LETTER
Attitudes toward AI-generated risk prediction in patients with early breast cancer: an international multicenter survey
Frederik Voigt Carstensena  , Sofie A.M. Gernaatb
, Sofie A.M. Gernaatb  , Friederike Banningc
, Friederike Banningc  , Eva Batistad
, Eva Batistad  , Desiree van den Bongardf,g
, Desiree van den Bongardf,g  , Nadia Harbeckc,g
, Nadia Harbeckc,g  , Marle Hattinkb
, Marle Hattinkb  , Lorenzo Livih,i
, Lorenzo Livih,i  , Icro Meattinih,i
, Icro Meattinih,i  , Karin Meijere,f
, Karin Meijere,f  , Jens Petersena,j
, Jens Petersena,j  , Ivica Ratosak,l
, Ivica Ratosak,l  , Helena Verkooijenb
, Helena Verkooijenb  , Ivan Richter Vogeliusa
, Ivan Richter Vogeliusa  and Maja Vestmø Maraldoa
and Maja Vestmø Maraldoa 
aDepartment of Oncology, Copenhagen University Hospital, Rigshospitalet, Copenhagen, Denmark; bDivision of Imaging and Oncology, University Medical Center Utrecht, Utrecht University, Utrecht, The Netherlands; cBreast Center, Department of OB&GYN, LMU University Hospital, Munich, Germany; dBreast Unit, Champalimaud Foundation, Lisbon, Portugal; eDepartment of Radiation Oncology, Amsterdam UMC, Cancer Center Amsterdam, Amsterdam, Netherlands; fCancer Treatment and Quality of Life/Cancer Biology and Immunology, Amsterdam, Netherlands; gCCC Munich, LMU University Hospital, Munich, Germany; hDepartment of Experimental and Clinical Biomedical Sciences “M. Serio”, University of Florence, Florence, Italy; iRadiation Oncology Unit & Breast Unit, Azienda Ospedaliero-Universitaria Careggi, Florence, Italy; jDepartment of Computer Science, University of Copenhagen, Copenhagen, Denmark; kDivision of Radiotherapy, Institute of Oncology Ljubljana, Zaloska cesta 2, Ljubljana, Slovenia; lFaculty of Medicine, University of Ljubljana, Vrazov trg 2, Ljubljana, Slovenia
KEYWORDS: breast cancer; radiotherapy; artificial intelligence; risk prediction; chronic disease; cardiovascular disease; survey
Citation: ACTA ONCOLOGICA 2025, VOL. 64, 1125–1128. https://doi.org/10.2340/1651-226X.2025.44030.
Copyright: © 2025 The Author(s). Published by MJS Publishing on behalf of Acta Oncologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/).
Received: 31 May 2025; Accepted: 13 August 2025; Published: 26 August 2025
CONTACT: Frederik Voigt Carstensen frederik.voigt.carstensen@regionh.dk Solbakken 15, 2840 Holte, Denmark; Maja Vestmø Maraldo maja.vestmoe.maraldo@regionh.dk Tuxensvej 20, 2700 Brønshøj, Denmark
Competing interests and funding: The funding body had no role in the study design, data collection, analysis, or writing of the manuscript.
Introduction
An estimated 375.000 women in Europe are diagnosed with invasive breast cancer (BC) yearly [1]. Most patients (90%) have early BC and are eligible for curative treatment [2, 3]. Advances in BC care have increased survival, and currently 80% are alive 10 following diagnosis [4]. BC survivors are at increased risk of developing chronic diseases, such as cardiovascular disease (CVD), osteoporosis, interstitial lung disease, and weight gain [5, 6]. The elevated risks are partly due to side effects from chemotherapy, antibody-treatment, radiotherapy (RT), and antihormonal therapies [7, 8].
Most early BC patients receive postoperative RT as part of adjuvant treatment [3]. For RT planning, a computed tomography (CT)-scan is performed, which may harbor information on the patients’ health status but is normally used only for RT planning. The ARTILLERY [9] project aims to develop risk prediction models through trustworthy artificial intelligence (AI) models estimating the risk of common chronic diseases based on RT planning CT-scans of early BC patients. The chronic diseases include CVD, osteoporosis, chronic obstructive pulmonary disease (COPD), and unfavorable body composition. Early identification of early BC patients at high-risk of developing chronic diseases can result in early diagnosis and timely treatment, with a consequent increase in health, quality of life, and survival. In a 2021 study, Gal et al. [10] used a deep learning algorithm to quantify coronary artery calcium (CAC) from planning CT-scans in a cohort of nearly 16.000 BC patients, revealing that elevated CAC scores were significantly associated with increased CVD risk. However, to our knowledge, no prospective studies have used such additional information as the basis for a structured patient intervention. Also, patients’ attitudes toward secondary usage of the RT planning CT-scans have not previously been investigated. The aim of our study is to investigate if early BC patients referred for postoperative RT are interested in such an AI-generated risk prediction.
Patients/material and methods
The survey was designed as a cross-sectional study including European early BC patients planned for postoperative RT. This study aimed to recruit 40 consecutive participants from nine RT institutions in Denmark (n = 3), Netherlands (n = 2), Germany (n = 1), Slovenia (n = 1), Portugal (n = 1), and Italy (n = 1).
Patients were asked to answer the survey either prior to or directly following their planning CT-scan. Participation was voluntary, and responses had no impact on the patients’ scheduled treatment. Survey data were entered locally into a centralized REDCap (Research Electronic Data Capture) database. The requirement for ethical approval was determined independently by each participating institution in accordance with local regulations and guidelines.
The survey was developed by the ARTILLERY members of the study, in English, and translated locally. Danish BC representatives reviewed the content for clarity, prompting minor revisions.
The survey was administered on paper and consisted of two pages: one with study and ARTILLERY project information, and another with the survey items. The questionnaire included two demographic items (age and smoking status) and four items assessing interest in AI-generated risk predictions for CVD, osteoporosis, COPD, and unfavorable body composition. The response scale was self-constructed, and response options were ‘Yes’, ‘No’, and ‘Don’t know’.
Data analysis
Descriptive statistics and exploratory multivariate logistic regression were conducted using ‘R for Windows’ version 4.5.0 and ‘R Studio’ version 2024.12.1. The variables included in the multivariate logistic regression were age (continuous), smoking status (binary), country of origin (nominal categorical), and a non-positive attitude toward AI-generated risk predictions (binary).
Results
Data were collected from February 2024 to May 2025. A total of 349 survey responses were collected (Denmark n = 120, Netherlands n = 69, Germany n = 40, Slovenia n = 40, Portugal n = 40, and Italy n = 40). The only variable with missing data was age, which was missing in less than 1% of participants. For all participants, the median age was 59 (range: 28–94) years, and 14.3% (CI95% 10.8–18.4) were active smokers. The majority reported positive attitudes toward a risk prediction, with 87.4% (CI95% 83.4–90.7), 89.1% (CI95% 85.4–92.1), 87.3% (CI95% 83.4–90.7), and 85.1% (CI95% 80.9–88.7) being positive for CVD, osteoporosis, COPD, and unfavorable body composition, respectively. Figure 1 shows the attitudes toward AI-generated risk prediction of chronic diseases by country. Notably, a less positive attitude was observed in Italy, with 70.0% (CI95% 53.5–83.4) answering positively for CVD, 70.0% (CI95% 53.5–83.4) for osteoporosis, 70.0% (CI95% 53.5–83.4) for COPD, and 65.0% (CI95% 48.3–79.4) for unfavorable body composition. Median age for Italian participants was 64 (range: 34–94) years, and 30% (CI95% 16.6–46.6) were active smokers.
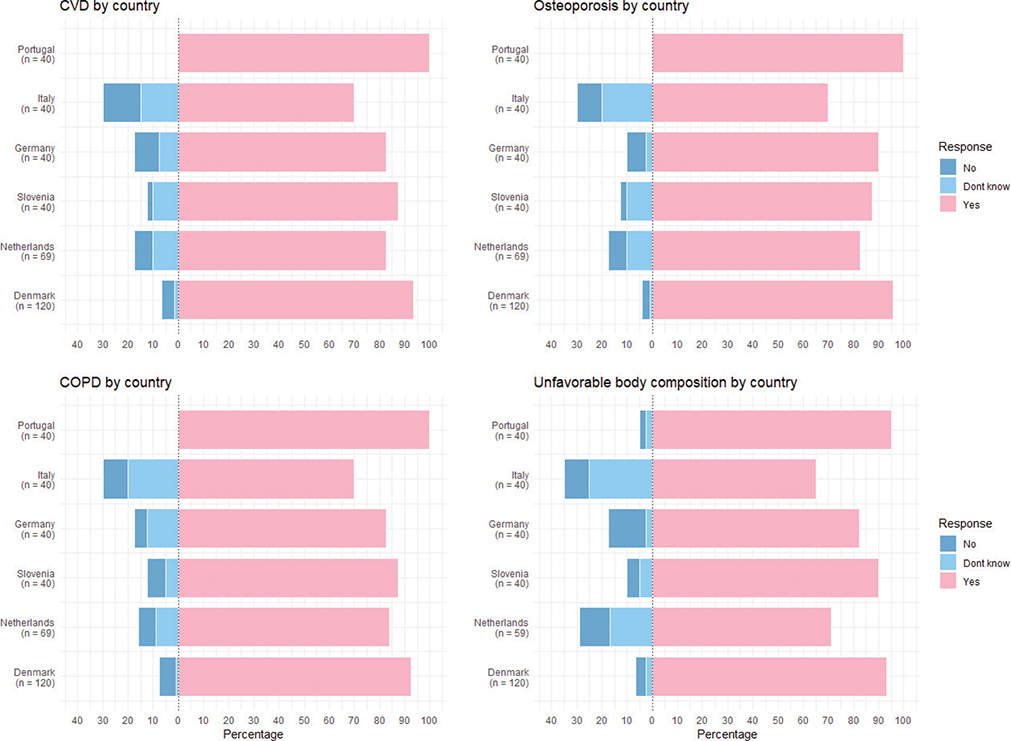
Figure 1. Attitudes toward AI-generated risk prediction of non-communicable diseases shown on country level in percentages.
Logistic regression was performed to assess associations between age, smoking status, and country, with non-positive attitudes toward AI-generated risk predictions. Older age was significantly associated with non-positive attitudes across all conditions: CVD (OR = 1.05; CI95% 1.02–1.08; p < 0.001), osteoporosis (OR = 1.04; CI95% 1.02–1.08; p < 0.01), COPD (OR = 1.06; CI95% 1.06–1.10; p < 0.001), and unfavorable body composition (OR = 1.03; CI95% 1.00–1.06; p < 0.05).
Smoking status was not significantly associated with non-positive attitudes for any of the outcomes.
A significant association was found between Italian nationality and non-positive attitudes toward AI risk predictions for all conditions: CVD (OR = 3.08; CI95% 1.32–6.88; p < 0.01), osteoporosis (OR = 3.95; CI95% 1.68–8.96; p < 0.01), COPD (OR = 3.03; CI95% 1.28–6.91; p < 0.01), and unfavorable body composition (OR = 3.38; CI95% 1.55–7.18; p < 0.01).
Discussion and conclusion
This European multicenter survey demonstrates a strong interest among early BC patients in receiving a predicted risk of developing common chronic diseases using AI. Across the cohort, over 85% of participants expressed a positive attitude, highlighting patients’ acceptance of integrating AI-systems into the standard oncology care pathway.
These findings are particularly relevant, given the high survival rates among early BC patients, which increase the patient awareness of the risks of developing treatment-induced chronic diseases over time. The RT planning CT-scans present an obvious tool for proactive health management, but these imaging datasets remain an underutilized resource.
Notably, a geographical variation was observed, with Italian participants showing comparatively lower interest in AI-based risk predictions. Additionally, Italian respondents had the highest median age and smoking prevalence. While reasons for this discrepancy remain speculative, cultural attitudes toward health technology and varying levels of digital health literacy may contribute. Also, we found that a higher age was associated with a lower interest in AI-generated risk predictions. Understanding factors like age and regional differences can help tailor communication and improve acceptance. These data are valuable for developing patient-centered strategies to implement AI-based risk tools effectively.
Our study was limited by the potential nonresponse bias as the patients who did not want to participate could be more skeptical toward the use of AI or risk predictions. Additionally, the hypothetical nature of the survey items is a limitation, as no information on the performance of the proposed risk prediction models was available to participants.
Overall, these findings support the acceptability of implementing AI-generated risk prediction in clinical oncology settings but highlight the need for patient-centered communication strategies and potential cultural adaptation. Through a prospective trial, ARTILLERY will provide insight into longitudinal outcomes following the implementation of AI risk prediction models, including their impact on early diagnosis of chronic diseases, patient satisfaction, and quality of life.
Acknowledgments
This work was supported by the European Union’s Horizon Europe under Grant No. 101080983.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Ethics declarations & trial registry information
According to the regulations of Capital Region of Denmark’s Department of Research Law, this study did not require ethical approval as it involved anonymous survey data, and the data were not presented to participants. Informed consent was implied through voluntary participation.
Author contributions
Frederik V. Carstensen, Maja V. Maraldo, Sofie A.M. Gernaat, and Helena Verkooijen conceptualized and designed the study.
Frederik V. Carstensen, Maja V. Maraldo, Jens Petersen, and Ivan R. Vogelius assisted in the study design and coordinated local approvals, participant recruitment, and data acquisition in Copenhagen, Herlev and Næstved, Denmark.
Nadia Harbeck and Friederike Banning assisted in the study design and coordinated local approvals, participant recruitment, and data acquisition in Munich, Germany.
Eva Batista assisted in the study design and coordinated local approvals, participant recruitment, and data acquisition in Lisbon, Portugal.
Merle Hattink, Sofie A.M. Gernaat, and Helena Verkooijen assisted in the study design, coordinated local approvals, participant recruitment, and data acquisition in Utrecht, Netherlands.
Desiree van den Bongard and Karin Meijer assisted in the study design and coordinated local approvals, participant recruitment, and data acquisition in Amsterdam, Netherlands.
Icro Meattini and Lorenzo Livi assisted in the study design and coordinated local approvals, participant recruitment, and data acquisition in Florence, Italy.
Ivica Ratosa assisted in the study design and coordinated local approvals, participant recruitment, and data acquisition in Ljubljana, Slovenia.
Frederik V. Carstensen and Maja V. Maraldo conducted the statistical analysis and drafted the initial manuscript.
All authors provided critical manuscript revisions and editing. All authors approved the final version of the manuscript and agree to be accountable for all aspects of the work.
References
[1] European Commission. Breast cancer burden EU-27 [Internet]. [cited 2025 Mar 20]. Available from: https://ecis.jrc.ec.europa.eu/cancer-factsheets-eu-27-countries-2022
[2] SEER*Explorer: An interactive website for SEER cancer statistics [Internet]. Surveillance Research Program, National Cancer Institute. [cited 2025 Apr 16]. Available from: https://seer.cancer.gov/statistics-network/explorer/
[3] DBCG. Danish Breast Cancer Group quality database for breast cancer – National Annual Report 2023. Regionernes Kliniske Kvalitetsudviklingsprogram; 2024. Available from: https://dbcg.dk/kvalitetsdatabasen/rapporter [cited 2025 Mar 20]
[4] Lundberg FE, Andersson TML, Lambe M, Engholm G, Mørch LS, Johannesen TB, et al. Trends in cancer survival in the Nordic countries 1990–2016: the NORDCAN survival studies. Acta Oncol. 2020;59(11):1266–74. https://doi.org/10.1080/0284186X.2020.1822544
[5] Ng HS, Vitry A, Koczwara B, Roder D, McBride ML. Patterns of comorbidities in women with breast cancer: a Canadian population-based study. Cancer Causes Control. 2019;30(9):931–41. https://doi.org/10.1007/s10552-019-01203-0
[6] Rey D, Touzani R, Bouhnik AD, Rousseau F, Monet A, Préau M, et al. Evolution of physical activity and body weight changes in breast cancer survivors five years after diagnosis – VICAN 2 & 5 French national surveys. Breast. 2021;59:248–55. https://doi.org/10.1016/j.breast.2021.07.012
[7] Arneja J, Brooks JD. The impact of chronic comorbidities at the time of breast cancer diagnosis on quality of life, and emotional health following treatment in Canada. PLoS One. 2021;16(8):e0256536. https://doi.org/10.1371/journal.pone.0256536
[8] Taylor CW, Kirby AM. Cardiac side-effects from breast cancer radiotherapy. Clin Oncol (R Coll Radiol). 2015;27(11):621–9. https://doi.org/10.1016/j.clon.2015.06.007
[9] ARTILLERY. Study group webpage [Internet]. Available from: https://artillery-project.eu/
[10] Gal R, van Velzen SGM, Hooning MJ, Emaus MJ, van der Leij F, Gregorowitsch ML, et al. Identification of risk cardiovascular disease by automatic quantification of coronary artery calcifications on radiotherapy planning CT scans in patients with breast cancer. JAMA Oncol. 2021;7(7):1024–32. https://doi.org/10.1001/jamaoncol.2021.1144