between gestational diabetes mellitus (GDM) and periodontal dysbiosis by evaluating specific periodontal pathobionts (Porphyromonas gingivalis, Prevotella intermedia, Tannerella forsythia, and Treponema denticola), clinical periodontal parameters, and periodontal inflamed surface area (PISA).
Materials and methods: 101 GDM and 98 non-GDM women of 16–36 weeks gestation were included. Clinical periodontal parameters were measured, and PISA values calculated. Subgingival plaque samples, collected from the deepest pockets, were analyzed, to assess the amount of periodontopathogens. Associations were assessed using binary logistic regression and path analysis.
Results: The GDM group showed higher clinical parameters and PISA values (p < 0.001). While P. gingivalis levels were similar in both groups (p = 0.924) and unrelated to the presence of GDM in path analysis (p = 0.055), P. intermedia and T. denticola levels were found higher in the non-GDM group (p < 0.05, p < 0.001) and negatively associated with the presence of GDM (p < 0.001, p = 0.002). P. intermedia increased with gestation week (p = 0.044). Elevated T. forsythia levels were observed to increase GDM risk 1.208-fold in regression analysis (p = 0.002).
Conclusion: Increasing sex hormone levels accompanied by the presence of GDM may alter subgingival pathobiont composition. In patients with GDM, there is a heightened burden of T. forsythia in subgingival zones, where P. intermedia finds less favorable conditions.
KEYWORDS: pregnancy; gestational diabetes mellitus; subgingival pathobionts; periodontal inflammation
Citation: ACTA ODONTOLOGICA SCANDINAVICA 2026; VOL. 85: 179–189. DOI: https://doi.org/10.2340/aos.v85.45789.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Odontologica Scandinavica Society. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), allowing third parties to copy and redistribute the material in any medium or format and to remix, transform, and build upon the material, with the condition of proper attribution to the original work.
Received: 14 December 2025; Accepted: 10 March 2026; Published: 15 April 2026.
CONTACT: Ulku Baser baserulk@istanbul.edu.tr Istanbul University, Faculty of Dentistry, Department of Periodontology, Süleymaniye, Prof. Dr. Cavit Orhan Tütengil Sk. No:4 D:6, 34116, Fatih, Istanbul, Türkiye
Competing interests and funding: The authors report there are no competing interests to declare.
This study was funded by the Scientific Research Projects Coordination Unit of Istanbul University (Project No: 34298), Istanbul, Türkiye.
Introduction
The etiopathogenesis of periodontal diseases is described by the polymicrobial synergy and dysbiosis model of Hajishengallis & Lamont [1]. This model suggests that pathobionts and certain keystone pathogens proliferate in proportion to symbiotic bacteria. The immune subversion mechanisms that these microorganisms possess allow them to induce an inflammatory response, which, despite its activation, fails to translate into an effective immune response. Consequently, inflammophilic bacteria capitalize on the inflammatory environment, intensifying the inflammation and accelerating periodontal destruction [2].
The transition into a diseased condition is significantly influenced by immunopathogenesis, which, is itself shaped by risk factors, including genetic predisposition, environmental factors, and systemic conditions [3].
As a metabolic disease characterized by systemic inflammation, diabetes has long been studied in conjunction with periodontitis, to explore their potentially common inflammatory etiopathogenesis. The interplay between diabetes and periodontal inflammation is well-documented, with overt diabetes exacerbating periodontitis and vice versa, that is, periodontitis potentially worsening glycemic control [3–5].
Gestational diabetes mellitus (GDM) is defined as glucose intolerance with onset diagnosis during pregnancy. It is characterized by insulin resistance stemming from the effect of sex steroid hormones [6]. The blocking effect these hormones may exert on insulin is described as a contra-insulin effect, usually observed at about 20–24 weeks of pregnancy [7]. Inflammation represents an important and frequent contributor to complications of pregnancy [8]. Globally, GDM represents the majority (84%) of hyperglycemia cases in pregnancy, as stated by the International Diabetes Federation (IDF), which estimates that one in six live births are affected by maternal hyperglycemia [9].
Both pregnancy and diabetes impact periodontal health independently. GDM, being a pregnancy-specific form of diabetes, is a global problem affecting both maternal and infant health. Its interaction with periodontal disease may influence maternal and infant health locally and systemically, by altering the periodontal microbiota.
During pregnancy, there is a marked rise in estrogen and progesterone levels [10]. Fluctuations in these hormone levels, specifically increasing until the 8th month of pregnancy, can lead to higher vascular permeability [11]. This change often manifests within the oral cavity as pregnancy gingivitis, with increases in probing pocket depth (PPD) and bleeding on probing (BoP) [12, 13]. The resulting inflammatory condition fosters the presence of bacteria that thrive in a periodontally-compromised oral environment, which is also exacerbated by glycemic dysregulation and altered immune response.
Several bacterial species have been shown to increase during pregnancy. Porphyromonas gingivalis stands out as a keystone pathogen [1] with potent virulence factors such as gingipains and endotoxins promoting periodontal damage even at low abundance. It has been associated with worsening gingival inflammation during pregnancy [14] as well as pregnancy complications [15–17]. Bacteria such as Prevotella intermedia and Prevotella melaninogenica can utilize estradiol and progesterone as substitutes for vitamin K, promoting their proliferation in the subgingival environment [11, 18]. Studies have reported a peak in P. intermedia levels during the second trimester, which coincides with heightened gingival inflammation and subsides postpartum [11, 19–22]. These shifts in the subgingival microbiota also include other pathogenic species, such as Tannerella forsythia and Treponema denticola, both members of the red complex detected in the subgingival microbiota during pregnancy, with their virulence factors contributing to epithelial invasion, immune evasion, and enhanced tissue breakdown [15, 23]. Accumulated evidence implicates the role of P. gingivalis, T. forsythia, T. denticola, and P. intermedia, in pregnancy complications, including preterm delivery [15–17, 24–27]. These four bacterial species were selected for analysis based on their well-established role in periodontal disease progression and their reported associations with pregnancy complications. A recent systematic review [28] has highlighted the underexplored biological mechanisms connecting periodontal disease and GDM; hence, investigating the abundance of these bacteria in the context of GDM could provide a targeted frame to reflect potential microbial and clinical patterns.
The null hypothesis of our study states that GDM patients have the same periodontal microbiota as a pregnant woman without GDM. In addition to investigating the relationship between periodontopathogens and GDM, we calculated the periodontally inflamed surface area (PISA) to quantify the inflammatory condition in both patient groups. The aim of this study is to evaluate the subgingival pathobiont composition of pregnant women with and without gestational diabetes and investigate its associations with clinical parameters and PISA.
Materials and methods
Study design and population
This cross-sectional study was conducted between March 2019 and June 2021 at two main research hospitals in Istanbul, Turkiye: (1) the Division of Endocrinology and Metabolism, Department of Internal Medicine, Faculty of Medicine, Istanbul University; and (2) the Department of Obstetrics and Gynecology, Zeynep Kamil Gynecology and Paediatrics and Research Hospital. The study was conducted and reported in accordance with the STROBE guidelines for observational studies [29]. Out of 1122 patients examined, 101 GDM and 98 non-GDM patients were included (Figure 1).
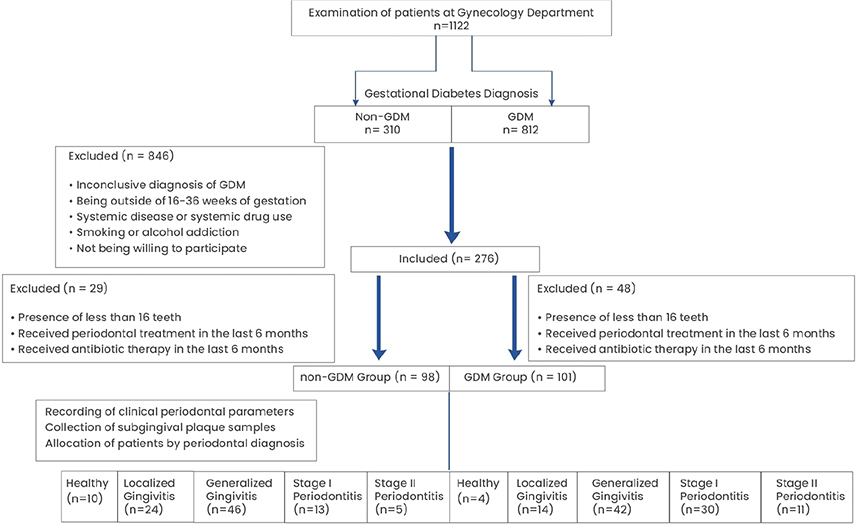
Figure 1. Flowchart of the study.
Inclusion criteria:
- Gestational age of 16–36 weeks.
- Diagnosed with or without GDM and no other systemic diseases.
- Have more than 16 teeth excluding third molars.
Exclusion criteria:
- History of cardiovascular, cerebrovascular, peripheral arterial, respiratory diseases, mental disorders (e.g. depression), rheumatoid arthritis, and osteoporosis.
- Smoking or alcohol addiction.
- Use of antibiotics within 6 weeks prior to sampling.
- Use of medication affecting periodontal tissues.
This study was conducted in accordance with the Declaration of Helsinki as per the 2013 revision and approved by the Clinical Research Ethics Committee of Istanbul University Faculty of Dentistry (Approval No: 2019/22). Informed consent was obtained from all patients prior to their inclusion in the study.
Diagnosis of GDM
GDM was diagnosed according to the American Diabetes Association criteria (2013), that is, a 75 g oral glucose tolerance test was administered at 16–36 weeks of gestation, with fasting plasma glucose measured at 1 and 2 h. Threshold values for GDM diagnosis were fasting ≥ 92 mg/dl (5.1 mmol/l); 1 h ≥ 180 mg/dl (10.0 mmol/l); and 2 h ≥ 153 mg/dl (8.5 mml/l).
Clinical examination
Demographic information, medical and dental anamnesis of the participants were recorded via a questionnaire.
Clinical periodontal parameters including gingival index (GI) [12], plaque index (PI) [30], PPD, clinical attachment level (CAL), and BoP [31] were recorded. Oral examination was performed using a Williams* type periodontal probe, recording measurements at six sites per tooth (mesiobuccal, midbuccal, distobuccal, mesiolingual, midlingual and distolingual). All measurements were performed by a single investigator (F.C.).
To quantify the amount of inflamed periodontal tissue and as such, the inflammatory burden, we calculated PISA values, using the formula by Nesse et al. [32], based on CAL/PPD, gingival recession and BoP data. PISA is expressed in square millimeters (mm2), representing the total surface area of the bleeding pocket epithelium.
Clinical diagnosis was done according to the 2017 Classification of Periodontal and Peri-Implant Diseases and Conditions [33]. The patients were categorized into five groups: Healthy, with Localized Gingivitis, with Generalized Gingivitis, with Periodontitis Stage I or with Periodontitis Stage II.
Subgingival plaque sampling
Subgingival plaque samples were collected from each participant’s four teeth with the deepest pockets. Cotton pellets were placed in the buccal and lingual sulci to prevent salivary contamination. Supragingival plaque was carefully wiped from the tooth surfaces using a sterile cotton pellet, and then subgingival plaque samples were obtained using standard absorbent paper points (#40). A total of 16 samples were taken from 4 sites per tooth (mesiobuccal, distobuccal, mesiolingual/-palatal and distolingual/-palatal) where paper points were inserted and left in place for 30 s. Samples contaminated with blood during collection were excluded. The collected paper points were placed in two Eppendorf tubes of eight samples each. Samples were initially stored in a refrigerator at +4°C, then transferred to −80°C ultra-low temperature freezers (New Brunswick Scientific, USA) for long-term storage.
DNA extraction procedure from plaque sample
Genomic DNA was extracted from paper points collected from patients using E.Z.N.A Bacterial DNA Kit (Omega Bio-tek†). Initially, 100 µl of TE buffer was added to the paper points which were then vortexed thoroughly, and subsequently, manufacturer’s protocol was followed as indicated. Finally, DNA was eluted in 60 µl of elution buffer. DNA concentration and purity were assessed using a NanoDrop spectrophotometer (Thermo Fisher Scientific‡), measuring absorbance at 230, 260, and 280 nm and calculating 260/280 and 260/230 ratios. Isolated DNA samples were stored at −20°C until further analysis.
Quantitative PCR analysis
Species-specific quantitative PCR (qPCR) method was used to determine the relative amounts of P. gingivalis, P. intermedia, T. forsythia, and T. denticola in the isolated DNA samples. The specific primers for each target bacteria are shown in Table 1.
Each 20 μL reaction mixture contained 10 μL of 2x GoTaq qPCR Master Mix (Promega), 0.5 μM of each primer, 2 μL of template DNA, and nuclease-free water. Amplification was conducted with an initial enzyme activation at 95°C for 2 min, followed by 40 cycles of 95°C for 15 s and 60°C for 1 min. A melting curve analysis (60–95°C) was performed to confirm amplification specificity. Two no-template controls were included in each plate to rule out possible contamination. All reactions were performed in a LightCycler480 Real-Time PCR System (Roche§).
Following qPCR, Ct values were obtained and used for relative quantification analysis. Comparative analyses between patient groups were performed by using fold changes calculated by Ct values. For visualization purposes, Ct values were normalized to the average Ct values (Ct_sample – Ct_average) of positive samples for each target microorganism. Group and sampling time comparisons were made using the fold-change rates obtained for each sample.
The primary outcome measure of this study is the quantification of P. gingivalis, P. intermedia, T. forsythia, and T. denticola in subgingival plaque samples using qPCR to compare bacterial loads between pregnant women with and without GDM. The secondary outcome measures include clinical periodontal parameters, PISA, and periodontal diagnosis classification to assess differences in periodontal health between the groups. The bacterial quantification values and all clinical periodontal parameters were treated as continuous variables, while sociodemographic features and periodontal diagnosis groups were categorized for statistical analysis.
Statistical analysis
Our reference in estimating sample size were the studies by Gogeneni et al. [34] and Ganiger et al. [35]. The sample size was calculated using G*Power (version 3.1.9.2¶) with a two-sided independent samples t-test, Type 1 error of 5%, Type 2 error of 20% corresponding to 80% Power, and an effect size of 0.45, yielding 79 participants per group. To account for potential losses, 100 participants per group were planned, totaling to a sample size of 200.
Data were analyzed using IBM SPSS V23‖. Normality was assessed with the Kolmogorov-Smirnov Test. Due to violations of normality assumptions in several variables, non-parametric analyses were conducted. Mann Whitney U Test was used for group comparisons of continuous variables. Categorical data were analyzed using the Pearson Chi-Square Test, Yates’ correction, or Fisher’s Exact Test, as appropriate. Binary logistic regression analysis was used to examine the risk factors affecting GDM. Model fit was evaluated using the Hosmer-Lemeshow goodness-of-fit test. Bacterial count was compared across the periodontal condition subgroups using Mann Whitney U test. Path analysis was used to identify potential factors affecting bacterial count, where clinical periodontal parameters, systemic and behavioral factors were treated as predictors, based on biological plausibility and existing literature [11, 14, 15, 36, 37]. Results were presented as number (percentage) for categorical data and mean ± standard deviation and median (minimum–maximum) for quantitative data, with significance set at P < 0.05 for all analyses.
Results
Continuous variables were categorized for descriptive comparison. Age was grouped as 17–25, 26–35, and ≥ 36 years [38,39]. BMI was classified according to the WHO criteria as < 25 (normal), 25–29.9 (overweight), and ≥ 30 kg/m² (obese) [40]. Education level and income levels were categorized into low, middle, and high based on self-reported answers; numeric thresholds were not applied. Number of children and pregnancies were grouped into clinically relevant ranges (e.g. 0–1, 2, and ≥ 3). Brushing habits and interdental cleaning habits were categorized by frequency as none, irregular and twice or once a day, respectively. Statistically significant differences were found between the groups for age, BMI, number of children, and socio-economic level (P < 0.001). Number of pregnancies differed between the groups with GDM group participants mostly on their third pregnancy and above, while the non-GDM group participants were either in their first or second pregnancy (P = 0.009). Gestational week was found to be similar as 28.56 ± 3.6 in the non-GDM group and 28.95 ± 2.5 in the GDM group (P = 0.866). Brushing habits were found to be significantly different between the groups, while interdental cleaning was similar and very close to statistical significance (P < 0.001 and P = 0.052, respectively) (Table 2).
| Group | p | ||
| Non-GDM (n = 98) | GDM (n = 101) | ||
| Age (year) | |||
| Median (Min–Max) | 28 (17–44) | 34 (20–43) | < 0.001b |
| Ages 17–25 | 27 (27.6) | 11 (10.9) | < 0.001c |
| Ages 26–35 | 52 (53.1) | 56 (55.4) | |
| Ages 36 and above | 19 (19.4) | 34 (33.7) | |
| BMI n (%) | |||
| Median (Min–Max) | 27.8 (18.7–44.9) | 31.6 (19.5–46.7) | < 0.001b |
| Normal weight (BMI < 25) | 28 (28.6) | 8 (7.9) | < 0.001a |
| Overweight (BMI 25–29.9) | 35 (35.7) | 31 (30.7) | |
| Obesity (BMI ≥ 30) | 35 (35.7) | 62 (61.4) | |
| Education level n (%) | |||
| Low (Literate/Elementary) | 44 (44.9) | 61 (60.4) | 0.03c |
| Middle (Secondary education) | 22 (22.4) | 17 (16.8) | |
| High (University) | 32 (32.7) | 23 (22.8) | |
| Income rate n (%) | |||
| Low | 61 (62.2)A | 76 (75.2)A | 0.026c |
| Middle | 23 (23.5)A | 20 (19.8)A | |
| High | 14 (14.3)A | 5 (5)B | |
| GDM history % | |||
| No GDM history | 44 (97.8)A | 49 (69)A | < 0.001c |
| GDM history present | 1 (2.2)A | 22 (31)B | |
| Nr. of children n (%) | |||
| 0–1 child | 77 (78.6) | 56 (55.4) | < 0.001c |
| 2 children | 17 (17.3) | 31 (30.7) | |
| 3 children and above | 4 (4.1) | 14 (13.9) | |
| Brushing habits n (%) | |||
| No brushing | 32 (32.7)A | 9 (8.9)B | < 0.001a |
| 2 times a day | 34 (34.7)A | 30 (29.7)A | |
| Irregular | 32 (32.7)A | 62 (61.4)B | |
| Interdental cleaning n (%) | |||
| No cleaning | 65 (66.3) | 82 (81.2) | 0.052 |
| Irregular | 26 (26.5) | 16 (15.8) | |
| Once a day | 7 (7.1) | 3 (3) | |
| GDM: gestational diabetes mellitus; BMI: body mass index. aPearson Chi-Square Test; bMann Whitney U; cFisher’s Exact Test used for 2x2 comparisons or Somers’d/Gamma association for ordinal data, used where appropriate; A–B: There is no difference between groups with the same letter; Significance set at p < 0.05. |
|||
Statistically significant differences were observed in PI, GI, PPD, BoP, and CAL values between the groups, with all parameters higher in the GDM group (P = 0.006 for PI and P ≤ 0.001 for the other parameters). PISA values were also significantly higher in the GDM group compared to the non-GDM group (P < 0.001) (Figure 2).
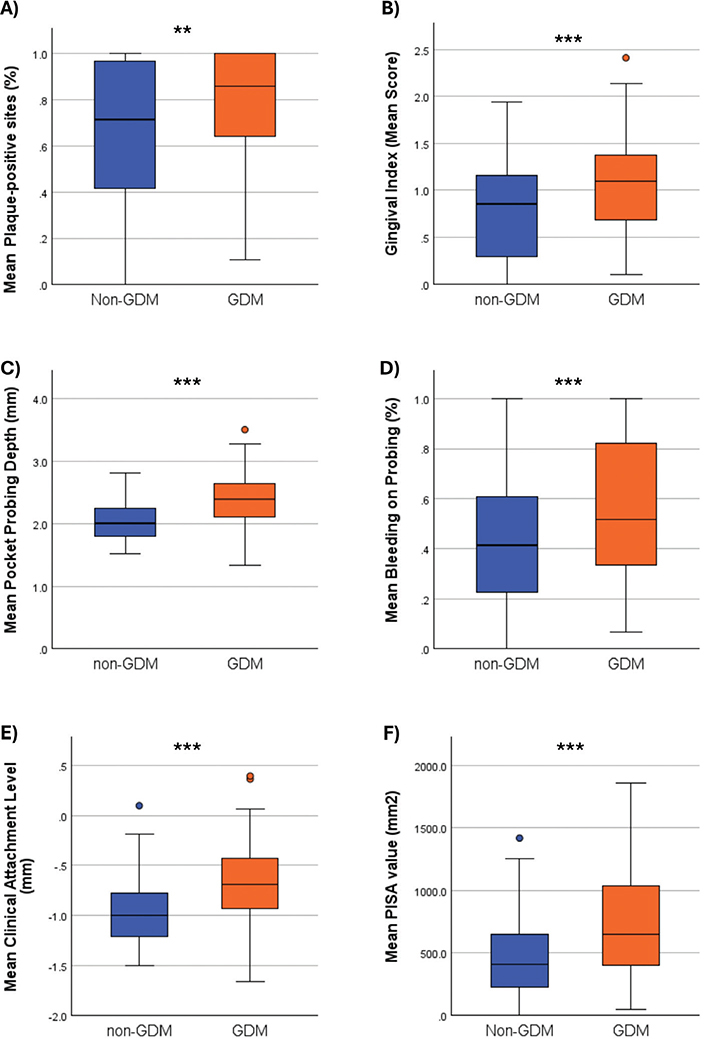
Figure 2. Comparison of periodontal parameters (A) PI: Plaque Index; (B) GI: Gingival Index, (C) PPD: Periodontal Pocket Depth; (D) BoP: Bleeding on Probing; (E) CAL: Clinical Attachment Loss; (F) PISA: Periodontal inflamed surface area. Mann Whitney U Test. * p < 0.05. ** p < 0.01. *** p < 0.001.
Of the 98 non-GDM patients, bacterial counts were evaluated in 92; among the 101 GDM patients, only one dataset was missing. The exact n values for each subgroup are indicated in Figure 3. No statistically significant difference was found for P. gingivalis prevalence between the GDM and non-GDM groups (P = 0.924). However, P. intermedia (P = 0.044), T. forsythia (P = 0.002) and T. denticola (P < 0.001) distributions differed significantly with T. forsythia more frequent in the GDM group, while P. intermedia and T. denticola were significantly more prevalent in the non-GDM group (Figure 3).
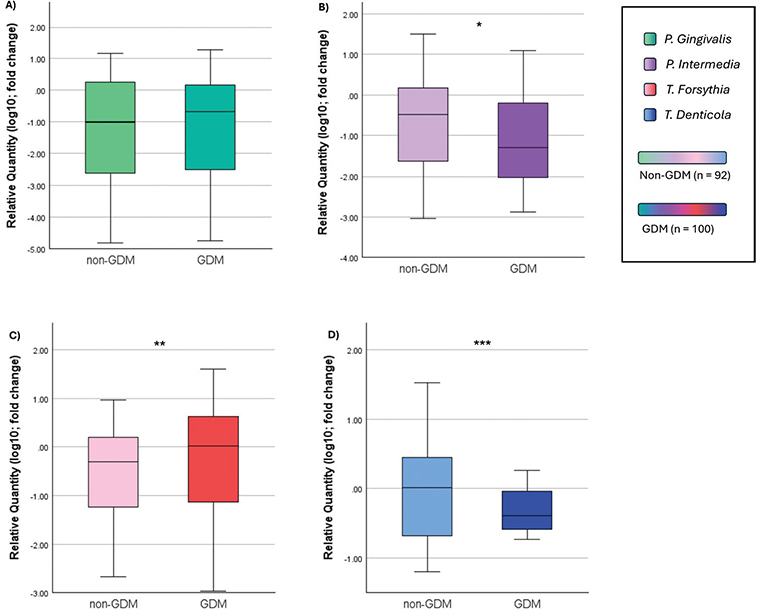
Figure 3. Periodontopathogen distribution without classification of periodontal disease. (A) P. gingivalis – Porphyromonas gingivalis; (B) P. intermedia: Prevotella intermedia; (C) T. forsythia: Tanerella forsythia; (D) T. denticola – Treponema denticola. Values were transformed using log(10) to normalize distributions for graphical visualization only; statistical analysis was conducted on raw data. Mann Whitney U test. * p < 0.05. ** p < 0.01. *** p < 0.001.
No significant difference in bacterial levels was observed in healthy periodontium between GDM and non-GDM groups (Figure 4).
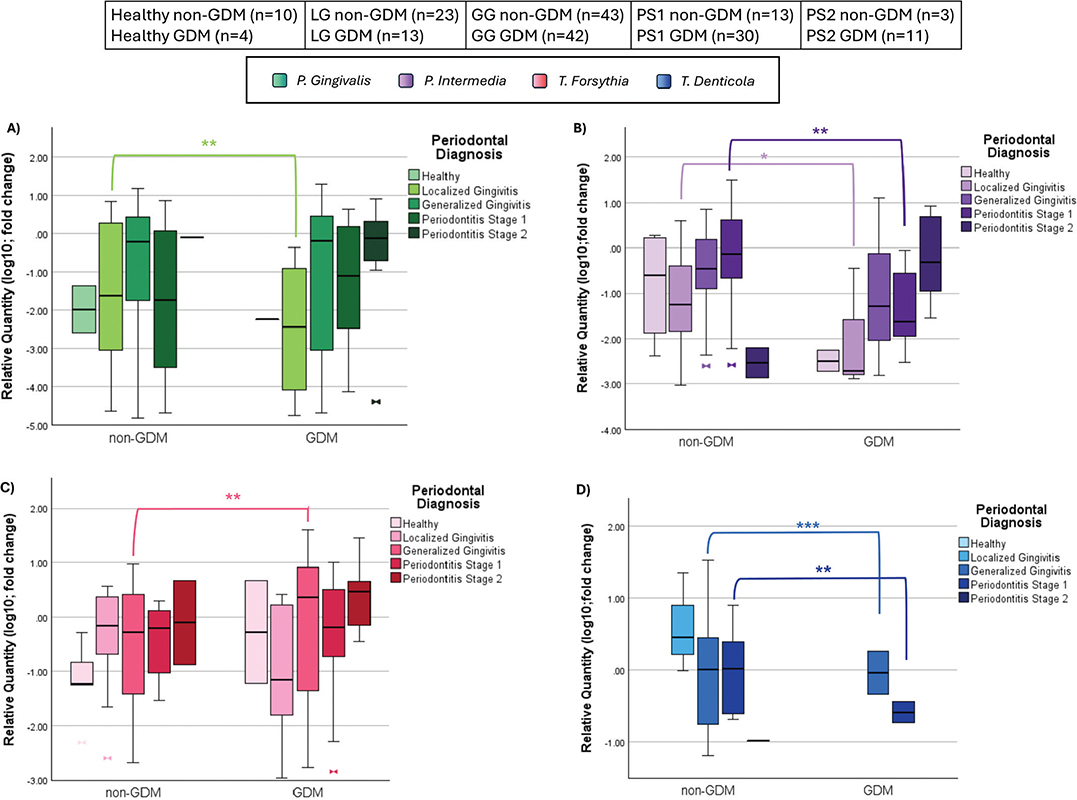
Figure 4. (A) Periodontopathogen distribution across diagnosis in P. gingivalis; (B) Periodontopathogen distribution across diagnosis in P. intermedia; (C) Periodontopathogen distribution across diagnosis in T. forsythia. (D) Periodontopathogen distribution across diagnosis in T. denticola (LG – Localized Gingivitis, GG – Generalized Gingivitis, PS1 – Periodontitis Stage 1, PS2 – Periodontitis Stage 2); non-GDM: pregnant without gestational diabetes mellitus, GDM: pregnant with gestational diabetes mellitus, n: number). Values were transformed using log(10) to normalize distributions for graphical visualization only; statistical analysis was conducted on raw data. Mann Whitney U Test. * p < 0.05. ** p < 0.01. *** p < 0.001.
P. gingivalis and P. intermedia distributions differed significantly between 13 GDM and 23 non-GDM women diagnosed with localized gingivitis (Figure 4A and B; P = 0.004), while T. forsythia and T. denticola showed no significant difference (Figure 4C and D).
No significant differences were found for P. gingivalis and P. intermedia levels between 42 GDM and 43 non-GDM women with generalized gingivitis. T. forsythia levels were higher in the GDM group (median 1.93 vs. 0.14; P = 0.022) while T. denticola levels were higher in the non-GDM group (P = 0.001) (Figure 4).
Among 30 GDM and 13 non-GDM women with stage I periodontitis, P. intermedia differed significantly (median 0 in GDM vs. 0.44 in non-GDM; p = 0.016) and T. denticola also showed a significant difference (P = 0.008); however, no significant differences were observed for P. gingivalis (P = 0.642) or T. forsythia (P = 0.403) (Figure 4).
There was no statistically significant difference between bacterial levels in 11 GDM and 3 non-GDM women with stage II periodontitis (P = 0.301) (Figure 4).
Regression analysis revealed that the risk of GDM increases with age (1.114 times, P < 0.001), number of children (1.746 times, P < 0.001), PI (5.241 times, P = 0.002), BoP (7.284 times, P < 0.001) and SCD (11,898 times, P < 0.001). Additionally, higher levels of T. forsythia (1.208 times, P = 0.002), increased periodontal disease severity (1.717 times, P = 0.001) and PISA values (1.002 times, P < 0.001) were also associated with increasing GDM risk. Notably, the risk of GDM was 19,755 times higher in those with GDM history from their previous pregnancy (P = 0.004). Table 3 presents unadjusted odds ratios (ORs), 95% confidence intervals (CIs), and p-values for each independent variable.
Significant path coefficients were identified for all the analyzed bacteria. For P. gingivalis, an increase in PI by one unit corresponded to a rise of 1.159 units (p = 0.039), while a unit increase in the GI led to a decrease of 0.856 units (P = 0.006). Similarly, P. gingivalis levels increased by 0.003 units with each unit increase in PISA value (P < 0.001). Compared to healthy individuals, levels of P. gingivalis were 0.931 units lower in stage I periodontitis (P = 0.018) and 1.316 units lower in stage II periodontitis (P = 0.028) (Figure 5).
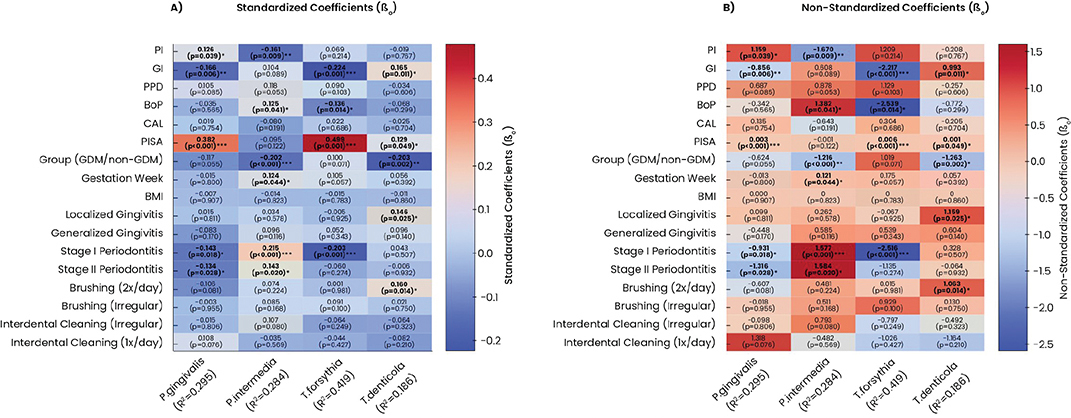
Figure 5. Heatmap of path analysis results with A. standardized and B. non-standardized coefficients. β0: Standardized Coefficients; β1: Non-Standardized Coefficients; P. gingivalis: Porphyromonas gingivalis; P. intermedia: Prevotella intermedia; T. denticola: Treponema denticola; T. forsythia: Tannerella forsythia; PI: Plaque Index; GI: Gingival Index; PPD: Periodontal Pocket Depth; BoP: Bleeding on Probing; CAL: Clinical Attachment Loss; PISA: Periodontally Inflamed Surface Area; non-GDM: pregnant without gestational diabetes mellitus; GDM: pregnant with gestational diabetes; BMI: Body Mass Index. * p < 0.05. ** p < 0.01. *** p < 0.001.
For P. intermedia, a one-unit rise in PI resulted in a decrease of 1.67 units (P = 0.009), while a unit increase in BoP increased its levels by 1.382 units (P = 0.041). The presence of GDM corresponded to 1.216 fewer units of P. intermedia compared to the non-GDM group (P < 0.001). Each additional week of gestation increased its levels by 0.121 units (P = 0.044) Those with stage I periodontitis showed an increase of 1.577 units (P < 0.001), while stage II periodontitis was associated with a 1.584-unit increase (P = 0.020) as compared to periodontally healthy individuals (Figure 5).
For T. forsythia, a one-unit increase in GI and BoP led to decreases of 2.217 units (P < 0.001) and 2.539 units (P = 0.014), respectively. In contrast, PISA value was positively associated with bacterium levels rising by 0.006 units per unit increase (P < 0.001). Compared to periodontally healthy individuals, T. forsythia levels were 2.516 units lower in stage I periodontitis participants (Figure 5).
For T. denticola, GI increases corresponded to a 0.933-unit rise (P = 0.011), and PISA value increases led to a 0.001-unit rise (P = 0.049). In individuals with GDM, T. denticola levels were 1.263 units lower than non-GDM individuals (P = 0.002). Healthy individuals had 1.159 more units compared to those with localized gingivitis (P = 0.025), while non-brushers had 1.063 more units than those brushing twice daily (P = 0.014) (Figure 5).
Discussion
The surge of sex steroid hormones observed during pregnancy may be a cause for alterations in periodontal tissues and microbial profile responsible for periodontal disease. On the other hand, these hormones can lead to a condition known as insulin resistance, by reducing the effectiveness of insulin [7]. Our aim in this study was to evaluate how the systemic burden of these two different conditions manifests in the local oral tissues through periodontal pathobionts. Our findings indicate that periodontal microbiota and periodontal inflammation differ between patients with GDM and those without GDM.
According to our clinical parameters, periodontal inflammation was found higher in the GDM group. While P. intermedia and T. denticola were found higher in the non-GDM group, T. forsythia was found higher in the GDM group. P. gingivalis was found to be associated with the severity of periodontal disease rather than the presence or absence of GDM. Its levels in the subgingival plaque samples showed a statistically significant relationship with PISA values (P < 0.001) and it was more prevalent in patients with stage I and II periodontitis. Overall, the higher inflammatory burden in the GDM group with larger inflamed sulcular surface and increased bleeding [41] may shift the subgingival niche by increasing the availability of host-derived substrates within a low-redox environment. This shift may generate a nutrient-rich medium which can selectively favor these inflammophilic pathogens [42].
Individuals with GDM often present with older age, higher BMI, greater parity, a history of GDM in previous pregnancies, and less favorable sociodemographic conditions [43]. These characteristics were also observed in our study, consistent with existing literature [44]. Many studies have identified age as a risk factor for GDM development [34, 35, 45]. In addition to age, the difference we observed in periodontal condition could also be attributed to behavioral factors. However, due to the systemic burden of gestational diabetes, the distribution of microbiota still shows some differences regardless of these risk factors.
To our knowledge, the existing literature specifically investigating the presence of periodontopathogens in oral microflora in relation to gestational diabetes includes the studies by Gogeneni et al. [34], Ganiger et al. [35] and Dasanayake et al. [43].
Gogeneni et al. reported GDM to be associated with heightened infection by P. gingivalis, Filifactor alocis, and T. denticola, where the latter was only found statistically higher in GDM patients with gingivitis and an increase in systemic C-Reactive Protein levels, which contrasts our findings. However, Gogeneni et al. performed a conventional PCR analysis with binary results to evaluate the bacterial presence in saliva samples, which is not a specific reflection of periodontal microbial flora [34].
Ganiger et al. [35] found P. gingivalis and P. intermedia to be higher in GDM patients. Conversely, Dasanayake et al. [43] found no difference in P. gingivalis levels between GDM, and healthy pregnant women based on dental plaque and vaginal samples which is parallel to our results.
P. intermedia, the main pathogen responsible for pregnancy gingivitis [18–21], thrives in a progesterone-rich environment, linking it to pregnancy-related periodontal disease, as also demonstrated by previous studies [46, 47]. In our study, P. intermedia levels were significantly higher in the non-GDM group (P < 0.001). P. intermedia was detected in increasing numbers as the gestation week progressed. This trend coincides with the hormonal surge going into the third trimester, supporting the relationship between P. intermedia and pregnancy hormonal changes. Its relationship with BoP could be attributed to vascular changes at the bottom level of gingival pocket depth due to the effect of sex hormone surge during pregnancy [20].
T. forsythia, one of the main culprits of periodontal disease and tissue destruction, is frequently isolated from areas where the disease is actively progressing [23]. In our study, T. forsythia levels were significantly higher in areas with elevated GI and PISA value, which supports the thesis that it is responsible for active disease. T. forsythia levels were also significantly higher in GDM individuals (P = 0.002), suggesting its active involvement in periodontal disease and its modulation by GDM factors. Dasanayake et al. [43] reported higher T. forsythia levels in the vaginal fluids of GDM patients but found no difference in serum IgG or plaque samples. Similarly, Akherati et al. [48] observed higher prevalence of T. forsythia in the subgingival plaque of Type 2 diabetes mellitus patients with moderate periodontitis compared to non-diabetic patients with moderate periodontitis. Our binary logistic regression analysis indicated that the risk of GDM increased 1.208-fold with an increase in T. forsythia levels (P = 0.002). T. forsythia count was found to be associated with both increased PISA value and GDM. Capillary density in gingival tissues of patients with GDM was found to be higher compared to healthy non-pregnant controls, suggesting a cumulative effect of pregnancy and GDM in gingival microcirculation [41]. In this aspect, the additional systemic burden of GDM and increased sulcular fluid [41, 49], as seen through the higher PISA and BoP in the GDM group of our study, may increase the influx of serum proteins and hemoglobin into the pocket. This influx may expand local heme/iron availability (including albumin-associated pools and hemoglobin-derived heme) creating a niche which may favor a heme-scavenging pathobiont such as T. forsythia [50].
T. denticola, a highly mobile, invasive, gram-negative anaerobe and red complex member, is frequently isolated from periodontitis sites and associated with pregnancy complications [16]. Although it was only detected in a limited group of participants in our study, T. denticola was observed in greater numbers in the non-GDM group. According to path analysis, its association with GI and PISA might indicate that T. denticola is associated with periodontal inflammation (P = 0.011 and P = 0.049 respectively).
The strengths of our study include the adequate sample size, use of species-specific qPCR, plaque collection from the deepest subgingival sites throughout the mouth, comprehensive periodontal assessments of six sites per tooth for all clinical parameters and the incorporation of PISA values as a quantification of the inflamed periodontal surface area. Our PISA results align with Chaparro et al. who also found significantly higher PISA values in GDM patients compared to healthy pregnant women [51]. While we identified significant associations, one limitation of our study is its cross-sectional design, which limits the ability to establish a causality. Additionally, our sample was not matched for age, which, since GDM individuals are inherently older, may be a potential confounder in the observed associations. Dietary habits were also not assessed in our study, which may limit the interpretation of potential drivers of the hyperglycemic state and its relationship with the oral microbial profiles.
In considering the generalizability of this study’s findings, it is important to note that participants were recruited from tertiary care hospitals in Istanbul; hence, the results may not fully represent the wider population of pregnant women in Turkey – especially those living in areas with limited access to healthcare. Additionally, due to the restrictive exclusion criteria, our population may not capture the full spectrum of medical and dental complexity potentially seen in everyday practice; however, this was necessary to reduce the confounding factors. Future studies in more diverse populations, incorporating dietary assessments are needed to confirm the broader applicability of these findings.
Conclusion
In individuals with GDM, increasing sex hormone levels combined with dysregulated glucose metabolism, may contribute to alterations in subgingival pathobiont composition. There may be an interplay between this environment and elevated T. forsythia levels in this subgingival area, whereas this environment may create a niche that disfavors P. intermedia. Consequently, GDM is associated with a distinct periodontal microbial profile and increased inflammatory burden.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this manuscript, the authors only used ChatGPT to improve its linguistic quality through inquiry of wording suggestions. After using this tool, the authors reviewed and edited the content as needed and take full responsibility for the publication’s content.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Author contributions
All authors have made substantial contributions to this study. Fatih Cömert: Investigation, Data curation, Methodology, Project administration, Formal analysis, Resources and Final Approval. Oya Kaya-Şimşek: Investigation, Writing – review & editing, Resources and Final Approval. Oya Demirci: Methodology, Investigation and Final Approval. Nursen Topçuoğlu: Methodology, Investigation, Formal analysis, Writing – review & editing and Final Approval. Funda Yalçın: Conceptualization, Supervision, Writing – review & editing, Project administration, Validation and Final Approval. Silvi Domnori: Data curation, Formal analysis, Visualization, Writing – original draft, Writing – review & editing, Software and Final Approval. Ulku Baser: Conceptualization, Methodology, Supervision, Project administration, Writing – original draft, Writing – review & editing, Funding acquisition, and Final Approval.
Footnotes
* Hu-Friedy, Williams Probe 0.5 mm, REF PQW, USA.
† Omega Bio-tek, E.Z.N.A.® Bacterial DNA Kit, Cat. No. D3350-02, Norcross, GA, USA.
‡ Thermo Fisher Scientific, NanoDrop 2000 Spectrophotometer, Waltham, MA, USA.
§ Roche Diagnostics, LightCycler 480 Real-Time PCR System, Basel, Switzerland.
‖ IBM Corp., IBM SPSS Statistics for Windows, Version 23.0, Armonk, NY, USA.
¶ Heinrich Heine University, G*Power, Version 3.1.9.2, Düsseldorf, Germany.
References
[1] Hajishengallis G, Lamont RJ. Beyond the red complex and into more complexity: the polymicrobial synergy and dysbiosis (PSD) model of periodontal disease etiology. Mol Oral Microbiol. 2012;27(6):409–19. https://doi.org/10.1111/j.2041-1014.2012.00663.x
[2] Suarez LJ, Garzon H, Arboleda S, Rodriguez A. Oral dysbiosis and autoimmunity: from local periodontal responses to an imbalanced systemic immunity. A review. Front Immunol. 2020;11:591255. https://doi.org/10.3389/fimmu.2020.591255
[3] Bascones-Martinez A, Gonzalez-Febles J, Sanz-Esporrin J. Diabetes and periodontal disease. Review of the literature. Am J Dent. 2014;27(2):63–7.
[4] Negrato CA, Tarzia O, Jovanovic L, Chinellato LE. Periodontal disease and diabetes mellitus. J Appl Oral Sci. 2013;21(1):1–12. https://doi.org/10.1590/1678-7757201302106
[5] Sima C, Glogauer M. Diabetes mellitus and periodontal diseases. Curr Diab Rep. 2013;13(3):445–52. https://doi.org/10.1007/s11892-013-0367-y
[6] Buchanan TA, Xiang A, Kjos SL, Watanabe R. What is gestational diabetes? Diabetes Care. 2007;30 Suppl 2:S105–11.
[7] Barbour LA, McCurdy CE, Hernandez TL, Kirwan JP, Catalano PM, Friedman JE. Cellular mechanisms for insulin resistance in normal pregnancy and gestational diabetes. Diabetes Care. 2007;30 Suppl 2:S112–9. https://doi.org/10.2337/dc07-s202
[8] Best LG, Saxena R, Anderson CM, Barnes MR, Hakonarson H, Falcon G, et al. Two variants of the C-reactive protein gene are associated with risk of pre-eclampsia in an American Indian population. PLoS One. 2013;8(8):e71231.
[9] Wang H, Li N, Chivese T, Werfalli M, Sun H, Yuen L, et al. IDF diabetes atlas: estimation of global and regional gestational diabetes mellitus prevalence for 2021 by International Association of Diabetes in Pregnancy Study Group’s Criteria. Diabetes Res Clin Pract. 2022;183:109050. https://doi.org/10.1016/j.diabres.2021.109050
[10] Zachariasen RD. Ovarian hormones and oral health: pregnancy gingivitis. Compendium. 1989;10(9):508–12.
[11] Mariotti A. Sex steroid hormones and cell dynamics in the periodontium. Crit Rev Oral Biol Med. 1994;5(1):27–53. https://doi.org/10.1177/10454411940050010201
[12] Loe H, Silness J. Periodontal disease in pregnancy. I. Prevalence and severity. Acta Odontol Scand. 1963;21:533–51. https://doi.org/10.3109/00016356309011240
[13] Miyazaki H, Yamashita Y, Shirahama R, Goto-Kimura K, Shimada N, Sogame A, et al. Periodontal condition of pregnant women assessed by CPITN. J Clin Periodontol. 1991;18(10):751–4. https://doi.org/10.1111/j.1600-051X.1991.tb00067.x
[14] Carrillo-de-Albornoz A, Figuero E, Herrera D, Cuesta P, Bascones-Martinez A. Gingival changes during pregnancy: III. Impact of clinical, microbiological, immunological and socio-demographic factors on gingival inflammation. J Clin Periodontol. 2012;39(3):272–83. https://doi.org/10.1111/j.1600-051X.2011.01800.x
[15] Offenbacher S, Jared HL, O’Reilly PG, Wells SR, Salvi GE, Lawrence HP, et al. Potential pathogenic mechanisms of periodontitis associated pregnancy complications. Ann Periodontol. 1998;3(1):233–50. https://doi.org/10.1902/annals.1998.3.1.233
[16] Cassini MA, Pilloni A, Condo SG, Vitali LA, Pasquantonio G, Cerroni L. Periodontal bacteria in the genital tract: are they related to adverse pregnancy outcome? Int J Immunopathol Pharmacol. 2013;26(4):931–9. https://doi.org/10.1177/039463201302600411
[17] Ercan E, Eratalay K, Deren O, Gur D, Ozyuncu O, Altun B, et al. Evaluation of periodontal pathogens in amniotic fluid and the role of periodontal disease in pre-term birth and low birth weight. Acta Odontol Scand. 2013;71(3–4):553–9. https://doi.org/10.3109/00016357.2012.697576
[18] Kornman KS, Loesche WJ. Effects of estradiol and progesterone on Bacteroides melaninogenicus and Bacteroides gingivalis. Infect Immun. 1982;35(1):256–63. https://doi.org/10.1128/iai.35.1.256-263.1982
[19] Gursoy M, Haraldsson G, Hyvonen M, Sorsa T, Pajukanta R, Kononen E. Does the frequency of Prevotella intermedia increase during pregnancy? Oral Microbiol Immunol. 2009;24(4):299–303.
[20] Borgo PV, Rodrigues VA, Feitosa AC, Xavier KC, Avila-Campos MJ. Association between periodontal condition and subgingival microbiota in women during pregnancy: a longitudinal study. J Appl Oral Sci. 2014;22(6):528–33. https://doi.org/10.1590/1678-775720140164
[21] Jelihovschi I, Dorneanu O, Badescu A, Serban R, Ursu RG, Martu IA, et al. Increased detection rate of Prevotella Intermedia among pregnant periodontitis patients in Romania. Biomed J Sci Tech Res. 2018;2(10.26717). https://doi.org/10.26717/BJSTR.2017.01.000646
[22] Raber-Durlacher JE, Leene W, Palmer-Bouva CC, Raber J, Abraham-Inpijn L. Experimental gingivitis during pregnancy and post-partum: immunohistochemical aspects. J Periodontol. 1993;64(3):211–8. https://doi.org/10.1902/jop.1993.64.3.211
[23] Sharma A. Virulence mechanisms of Tannerella forsythia. Periodontol 2000. 2010;54(1):106–16. https://doi.org/10.1111/j.1600-0757.2009.00332.x
[24] Wang Y, Cupul-Uicab LA, Rogan WJ, Eggesbo M, Travlos G, Wilson R, et al. Recreational exercise before and during pregnancy in relation to plasma C-reactive protein concentrations in pregnant women. J Phys Act Health. 2015;12(6):770–5. https://doi.org/10.1123/jpah.2013-0390
[25] Fardini Y, Chung P, Dumm R, Joshi N, Han YW. Transmission of diverse oral bacteria to murine placenta: evidence for the oral microbiome as a potential source of intrauterine infection. Infect Immun. 2010;78(4):1789–96. https://doi.org/10.1128/IAI.01395-09
[26] Tellapragada C, Eshwara VK, Acharya S, Bhat P, Kamath A, Vishwanath S, et al. Prevalence of clinical periodontitis and putative periodontal pathogens among south indian pregnant women. Int J Microbiol. 2014;2014:420149. https://doi.org/10.1155/2014/420149
[27] Carta G, Persia G, Falciglia K, Iovenitti P. Periodontal disease and poor obstetrical outcome. Clin Exp Obstet Gynecol. 2004;31(1):47–9.
[28] Garcia-Martos JM, Valverde-Bolivar FJ, Campillo-Lopez MT, Delgado-Rodriguez M. Association between periodontal disease and gestational diabetes: systematic review and meta-analysis. Prim Care Diabetes. 2025;19(1):1–6. https://doi.org/10.1016/j.pcd.2024.11.003
[29] von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, et al. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Lancet. 2007;370(9596):1453–7. https://doi.org/10.1016/S0140-6736(07)61602-X
[30] O’Leary TJ, Drake RB, Naylor JE. The plaque control record. J Periodontol. 1972;43(1):38. https://doi.org/10.1902/jop.1972.43.1.38
[31] Ainamo J, Bay I. Problems and proposals for recording gingivitis and plaque. Int Dent J. 1975;25(4):229–35.
[32] Nesse W, Abbas F, van der Ploeg I, Spijkervet FK, Dijkstra PU, Vissink A. Periodontal inflamed surface area: quantifying inflammatory burden. J Clin Periodontol. 2008;35(8):668–73. https://doi.org/10.1111/j.1600-051X.2008.01249.x
[33] Tonetti MS, Greenwell H, Kornman KS. Staging and grading of periodontitis: framework and proposal of a new classification and case definition. J Periodontol. 2018;89 Suppl 1:S159–72.
[34] Gogeneni H, Buduneli N, Ceyhan-Ozturk B, Gumus P, Akcali A, Zeller I, et al. Increased infection with key periodontal pathogens during gestational diabetes mellitus. J Clin Periodontol. 2015;42(6):506–12. https://doi.org/10.1111/jcpe.12418
[35] Ganiger K, Sridharan S, Rahul A, Satyanarayana A. Quantitative analysis of key periodontopathic bacteria in gestational diabetic and non-diabetic women. J Diabetes Metab Disord. 2019;18(2):363–9. https://doi.org/10.1007/s40200-019-00420-3
[36] Figuero E, Carrillo-de-Albornoz A, Herrera D, Bascones-Martinez A. Gingival changes during pregnancy: I. Influence of hormonal variations on clinical and immunological parameters. J Clin Periodontol. 2010;37(3):220–9. https://doi.org/10.1111/j.1600-051X.2009.01516.x
[37] Carrillo-de-Albornoz A, Figuero E, Herrera D, Bascones-Martinez A. Gingival changes during pregnancy: II. Influence of hormonal variations on the subgingival biofilm. J Clin Periodontol. 2010;37(3):230–40. https://doi.org/10.1111/j.1600-051X.2009.01514.x
[38] Li Y, Ren X, He L, Li J, Zhang S, Chen W. Maternal age and the risk of gestational diabetes mellitus: a systematic review and meta-analysis of over 120 million participants. Diabetes Res Clin Pract. 2020;162:108044.
[39] Lappe V, Greiner GG, Linnenkamp U, Viehmann A, Adamczewski H, Kaltheuner M, et al. Gestational diabetes in Germany-prevalence, trend during the past decade and utilization of follow-up care: an observational study. Sci Rep. 2023;13(1):16157. https://doi.org/10.1038/s41598-023-43382-6
[40] World Health Organization. A healthy lifestyle – WHO recommendations. 2010. Available from: https://www.who.int/europe/news-room/fact-sheets/item/a-healthy-lifestyle---who-recommendations
[41] Yilmaz D, Altas A. Evaluation of gingival microcirculation in patients with gestational diabetes mellitus. Microvasc Res. 2021;138:104222. https://doi.org/10.1016/j.mvr.2021.104222
[42] Hajishengallis G. The inflammophilic character of the periodontitis-associated microbiota. Mol Oral Microbiol. 2014;29(6):248–57. https://doi.org/10.1111/omi.12065
[43] Dasanayake AP, Chhun N, Tanner AC, Craig RG, Lee MJ, Moore AF, et al. Periodontal pathogens and gestational diabetes mellitus. J Dent Res. 2008;87(4):328–33. https://doi.org/10.1177/154405910808700421
[44] Keshavarz M, Cheung NW, Babaee GR, Moghadam HK, Ajami ME, Shariati M. Gestational diabetes in Iran: incidence, risk factors and pregnancy outcomes. Diabetes Res Clin Pract. 2005;69(3):279–86.
[45] Xiong X, Elkind-Hirsch KE, Vastardis S, Delarosa RL, Pridjian G, Buekens P. Periodontal disease is associated with gestational diabetes mellitus: a case-control study. J Periodontol. 2009;80(11):1742–9. https://doi.org/10.1902/jop.2009.090250
[46] Jensen J, Liljemark W, Bloomquist C. The effect of female sex hormones on subgingival plaque. J Periodontol. 1981;52(10):599–602. https://doi.org/10.1902/jop.1981.52.10.599
[47] Muramatsu Y, Takaesu Y. Oral health status related to subgingival bacterial flora and sex hormones in saliva during pregnancy. Bull Tokyo Dent Coll. 1994;35(3):139–51.
[48] Akherati M, Shafaei E, Salehiniya H, Abbaszadeh H. Comparison of the frequency of periodontal pathogenic species of diabetics and non-diabetics and its relation to periodontitis severity, glycemic control and body mass index. Clin Exp Dent Res. 2021;7(6):1080–8. https://doi.org/10.1002/cre2.453
[49] Sakallioglu EE, Lutfioglu M, Sakallioglu U, Diraman E, Keskiner I. Fluid dynamics of gingiva in diabetic and systemically healthy periodontitis patients. Arch Oral Biol. 2008;53(7):646–51. https://doi.org/10.1016/j.archoralbio.2007.12.013
[50] Bielecki M, Antonyuk S, Strange RW, Smalley JW, Mackiewicz P, Smiga M, et al. Tannerella forsythia Tfo belongs to Porphyromonas gingivalis HmuY-like family of proteins but differs in heme-binding properties. Biosci Rep. 2018;38(5):BSR20181325 https://doi.org/10.1042/BSR20181325
[51] Chaparro A, Zuniga E, Varas-Godoy M, Albers D, Ramirez V, Hernandez M, et al. Periodontitis and placental growth factor in oral fluids are early pregnancy predictors of gestational diabetes mellitus. J Periodontol. 2018;89(9):1052–60. https://doi.org/10.1002/JPER.17-0497