SHORT REPORT
Impact of age and comorbidities on real-world outcomes in advanced breast cancer patients treated with palbociclib in first line: a nation-wide Danish retrospective study
Alan Celik  a,b, Laurits Sebastian Dahla, Rasmus Garlya, Vesna Glavicicc, Maja Bendtsen Sharmad, Sophie Yammenie, Humma Khanf, Daniel Sloth Haubergf, Hanne Schultz Kapelf, Ann Knoop
a,b, Laurits Sebastian Dahla, Rasmus Garlya, Vesna Glavicicc, Maja Bendtsen Sharmad, Sophie Yammenie, Humma Khanf, Daniel Sloth Haubergf, Hanne Schultz Kapelf, Ann Knoop  g and Tobias Berg
g and Tobias Berg  a,g
a,g
aDanish Breast Cancer Group, Rigshospitalet, Copenhagen University Hospital, Copenhagen, Denmark; bZealand University Hospital, Emergency Department, Nykoebing F, Denmark; cDepartment of Oncology, Zealand University Hospital, Naestved, Denmark; dDepartment of Oncology, Aarhus University Hospital, Aarhus, Denmark; eDepartment of Oncology, Aalborg University Hospital, Aalborg, Denmark; fPfizer ApS, Ballerup, Denmark; gDepartment of Clinical Oncology, Rigshospitalet, Copenhagen University Hospital, Copenhagen, Denmark
Abstract
Background and purpose: Palbociclib, a cyclin-dependent kinase 4/6 (CDK4/6) inhibitor, combined with aromatase inhibitors (AI), can be used in first-line treatment for estrogen receptor (ER) positive, human epidermal growth factor receptor 2 (HER2) normal advanced breast cancer (ABC). This study aims to assess the impact of age and comorbidities on the progression-free survival (PFS) and overall survival (OS) of patients treated with palbociclib and AI.
Materials and methods: This nationwide, retrospective cohort study included 604 women with ER positive, HER2 normal ABC treated with palbociclib and an AI between 2017 and 2021. Data were obtained from the Danish Breast Cancer Group database. Survival outcomes were analyzed based on age, Charlson Comorbidity Index (CCI), number of comorbidities, and comorbidity type. PFS and OS were estimated using the Kaplan–Meier method.
Results: Median PFS for all patients was 30.6 months (95% confidence interval [CI], 27.4–34.2), and median OS was 55.6 months (95% CI, 51.8–58.9). Patients aged 65–75 years had significantly longer PFS and OS (p = 0.031 and p = 0.012) than patients aged under 65 and over 75 years. Visceral metastases were associated with shorter PFS and OS (p = 0.005). Comorbidity burden, including CCI score and comorbidity type, did not significantly affect survival outcomes.
Interpretation: In our data set, univariate analyses suggest age and visceral metastases to be potential factors influencing outcomes in patients with ABC treated with palbociclib plus an AI. Comorbidities, did not significantly impact survival, suggesting that palbociclib is well-tolerated in patients with varying health profiles.
ClinicalTrials.gov ID: NCT06307457.
KEYWORDS: Advanced breast cancer; Palbociclib; CDK4/6 inhibitor; survival; age; comorbidity
Citation: ACTA ONCOLOGICA 2025, VOL. 64, 778–783. https://doi.org/10.2340/1651-226X.2025.43226.
Copyright: © 2025 The Author(s). Published by MJS Publishing on behalf of Acta Oncologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/).
Received: 21 February 2025; Accepted: 11 June 2025; Published: 11 June 2025
CONTACT: Alan Celik alan.celik.01@regionh.dk Danish Breast Cancer Group, Rigshospitalet, Copenhagen University Hospital. Blegdamsvej 9, DK-2100, Copenhagen, Denmark
Supplemental data for this article can be accessed online at https://doi.org/10.2340/1651-226X.2025.43226
Competing interests and funding: AC: Travel: Astra Zeneca.
LSD: None.
RG: None.
VG: Travel: Pfizer, MSD, Daiichi Sankyo.
MBS: Travel: Gilead Sciences, MSD.
SY: Travel: Pfizer, MSD, Advisory board: MSD, Daiichi Sankyo, Roche
HK: Is current employee at Pfizer and own shares in Pfizer Inc.
DSH: Is current employee at Pfizer and own shares in Pfizer Inc.
HSK: Is a paid contractor to Pfizer
AK: Institutional grants from: Pfizer, Astra Zeneca, Novartis and Daiichi Sankyo. Advisory board: Astra Zeneca, Novartis, Gilead Sciences Denmark, Eli Lilly, MSD, Seagen Denmark Aps and Daiichi Sankyo. Travel: Daiichi Sankyo.
TB: Institutional grants from: Pfizer, Astra Zeneca, Novartis, Samsung Bioepis, Seattle Genetics, Merck, Eli Lilly and Daiichi Sankyo. Advisory board: Novartis. Travel: Daiichi Sankyo.
This study was conducted as a collaboration between Danish Breast Cancer Group and Pfizer. This study was sponsored by Pfizer.
Introduction
The treatment landscape for advanced breast cancer (ABC) has drastically evolved with the arrival of targeted therapies, offering new hope for improved outcomes. Among these, selective cyclin-dependent kinase 4/6 (CDK4/6) inhibitors combined with an aromatase inhibitor (AI) have emerged as a first-line standard of care for estrogen receptor (ER) positive, human epidermal growth factor receptor 2 (HER2) normal ABC. This treatment has shown substantial efficacy in clinical trials, markedly extending progression-free survival (PFS) and overall survival (OS) [1–6]. However, the real-world effectiveness of this regimen can be influenced by various patient-specific factors, notably age and the presence of comorbidities. Older patients or patients with high comorbidity burden are often not included in trials given strict inclusion/exclusion criteria.
Understanding the interplay between patient characteristics and treatment outcomes is crucial for optimizing therapy and personalizing care in ABC. Ageing organ function can impact pharmacokinetics, treatment tolerance, and patient outcomes. Similarly, comorbidities, which are more common in the aging population, may complicate cancer treatment due to potential drug interactions and alteration of drug effectiveness. Evaluating how these factors affect the effectiveness of palbociclib in combination with an AI is essential to refining treatment protocols and ensuring reasonable care across diverse patient populations.
Several real-world studies have evaluated palbociclib, both with and without an AI, in older patients and those with comorbidities, with only two studies formally reviewing impact of comorbidities on outcomes [7–12]. However, such data have not yet been explored in a Danish setting. This study aims to explore the impact of age and comorbidities on treatment outcomes of first-line therapy with the CDK4/6 inhibitor palbociclib in combination with an AI in ER-positive, HER2-normal ABC. This study builds on a previously published cohort, extending follow-up and focusing exclusively on first-line treatment with palbociclib combined with an AI [13].
Material and methods
Study design
This is a nationwide, retrospective cohort study involving all departments of oncology in Denmark.
Patient selection
All known women aged 18 years or above diagnosed with primary (de novo) or recurrent ABC were included. All patients included had ER positive, HER2 normal disease and initiated first-line treatment with palbociclib in combination with an AI between January 1st, 2017, and December 31st, 2021. ER and HER2 status were based on metastatic tissue or – if not available – on primary tumor tissue.
Data source
The Danish Breast Cancer Group (DBCG) database was utilized in this study to gather information on date of primary diagnosis, date of ABC diagnosis, treatment ((neo)adjuvant and for advanced disease), date of progression (based on imaging and/or clinical examination), reason for treatment discontinuation and location of metastases. Information regarding comorbidities was gathered from the Danish National Patient Registry. Information concerning vital status was obtained from the Danish Civil Registration System.
Study endpoints
Primary endpoints were PFS and OS for all patients.
Secondary endpoints were PFS and OS stratified by age (< 65, 65–75 and ≥ 75 years), comorbidities (severity, number, and type), type of metastases (visceral and bone-only disease), disease presentation (de novo metastatic or recurrent), and endocrine resistance. De novo metastatic disease was defined as distant metastases at diagnosis or within 90 days of (neo) adjuvant therapy initiation. Endocrine resistance was defined as recurrent advanced disease within 12 months of completing or during adjuvant endocrine therapy (ET); endocrine sensitivity was defined as recurrent advanced disease after 12 months of completing adjuvant ET; recurrent patients having not received adjuvant ET and patients with de novo ABC were also counted as sensitive.
Measures
Comorbidity-subgroup analyses were split into three: severity, number of comorbidities and type at ABC diagnosis. The Charlson Comorbidity Index (CCI) was used to score severity including diagnoses made within the 10 years preceding the ABC diagnosis [14]. Patients were subdivided into four groups based on their CCI-score (0, 1, 2, and 3+). Patients were further divided into three comorbidity groups based on number of comorbidities (0, 1, and 2+ comorbidities). Splitting the subgroup analysis according to number of comorbidities per patient therefore puts equal weight on all types of comorbidities. Patients were also subdivided based on type of comorbidity into three main groups: cardiac, vascular, and metabolic diseases, following the diseases used in the CCI.
Statistical analysis
PFS was defined as the time from date of ABC diagnosis to either progression or death of any cause, whichever occurred first. OS was defined as the time from the date of ABC diagnosis to death from any cause. Patients were followed up until February 1st, 2024, and were censored at this point in time, if progression-free and/or alive. Estimates were calculated using the Kaplan–Meier method. Differences in both, PFS and OS across subgroups, were assessed using the Log-Rank method (univariate analysis). As this was a descriptive, exploratory study, we focused on univariate analyses. This approach was chosen to allow a clear and direct interpretation of real-world patterns without the added complexity or assumptions of multivariable modeling. Follow-up duration was calculated using the reverse Kaplan–Meier method. PFS, OS and follow-up duration were reported as medians with confidence intervals (CIs).
Results
Study population
During the study period, 604 patients received palbociclib in combination with an AI as first-line therapy across all departments of oncology in Denmark (Figure 1). Patient characteristics are presented in Table 1.
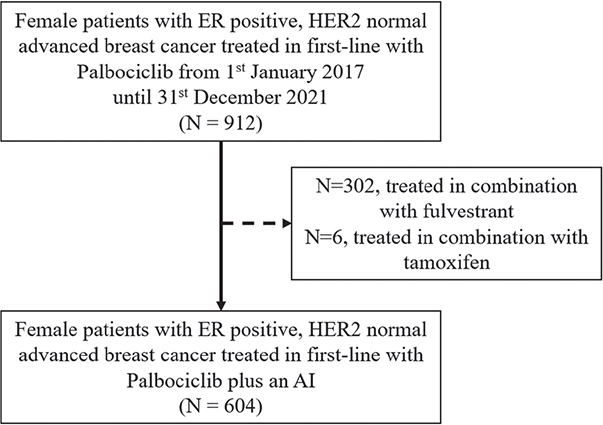
Figure 1. Flowchart of patient inclusion.
ER: estrogen receptor; HER2: human epidermal growth factor receptor 2; AI: aromatase inhibitor.
Median age was 68.9 years at baseline. A total of 66% (n = 400) of the patients had a CCI of 0, 17% (n = 101) a CCI of 1, 7.5% (n = 45) a CCI of 2, and 9.6% (n = 58) a CCI of 3 or more. In terms of the number of comorbidities, 147 patients (24%) had one comorbidity and 57 (9.4%) had two or more comorbidities at baseline. Both vascular and metabolic disease were present in 45 patients (7.5%) and 23 patients (3.8%) presented with cardiac disease. Most patients had visceral metastases, accounting for 321 patients (53%), while 145 (24%) had bone-only metastases. The remaining patients (23%) presented with metastases to distant lymph nodes, skin, muscle, or the contralateral breast, with or without concurrent bone metastases. Endocrine resistance was observed in 67 patients (11%), and 537 (89%) were classified as endocrine sensitive. Additionally, 198 patients (33%) presented with de novo metastatic disease.
Outcomes
For the overall 604 patients, median PFS was 30.6 months (95% CI, 27.4–34.2), and median OS was 55.6 months (95% CI, 51.8–58.9). A total of 322 patients died during the study period, and 430 patients had an event in form of progression or death during the study period. Kaplan–Meier graphs for PFS and OS are included in Supplementary Figure 1. Median follow-up for PFS and OS were 67.4 months (95% CI, 62.9–69.9) and 66.9 months (95% CI, 63.4–69.6), respectively.
Based on univariate analysis, age-groups showed a significant difference in terms of PFS and OS, with patients aged 65–75 years having a the longest median PFS and OS (p = 0.031 and p = 0.012). Patients with visceral metastases had a shorter median PFS and OS compared to patients without visceral involvement (p = 0.005 and p = 0.005). Endocrine-sensitive patients had a longer median PFS and OS compared to endocrine-resistant patients (p = 0.047 and p = 0.041). The median PFS in patients with de novo metastatic disease was 32.0 months (95% CI: 25.5–43.3) and median OS was 58.9 months (95% CI: 52.6–70.3).
In this univariate analysis, CCI did not significantly affect outcomes in terms of PFS and OS when stratified based on score (p = 0.061 and p = 0.080). Similarly, the number of comorbidities did not significantly affect PFS or OS (p = 0.2 and p = 0.093). Specific comorbidity subgroups, including cardiac, vascular, and metabolic diseases, did not show significant differences in outcomes.
All outcomes are summarized in Table 2.
| Characteristic | N | Median (95% CI) progression free survival | p1 | Median (95% CI) overall survival | p1 |
| All patients | 604 | 30.6 (27.4, 34.2) | 55.6 (51.8, 58.9) | ||
| Age group | 0.031 | 0.012 | |||
| Under 65 years | 212 | 25.8 (22.2, 30.7) | 52.6 (48.6, 59.2) | ||
| 65–75 years | 243 | 35.7 (30.4, 42.2) | 60.8 (55.6, 78.3) | ||
| Over 75 years | 149 | 29.8 (23.6, 37.2) | 50.1 (41.4, 58.6) | ||
| Visceral metastases | 0.005 | 0.005 | |||
| Non-Visceral | 283 | 34.6 (31.1, 39.8) | 59.2 (56.6, 69.8) | ||
| Visceral | 321 | 24.2 (19.8, 30.3) | 50.6 (44.0, 55.7) | ||
| Bone-only | 0.14 | 0.2 | |||
| Bone-only | 144 | 34.6 (27.7, 42.6) | 58.3 (51.8, NA) | ||
| Non-bone-only | 460 | 29.0 (25.4, 33.1) | 54.2 (50.1, 58.6) | ||
| Endocrine Status | 0.047 | 0.041 | |||
| Endocrine resistant | 67 | 21.7 (13.8, 37.2) | 43.5 (32.2, 59.2) | ||
| Endocrine sensitive | 537 | 31.8 (28.5, 34.6) | 56.6 (52.6, 60.8) | ||
| De novo metastatic | 198 | 32.0 (25.5, 43.3) | 58.9 (52.6, 70.3) | ||
| Charlson Comorbidity Index | 0.061 | 0.080 | |||
| 0 | 400 | 32.8 (28.3, 36.7) | 57.6 (53.9, 66.7) | ||
| 1 | 101 | 32.0 (26.0, 45.8) | 50.1 (43.6, 58.9) | ||
| 2 | 45 | 29.7 (17.1, 38.3) | 53.1 (43.3, NA) | ||
| 3+ | 58 | 21.5 (18.0, 33.2) | 45.2 (37.7, 58.3) | ||
| Number of comorbidities | 0.2 | 0.093 | |||
| 0 | 400 | 32.8 (28.3, 36.7) | 57.6 (53.9, 66.7) | ||
| 1 | 147 | 27.9 (23.8, 37.9) | 50.1 (44.5, 58.0) | ||
| 2+ | 57 | 23.6 (19.3, 35.6) | 52.7 (41.2, NA) | ||
| Type of comorbidity | |||||
| Cardiac | > 0.9 | 0.7 | |||
| No | 581 | 30.7 (27.3, 34.3) | 55.6 (51.8, 58.9) | ||
| Yes | 23 | 27.9 (22.2, NA) | 61.2 (45.6, NA) | ||
| Vascular | 0.9 | 0.3 | |||
| No | 559 | 30.3 (27.0, 34.2) | 56.1 (52.4, 59.2) | ||
| Yes | 45 | 32.3 (22.9, 44.9) | 47.4 (42.9, NA) | ||
| Metabolic | 0.4 | 0.7 | |||
| No | 559 | 31.1 (27.7, 34.6) | 56.1 (52.4, 59.2) | ||
| Yes | 45 | 25.5 (17.7, 36.5) | 50.1 (39.8, NA) | ||
| 1 Log-Rank (univariate analysis). | |||||
Discussion
Our study aimed to explore the impact of age and comorbidities on the treatment outcomes of first-line therapy with palbociclib and an AI in ER positive, HER2 normal ABC.
In our data set, univariate analyses suggest age as a significant factor influencing both PFS and OS. The superior outcomes in the 65–75 age group could be due to several factors, including differences in tumor biology and burden, treatment tolerance, and adherence to therapy. While older patients (above 75 years) showed a PFS similar to younger patients (29.8 months, 95% CI, 23.6–37.2), their OS was notably not surprisingly shorter (50.1 months, 95% CI, 41.4–58.6) [15]. In a recent study, in patients aged 75 years or older the point estimate of PFS was lower compared to the one found in our study (20.5 months (95% CI, 17.5–27.3)) with a median OS of 47.8 months (95% CI, 40.7–NA) [16].
The presence of visceral metastases was associated with significantly worse outcomes (p = 0.005 for both PFS and OS). These findings align with prior findings suggesting that visceral involvement is a marker of more aggressive disease [13]. Furthermore, we found that patients with endocrine sensitive disease experienced significantly improved outcomes compared to those with resistant disease (p = 0.047 and p = 0.041, for PFS and OS respectively).
Interestingly, the severity of comorbidities, assessed through the CCI, and the number of comorbidities did not statistically significantly impact PFS or OS, but did however show a numerical trend favoring a CCI score of 0. Similarly, the number of comorbidities did not significantly affect survival outcomes (p = 0.093). Moreover, specific comorbid conditions, such as cardiac, vascular, and metabolic diseases, were not associated with significant differences in survival outcomes (p > 0.05 for all comparisons); however, sample sizes were small in these subgroups.
The lack of an impact of comorbidities on PFS and OS in our dataset might suggests that palbociclib in combination with an AI is a viable therapeutic option across a broad range of patient profiles, including those with multiple comorbidities or severe comorbidity burden. This is crucial in clinical practice, especially for older patients, who are more likely to present with comorbid conditions. ESMO-MCBS scores differ among the available three CDK4/6 inhibitors, with ribociclib receiving the most favorable ratings, though it is important to acknowledge that no specific CDK 4/6 inhibitor is recommended in first-line in the ESMO/ESO-guidelines [6, 17]. Additionally, ribociclib has been associated with a higher incidence of clinically significant (Class C/D) drug–drug interactions (especially regarding QT interval prolongation) compared to palbociclib, a concern particularly pertinent in older patients who often manage multiple comorbidities and medications [18]. No head-to-head comparisons between the three approved CDK 4/6 inhibitors have been made yet concerning outcomes. As this study focused on the use of palbociclib, we are unable to make any comparisons or conclusions about other available CDK4/6 inhibitors with respect to the outcomes stratified by comorbidities.
This study has several strengths and limitations. By utilizing the DBCG national database, we ensured that all known women in Denmark treated with palbociclib plus AI during the study period were included, minimizing geographical and socioeconomic bias. No information regarding performance status, quality of life during treatment, and data concerning the safety of treatment was available. In addition, due to the retrospective nature of our study, missingness cannot be excluded. The small sample sizes of some subgroups also presented a limitation. Finally, a key limitation of the study is the lack of multivariable analysis, which restricts our ability to assess potential confounding factors. However, given the descriptive aim of the study, we prioritized an unadjusted approach to capture clinically observable trends in a real-world setting. We recognize that future studies with larger and more uniform cohorts may benefit from multivariable analyses to further clarify these relationships. Accordingly, further assessments, including prospective validation of our findings are warranted.
Conclusion
In conclusion, our findings suggest that palbociclib in combination with an AI is an effective treatment across different patient subgroups in ABC, though certain groups may derive a greater benefit than others. Our real-world univariate analyses showed that patients aged 65–75 years, patients without visceral metastases, and endocrine-sensitive patients had the greatest survival benefit. Comorbidities did not significantly impact outcomes, indicating that palbociclib-based therapy may be well-tolerated across diverse health profiles. Imbalances between prognostic factors, such as age and comorbidity groups, may have influenced this analysis, highlighting the need for further investigation, including prospective studies.
Data availability statement
All data are stored in the DBCG database. The dataset can be made available to qualified researchers through application to the Danish Breast Cancer Group. Please contact dbcg.rigshospitalet@regionh.dk.
Ethics declarations & trial registry information
The study was approved by the Danish Breast Cancer Group’s oncological committee. The study was also registered and approved by the Capital Regions research overview (P- 2022-386) and the Center for Health constituting signed consent (R-22036282).
Author contributions
AC, HK, DSH, HSK, AK, and TB helped design this study. AC, LSD, and RG conducted the data curation. AC, HK, DSH, HSK, AK, and TB planned the statistical analyses and AC performed them. AC drafted the original manuscript. All authors reviewed, edited, and approved the final manuscript.
References
[1] Sledge GWJ, Toi M, Neven P, Sohn J, Inoue K, Pivot X, et al. MONARCH 2: abemaciclib in combination with fulvestrant in women with HR+/HER2-advanced breast cancer who had progressed while receiving endocrine therapy. J Clin Oncol. 2017;35:2875–84. https://doi.org/10.1200/JCO.2017.73.7585
[2] Goetz MP, Toi M, Campone M, Sohn J, Paluch-Shimon S, Huober J, et al. MONARCH 3: abemaciclib as initial therapy for advanced breast cancer. J Clin Oncol. 2017;35:3638–46. https://doi.org/10.1200/JCO.2017.75.6155
[3] Cristofanilli M, Turner NC, Bondarenko I, Ro J, Im S-A, Masuda N, et al. Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2-negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, PHA. Lancet Oncol. 2016;17:425–39. https://doi.org/10.1016/S1470-2045(15)00613-0
[4] Finn RS, Martin M, Rugo HS, Jones S, Im S-A, Gelmon K, et al. Palbociclib and letrozole in advanced breast cancer. N Engl J Med. 2016;375:1925–36. https://doi.org/10.1056/NEJMoa1607303
[5] Hortobagyi GN, Stemmer SM, Burris HA, Yap YS, Sonke GS, Paluch-Shimon S, et al. Updated results from MONALEESA-2, a phase III trial of first-line ribociclib plus letrozole versus placebo plus letrozole in hormone receptor-positive, HER2-negative advanced breast cancer. Ann Oncol. 2018;29:1541–7. https://doi.org/10.1093/annonc/mdy155
[6] Slamon DJ, Neven P, Chia S, Fasching PA, De Laurentiis M, Im S-A, et al. Phase III randomized study of ribociclib and fulvestrant in hormone receptor-positive, human epidermal growth factor receptor 2-negative advanced breast cancer: MONALEESA-3. J Clin Oncol. 2018;36:2465–72. https://doi.org/10.1200/JCO.2018.78.9909
[7] El Badri S, Tahir B, Balachandran K, Bezecny P, Britton F, Davies M, et al. Palbociclib in combination with aromatase inhibitors in patients ≥ 75 years with oestrogen receptor-positive, human epidermal growth factor receptor 2 negative advanced breast cancer: a real-world multicentre UK study. Breast. 2021;60:199–205. https://doi.org/10.1016/j.breast.2021.10.010
[8] Goyal RK, Chen H, Abughosh SM, Holmes HM, Candrilli SD, Johnson ML. Overall survival associated with CDK4/6 inhibitors in patients with HR+/HER2- metastatic breast cancer in the United States: a SEER-Medicare population-based study. Cancer. 2023;129:1051–63. https://doi.org/10.1002/cncr.34675
[9] Carola E, Pulido M, Falandry C, Paillaud E, Caillet P, Tassy L, et al. First-line systemic treatment with palbociclib in women aged ≥70 years presenting with hormone receptor-positive advanced breast cancer: results from the PALOMAGE program. J Clin Oncol. 2023;41:1018. https://doi.org/10.1200/JCO.2023.41.16_suppl.1018
[10] Brain E, Pulido M, Paillaud E, Grosjean J, Mina W, Caillet P, et al. Abstract P1-18-04: feasibility of palbociclib in women aged 70 and older with resistant and/or pretreated advanced breast cancer in the PALOMAGE study. Cancer Res. 2022;82:P1-18-04. https://doi.org/10.1158/1538-7445.SABCS21-P1-18-04
[11] Caillet P, Pulido M, Brain E, Falandry C, Desmoulins I, Ghebriou D, et al. PALOMAGE, a French real-world cohort of elderly women beyond age 70 with advanced breast cancer receiving palbociclib: baseline characteristics and safety evaluation. J Clin Oncol. 2025;39:1012. https://doi.org/10.1200/JCO.2021.39.15_suppl.1012
[12] Rugo HS, Liu X, Li B, McRoy L, Layman RM, Brufsky A. Real-world comparative effectiveness of palbociclib plus letrozole versus letrozole in older patients with metastatic breast cancer. Breast. 2023;69:375–81. https://doi.org/10.1016/j.breast.2023.03.015
[13] Garly R, Berg T, Jensen M-B, Knoop A, Volmer L, Glavicic V, et al. A retrospective, non-interventional study of breast cancer patients diagnosed with ER+/HER2 negative, locally advanced or metastatic breast cancer treated with palbociclib in Denmark. Acta Oncol. 2023;62:290–7. https://doi.org/10.1080/0284186X.2023.2194030
[14] Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83. https://doi.org/10.1016/0021-9681(87)90171-8
[15] Brain E, Chen C, Simon S, Pasupuleti V, Pfitzer KV, Gelmon KA. Palbociclib in older patients with advanced/metastatic breast cancer: a systematic review. Target Oncol. 2024;19:303–20. https://doi.org/10.1007/s11523-024-01046-z
[16] Brufsky A, Liu X, Li B, McRoy L, Chen C, Layman RM, et al. Real-world treatment patterns and effectiveness of palbociclib plus an aromatase inhibitor in patients with metastatic breast cancer aged 75 years or older. Front Oncol. 2023;13:1237751. https://doi.org/10.3389/fonc.2023.1237751
[17] Cardoso F, Paluch-Shimon S, Schumacher-Wulf E, Matos L, Gelmon K, Aapro MS, et al. 6th and 7th International consensus guidelines for the management of advanced breast cancer (ABC guidelines 6 and 7). Breast. 2024;76:103756. https://doi.org/10.1016/j.breast.2024.103756
[18] Kapagan T, Bulut N, Erdem GU. Polypharmacy and drug-drug interactions in metastatic breast cancer patients receiving cyclin-dependent kinase (CDK) 4/6 inhibitors. J Oncol Pharm Pract. 2024;30:1403–10. https://doi.org/10.1177/10781552231218959