ORIGINAL ARTICLE
How does pulmonary function impact QoL in patients with locally advanced NSCLC treated with chemoradiotherapy and durvalumab?
Frigg Å. Sommervolla, Henrik Horndalsveenb,c,d  , Dag Einar Sommervolle, Jussi Koivunenf,g
, Dag Einar Sommervolle, Jussi Koivunenf,g  , Tarje Onsøien Halvorsenh,i
, Tarje Onsøien Halvorsenh,i  , Bjørn Henning Grønbergh,i
, Bjørn Henning Grønbergh,i  , Marianne Aaneruda,j, Saulius Cicenask, Nina Helbekkmol, Jarkko Ahvonenm
, Marianne Aaneruda,j, Saulius Cicenask, Nina Helbekkmol, Jarkko Ahvonenm  , Maria Silvoniemin
, Maria Silvoniemin  , Gina Barrerao, Maria M. Bjaanæsb,c
, Gina Barrerao, Maria M. Bjaanæsb,c  , Vilde D. Haakensenb
, Vilde D. Haakensenb  , Åsa Öjlertb,c, Kersti Oselinp
, Åsa Öjlertb,c, Kersti Oselinp  , Åslaug Hellandb,c,d
, Åslaug Hellandb,c,d  and Tesfaye Madeboa,o
and Tesfaye Madeboa,o 
aDepartment of Clinical Science, University of Bergen, Bergen, Norway; bDepartment of Cancer Genetics, Institute for Cancer Research, Oslo University Hospital, Oslo, Norway; cDepartment of Oncology, Oslo University Hospital, Oslo, Norway; dDepartment of Clinical Medicine, University of Oslo, Oslo, Norway; eNMBU, Ås, Norway; fDepartment of Oncology and Radiotherapy, Oulu University Hospital, Oulu, Finland; gMedical Research Center Oulu, Oulu, Finland; hDepartment of Clinical and Molecular Medicine, NTNU, Norwegian University of Science and Technology, Trondheim, Norway; iDepartment of Oncology, St. Olavs Hospital, Trondheim University Hospital, Trondheim, Norway; jDepartment of Thoracic Medicine, Haukeland University Hospital, Bergen, Norway; kDepartment of Thoracic Surgery and Oncology, National Cancer Center, Affiliate of Vilnius University Hospital Santaros Klinikos, Vilnius, Lithuania; lDepartment of Pulmonology, University Hospital of North Norway, Tromsø, Norway; mTays Cancer Center, Department of Oncology, Tampere University Hospital, Tampere, Finland; nDepartment of Pulmonary Medicine, Turku University Hospital, Turku, Finland; oDepartment of Pulmonology, Stavanger University Hospital, Stavanger, Norway; pOncology and Haematology Clinic, North Estonia Medical Centre, Tallinn, Estonia
ABSTRACT
Background: Impaired pulmonary function is common among patients with lung cancer and may negatively affect health-related quality of life (HRQoL). The primary objective of the present sub-study of the DART-trial was to assess the overall quality of life changes during treatment and stratified by the presence of Chronic Obstructive Pulmonary Disease (COPD).
Methods: The investigator-initiated DART trial (NCT04392505) included patients with unresectable stage III non-small cell lung cancer (NSCLC) treated with chemoradiotherapy followed by durvalumab. Baseline pulmonary function was measured by spirometry, and patients were stratified by FEV1/FVC <70% (COPD) or ≥70% (non-COPD). HRQoL was assessed regularly using the EORTC QLQ-C30 and QLQ-LC13 questionnaires at screening and during treatment. A difference in mean score of ≥10 was defined as clinically significant.
Results: A total of 86 patients initiated durvalumab and completed at least one HRQoL assessment; pulmonary function data were available for 64 patients. For the overall cohort, quality of life scores remained stable throughout treatment. Patients with COPD consistently reported lower global health scores than those with preserved lung function. The global health score among patients with COPD was not significantly different at end of treatment compared to baseline, however significantly lower than patients without COPD. Symptom trajectories across QLQ-C30 scales were stable in both groups. Dyspnoea was more prevalent among patients with COPD. In the LC13 module, no clinically significant differences were observed except for dyspnoea, which was consistently higher among patients with COPD.
Interpretation: The HRQoL remained stable during chemoradiotherapy and durvalumab treatment in stage III NSCLC patients. Impaired lung function was associated with modestly lower HRQoL, though larger studies are needed to confirm subgroup effects.
KEYWORDS: Cancer; NSCLC; quality of life; COPD; lung
Citation: ACTA ONCOLOGICA 2026, VOL. 65, 232–240. https://doi.org/10.2340/1651-226X.2026.45040.
Copyright: © 2026 The Author(s). Published by MJS Publishing on behalf of Acta Oncologica. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/).
Received: 12 November 2025; Accepted: 19 February 2026; Published: 19 March 2026
CONTACT: Åslaug Helland ahelland@medisin.uio.no Oslo universitetssykehus HF, Radiumhospitalet, Postboks 4953 Nydalen, 0424 Oslo, Norway
Competing interests and funding: Henrik Horndalsveen: Advisory board: Johnson and Johnson. Honoraria: AstraZeneca, Pfizer, Roche.
Vilde Drageset Haakensen: Advisory board: AstraZeneca, Merck Sharp & Dome, Johnson & Johnson, Novartis, Bristol-Myers Squibb. Honoraria: AstraZeneca, Merck Sharp & Dome, Johnson & Johnson, Novartis, Bristol-Myers Squibb, Pfizer, Takeda.
Tesfaye Madebo: Advisory board: Johnson and Johnson. Honoraria: AstraZeneca, GlaxoSmithKline, Takeda AS, Merck Sharp & Dome.
Bjørn Henning Grønberg: Advisory board: Janssen, Accord, Merck Sharp & Dome, AstraZeneca, Pharmacosmos. Honoraria: AstraZeneca, Pfizer, Accord, Eli Lilly, Merck Sharp & Dome, Gilead, Bristol-Myers Squibb. Research funding: Roche, AstraZeneca.
Tarje Onsøien Halvorsen: Advisory board: AstraZeneca, Sanofi, Immedica. Honoraria: AstraZeneca, Takeda, Merck Sharp & Dome, Pfizer. Research funding: Roche, AstraZeneca
Jussi Koivunen: Honoraria: Roche, AstraZeneca, Johnson and Johnson, Bristol-Myers Squibb, Merck Sharp & Dome, Amgen, Merck KGaA, Novartis, Sanofi and Pfizer. Research Funding: Institutional grants from AstraZeneca and Roche outside of current study. Lecturing: Siemens Healthineers. Employment: Former employee of Faron Pharmaceuticals.
Kersti Oselin: Advisory board: Merck Sharp & Dome, AstraZeneca, Roche. Research Funding: Optellum.
Marianne Aanerud: Honoraria: Bristol-Myers Squibb, Astra Zeneca.
Jarkko Ahvonen: Advisory board: AstraZeneca.
Maria Silvoniemi: Advisory board: AstraZeneca, Merck Sharp & Dome, Johnson and Johnson, Bristol-Myers Squibb, Pfizer, Roche. Honoraria: AstraZeneca, Merck Sharp & Dome, Johnson and Johnson, Bristol-Myers Squibb, Pfizer, Roche, Boehringer-Ingelheim.
Saima Farooqi: Honoraria: Bristol-Myers Squibb.
Åsa Kristina Öjlert: Advisory board: Sanofi.
Åslaug Helland: Research Funding: Roche, AstraZeneca, Novartis, Incyte, Eli Lilly, Bristol-Myers Squibb, Ultimovacs, Merck, GlaxoSmithKline, Illumina, Nanopore, Johnson and Johnson, BeOne. Advisory boards and Honoraria: ABBVIE, Takeda, AstraZeneca, Roche, Pfizer, Janssen, EliLilly, Bristol-Myers Squibb, PierreFabre, Bayer, Merck Sharp & Dome, Novartis, Merck, Sanofi, Medicover. All funds go to Oslo University Hospital.
The remaining authors declare that they have no conflicts of interests.
The study has received an unrestricted research grant from ASTRA Zeneca.
Introduction
Lung cancer is one of the most common malignancies and the leading cause of cancer-related mortality worldwide [1]. In Norway, 3435 new cases were diagnosed in 2024, and 2243 patients died from the disease (Cancer Registry of Norway [https://www.fhi.no/contentassets/1d3cf9facb9747a1b9148cb23a7f7c54/cancer-in-norway-2024.pdf]). Approximately 20–30% of patients with non-small cell lung cancer (NSCLC) present with stage III, locally advanced disease [2].
For patients with good performance status and unresectable stage III NSCLC without genetic alterations in the genes Epidermal Growth Factor Receptor (EGFR) or Anaplastic Lymphoma Kinase (ALK), the standard of care is radiotherapy given concurrently with platinum-based doublet chemotherapy (chemoradiation), followed by up to 1 year of durvalumab in patients with tumours expressing the protein Programmed Death -Ligand 1 (PD-L1) [3]. This approach, modelled by the PACIFIC trial, achieves cure in a subset of patients, however, the median progression-free survival is limited, and only 42% of patients are alive at 5 years [4, 5].
Approximately 40–70% of NSCLC patients also suffer from Chronic Obstructive Pulmonary Disease (COPD) [6–8]. The two diseases share major risk factors such as smoking, environmental pollutants, and occupational carcinogen exposure. Proposed mechanisms linking COPD and lung cancer include genetic predisposition, epigenetic regulation, and chronic local and systemic inflammation [9].
The COPD has been identified as an independent risk factor for mortality in stage III NSCLC, particularly in squamous cell carcinoma [10–12], although some studies report conflicting results [13]. Nevertheless, it’s important to note that standard eligibility criteria for concurrent chemotherapy and radiotherapy trials in locally advanced NSCLC typically exclude individuals with advanced COPD and cardiac comorbidities. In addition, tobacco smoking in cancer patients is associated with increased treatment toxicity, higher risk of a treatment failure, and poorer quality of life [14].
The coexistence of COPD complicates diagnosis and management of NSCLC. Overlapping symptoms may delay diagnosis or lead to misinterpretation [15]. Radiotherapy and immunotherapy can further impair pulmonary function due to inflammation, pneumonitis, or fibrosis [16–19]. Baseline pulmonary function parameters, such as Forced Exporatory Volume in 1 Second (FEV1), Forced Vital Capacity (FVC and Diffusing Capasity of the Lungs for Carbon Monoxide (DLCO), may predict risk of radiation pneumonitis [20].
While survival remains a central outcome, health-related quality of life (HRQoL) is increasingly recognized as a critical factor in treatment planning, especially given the substantial symptom burden experienced by these patients, including fatigue, pain, and impaired physical and social functioning. Two systematic reviews – Reale et al. and Marandino et al. – highlight persistent deficiencies in the reporting and integration of HRQoL data in phase III oncology trials [21, 22]. Despite growing awareness, HRQoL outcomes are often under-reported, delayed or omitted, limiting their influence on clinical decision-making [2]. Moreover, the impact of baseline pulmonary function on HRQoL during curative-intent therapy for stage III NSCLC remains poorly understood and there is a need for further studies [22, 23].
In the Durvalumab after RadioTherapy Trial (the Dart Trial), patient well-being is monitored every 6–12 weeks using two validated instruments: the European Organisaton For Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire - 30 (QLQ-C30) (version 3) and the lung cancer-specific QLQ-LC13, capturing physical, emotional, and social dimensions. Here, we report changes in symptoms and functioning according to pre-treatment pulmonary function.
Material and methods
The DART-study
The DART-study is an open-label, multinational, investigator-initiated phase 2 trial aiming to identify biomarkers for stratifying treatment for patients with locally advanced non-small cell lung cancer (NSCLC). The study was conducted at 10 hospitals in Norway, Finland, Lithuania, and Estonia. Inclusion started on May 2020 and ended on December 2023. Patients with unresectable stage III NSCLC were enrolled and treated with curatively intended chemoradiotherapy (CRT), consisting of two cycles of platinum-based doublet chemotherapy every 3 weeks and radiotherapy 60–66 Gy in 30–33 fractions. Patients without disease progression following CRT received durvalumab 1500 mg every 4 weeks, preferably starting within 5 weeks of CRT completion, and continued until progression, intolerable toxicity, or a maximum duration of 12 months independent of PDL1 expression. Participants not starting durvalumab were excluded from further analyses. The follow-up includes a safety follow-up for up to 5 years, and a survival follow-up for up to a total of 10 years. Approval was granted by the Regional Committee for Medical and Health Research Ethics (reference 48665, November 28, 2019). All participants provided written informed consent. The trial is registered at ClinicalTrials.gov (NCT04392505). The primary endpoint in the DART-study was to determine how tumour mutational burden affected hazard ratio, with several secondary and exploratory endpoints, including the HRQoL analyses. The study was powered for the primary endpoint, and no estimation of power for the HRQoL analyses was performed. No specific inclusion requirements were defined regarding lung function, as this was a pragmatic trial with inclusion following clinical practice. For the analyses of HRQoL, we included patients completing at least one questionnaire at screening, and for the analyses related to lung function, we included patients with baseline measurement of lung function.
Quality of life measurements
HRQoL was assessed using the EORTC QLQ-C30 (Version 3) and the lung cancer module LC13 from the screening timepoint (before chemoradiation) and at cycle 3 (6 weeks) and thereafter every 12 weeks during durvalumab treatment. These instruments enable a thorough assessment of the patients’ quality of life, encompassing physical, emotional, and social dimensions. QLQ-C30 is a general questionnaire for cancer patients, while QLQ-LC13 incorporates dimensions highly relevant for lung cancer patients.
The primary objective of this study was to assess the overall quality of life changes during treatment and stratified by the presence of COPD. Through this approach, the DART study aims to gather nuanced insights into the experiences and needs of lung cancer patients with clinically relevant comorbidity. The baseline was at inclusion in the study, before starting treatment.
Pulmonary function
Pulmonary function tests, by spirometry, were conducted in adherence to the guidelines outlined by the American Thoracic Society/European Respiratory Society (ATS/ERS) and local standards for participants in the study, prior to study treatment.
Patients were classified according to the severity of airway limitation, as outlined by the latest Global Initiative for Chronic Obstructive Lung Disease (GOLD) strategy [24]. Specifically, individuals with a postbronchodilator FEV1/FVC ratio below 70% as assessed before starting chemoradiation, were identified as having COPD.
Statistical analysis
Scores for the QLQ-C30 and QLQ-LC13 questionnaires were calculated according to the EORTC Scoring Manual, with raw scores standardized by linear transformation to range from 0 to 100 [25]. A higher score represents a better level of functioning and global health status/QoL or greater symptom severity. Clinically meaningful changes were defined as an absolute change (increase or decrease) in score from baseline of ≥10 points [26]. The following items were analysed: Global Health, Emotional functioning, Fatigue, Pain, Dyspnoea, from the QLQ-C30, and Arm/Shoulder pain, Chest pain, Other pain, Cough, Hemoptysis, Dyspnoea from the QLQ-LC13. We have used data from baseline and at cycle 3, 6, 9, and 12 of durvalumab. Scores from patients with and without COPD were compared. Data were reported using descriptive statistics with percentages, means, and standard deviation. We did not perform imputations of missing data. The results are given as the mean values ± one standard deviation unless otherwise stated. R (version 4.5.1) was used to perform calculations and produce graphical figures.
Results
Patients
Of the 123 patients included in the DART-trial, 86 NSCLC patients started treatment with durvalumab. All 86 completed at least one QoL questionnaire and were included in the present analyses. Figure 1 shows a study flowchart illustrating patient enrolment, exclusions, and pulmonary function test availability.
Characteristics of the 86 patients are shown in Table 1. The study cohort consisted of 52 men (60.5%) and 34 women (39.5%), with a median age of 69 years (range 36–85). Pulmonary function was recorded for 64 patients (74.4%) (Table 1), of whom 40 (62.5%) of the patients had FEV1/FVC < 70%, and 24 (37.5%) had FEV1/FVC ≥ 70%. The patients receiving durvalumab received a median of 13 cycles (average 10 cycles). The median Progression Free Survival (PFS) was 18.9 months.
Baseline Patient Recorded Outcome Measurements (PROMs)
An overview of baseline patient-reported outcome scores stratified by pulmonary function is presented in Table 2. Patients with FEV1/FVC < 0.7 reported lower global health and some symptoms were more severe compared to those with FEV1/FVC ≥ 0.7. Specifically, patients with reduced lung function (FEV1/FVC < 0.7) reported numerically lower (but not significantly different) scores on global health (60.63 ± 18.10 vs. 66.67 ± 17.92) and reported higher levels of dyspnoea (45.83 ± 28.93 vs. 29.17 ± 28.34) in the EORTC QLQ-C30 symptom scale (v3). The emotional function was not significantly different in the two groups. There were no significant differences in the baseline scores as measured by EORTC QLQ-LC13 between patients with or without COPD.
| All patients (N = 86) | Patients (N = 40) FEV1/FVC < 0.7 | Patients (N = 24) FEV1/FVC ≥ 0.7 | ||||
| N | Score | N | Score | N | Score | |
| EORTC QLQ-C30 function scalea | ||||||
| Emotional function | 83 | 78.51 ± 20.75 | 40 | 78.54 ± 19.60 | 22 | 80.30 ± 17.92 |
| Global Health | 83 | 61.95 ± 20.04 | 40 | 60.63 ± 18.10 | 23 | 66.67 ± 24.10 |
| EORTC QLQ-C30 symptom scaleb | ||||||
| Fatigue | 82 | 31.03 ± 24.43 | 38 | 36.26 ± 24.60 | 23 | 22.22 ± 21.19 |
| Pain | 83 | 16.67 ± 24.00 | 40 | 20.83 ± 25.81 | 23 | 10.87 ± 19.85 |
| Dyspnoea | 86 | 34.50 ± 28.67 | 40 | 45.83 ±28.93 | 24 | 29.17 ± 28.34 |
| EORTC QLQ-LC13 symptom scaleb | ||||||
| Arm / shoulder pain | 86 | 14.73 ± 25.87 | 40 | 15.00 ± 26.09 | 24 | 15.28 ± 27.77 |
| Chest pain | 85 | 15.29 ± 23.32 | 40 | 14.17 ± 23.74 | 23 | 13.04 ± 19.43 |
| Other pain | 84 | 12.30 ± 20.55 | 39 | 11.11 ± 20.71 | 24 | 13.89 ± 21.80 |
| Cough | 86 | 45.35 ± 26.53 | 40 | 47.50 ± 28.13 | 24 | 41.67 ± 24.47 |
| Hemoptysis | 86 | 9.30 ± 20.86 | 40 | 12.50 ± 24.68 | 24 | 9.72 ± 20.80 |
| Dyspnoea | 86 | 26.87 ± 21.04 | 40 | 34.44 ± 23.84 | 24 | 18.06 ± 15.99 |
| N = number of patients. Scores are presented as mean value ± standard deviation. aHigher score indicates higher functioning (scale 0–100), bLower score indicates milder symptoms (scale 0–100). | ||||||
EORTC QLQ-C30 function scale
The mean overall global health (± SD) for all patients who initiated durvalumab treatment, across five time points (V00 (Screening), and cycle (C) C3, C6, C9, C12) are shown in Figure 2a. Overall, global health scores remained relatively stable across treatment cycles, with mean values consistently around 60–70. A slight increase was observed around C6, but this was not sustained, and the overall trajectory suggests no significant decline in perceived health status over time.
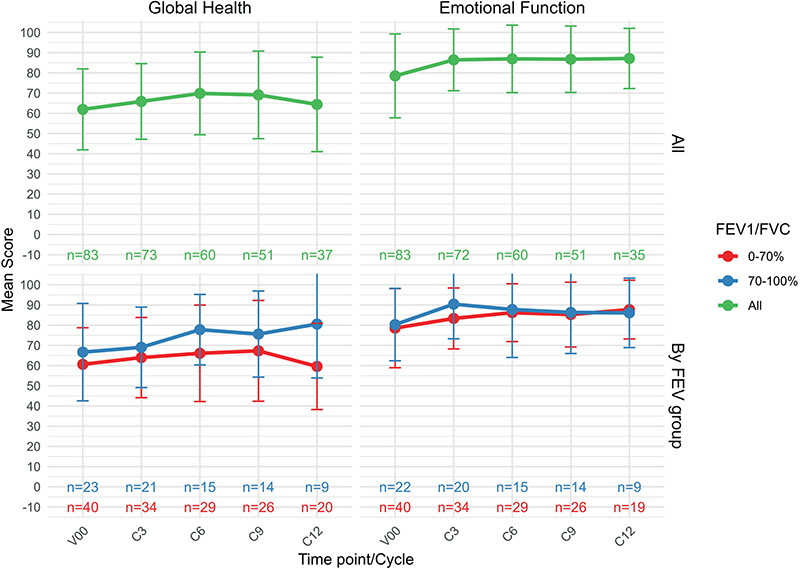
Figure 2. (a) Mean global health function (± SD) for all patients who initiated durvalumab treatment, across five time points (V00, (screening), C3 (cycle 3), C6 (cycle 6), C9 (cycle 9), C12 (cycle 12)). N* = Number of patients still on treatment. (b) Mean emotional function scores (± SD) for all patients who initiated durvalumab treatment, across five time points (V00, C3, C6, C9, C12). (c) Mean scores for global health for patients treated with durvalumab stratified by FEV1/FVC < or ≥ 0.7 (N = 64). (d) Mean scores for emotional function for patients treated with durvalumab stratified by FEV1/FVC group (N = 64).
Figure 2b shows the mean emotional function (± SD) for all patients who initiated durvalumab treatment, revealing a slight non-significant increase in emotional function during the treatment period.
Figure 2c shows mean global health scores stratified by pulmonary function (± SD). Overall, patients without COPD reported higher global health scores than those with COPD (approximately 6–20 points higher). Over the study period, mean global health scores showed a modest but steady increase among patients without COPD whereas those with COPD exhibited a slight initial improvement followed by a decline at the final assessment. Consequently, the difference between the two groups became more pronounced over time (Figure 2c). Importantly, there was no indication of a significant progressive decline in either group during treatment. Figure 2d shows that the emotional function increases non-significantly in both groups.
EORTC QLQ-C30 symptom scale
When stratified by pulmonary function, most QLQ-C30 symptom scores remained broadly stable during treatment, as shown in Figure 3. No clinically significant changes were observed for dyspnoea, fatigue or pain, in either groups, with both groups displaying stable trajectories across treatment cycles. Symptom scores for both fatigues, pain and dyspnoea (QLQ-C30 v3) were consistently higher in the impaired lung function group as compared to the patients with no COPD, although the difference diminished over time for all three symptoms over time.
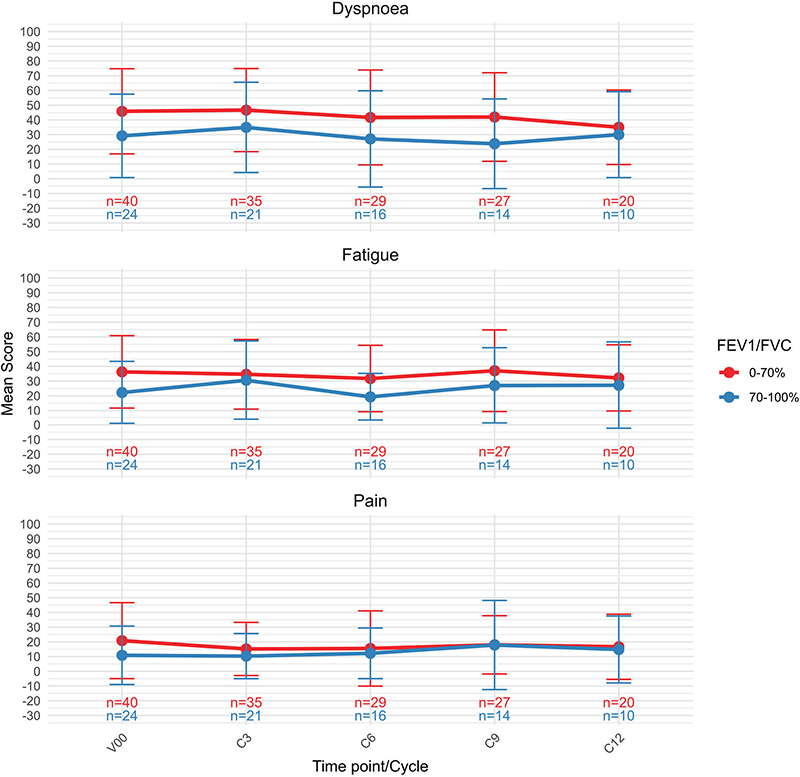
Figure 3. EORTC QLQ-C30 symptoms. A higher score represents more symptoms / higher burden. V00 = screening, C3 = cycle 3, C6 = cycle 6, C9 = cycle 9, C12 = cycle 12.
EORTC QLQ-LC13
Lung cancer specific symptoms were assessed thought EORTC QLQ-LC13. As shown in Figure 4, we see mean scores for six common symptoms stratified by FEV1/FVC. Across the LC13 symptom scales, most scores, except dyspnoea were similar between patients with COPD (FEV1/FVC <70%) and without COPD (FEV1/FVC ≥ 70%) at baseline. There was a clinically significant difference at baseline for dyspnoea, but this difference diminished over time, at Cycle 12, there was no significant difference.
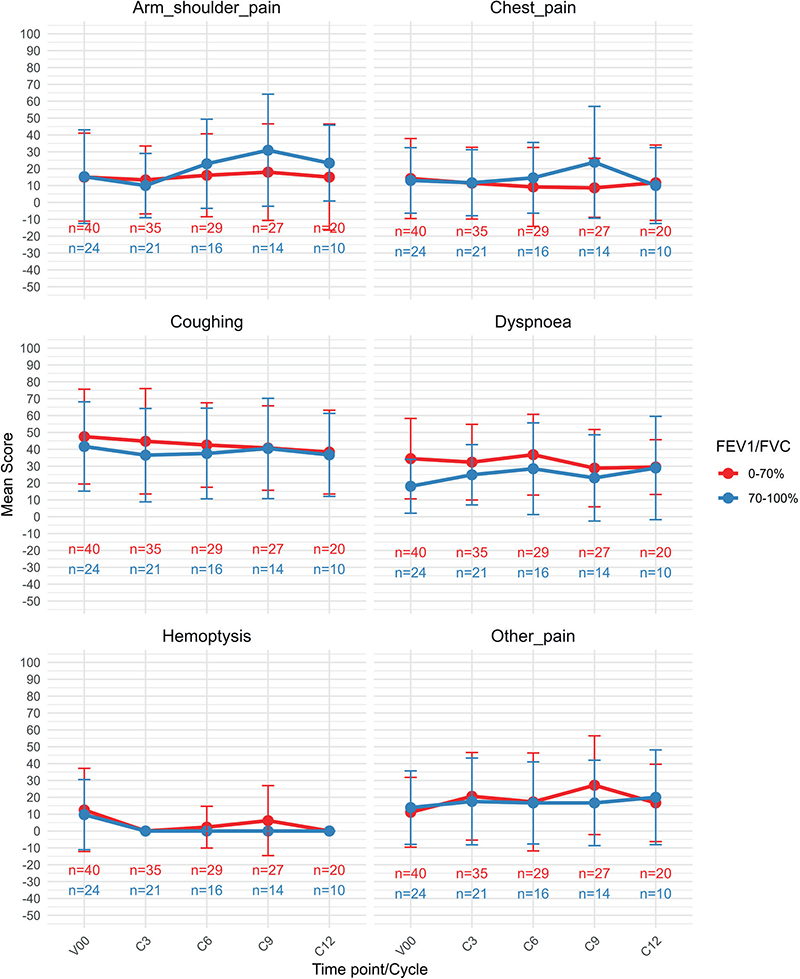
Figure 4. EORTC QLQ-LC13 symptoms. A higher score represents more symptoms / higher burden. V00 = screening, C3 = cycle 3, C6 = cycle 6, C9 = cycle 9, C12 = cycle 12.
Discussion and conclusion
In our study population, over 60% of patients had coexisting COPD, placing them at the higher end of the prevalence range reported in other settings (40–70% among NSCLC patients) [6–8]. The COPD group have more squamous cell carcinoma, more patients with a smoking history and more patients in Eastern Cooperative Oncology Group (ECOG) Performance Status 1. Generally, the patients with COPD are more frail, and it is important to learn more about how this impacts the treatment tolerability.
Our results show that there was no significant difference over time in overall global health as assessed through the EORTC QLQ-C30, for patients with locally advanced NSCLC treated with chemoradiation and durvalumab, when looking at the whole group (Figure 2a). This aligns well with a previous study in a similar population [27]. Hui and coworkers reported global health status and quality of life remained stable during treatment with chemoradiation ± durvalumab in the PACIFIC trial [27]. This may indicate that patients tolerate the treatment reasonably well and do not experience a decline in quality of life compared to their baseline level. However, when looking at global health in two groups with different pulmonary function (FEV1/FVC < and ≥ 0.7), we see that the patients with COPD showed a steady improvement in global health across all assessments except at Cycle 12, where a significant decline was observed. This deterioration suggests an increased treatment burden in this vulnerable cohort. The fact that one‑third of these patients are active smokers may further potentiate treatment‑related toxicity, accelerate decline in lung function, and ultimately contribute to poorer quality‑of‑life outcomes.
We identified clinically significant differences in the QLQ-C30 symptoms Fatigue, Pain and Dyspnoea at baseline, however diminishing difference during the treatment timeline. The number of responders is significantly lower at end of treatment as compared to baseline, and the decline in responders might of course influence the results. Patients with the most severe symptoms might not be represented at Cycle 12.
In the QLQ-LC13 symptom scale, no significant group differences were identified, except for dyspnoea at baseline, where the group with a poor pulmonary function reported higher scores, as would be expected. The two groups differ in several aspects, like smoking history and performance status, which might impact the overall well-being of the patients.
A limitation of this study is the relatively small sample size, and challenges in controlling for other factors impacting the HRQoL, which may reduce the statistical power and limit the generalizability of the findings. As expected, compliance with the questionnaires decrease over time, which partly is related to disease progression and deaths. Moreover, missing data on several items, including DLCO, could have offered further insight into the interplay between lung function and HRQoL, representing an important avenue for future research.
For patients with stage III NSCLC, chemoradiation followed by immunotherapy has significantly improved prognosis [4, 5, 19]. Despite the high prevalence of comorbidities such as COPD, our findings indicate that the treatment is generally well tolerated across patient groups. Although patients with impaired lung function reported lower overall global health scores at end of treatment, no substantial differences were observed in specific QLQ-C30 symptom domains between groups, at end of treatment. These results reinforce the safety and efficacy of this treatment approach, even in patients with compromised pulmonary function, and highlight the importance of individualized monitoring and multidisciplinary care to optimize clinical outcomes.
Acknowledgments
This work was supported by the regional Health Authorities in Norway (grant ID 26011) and the South-Eastern Regional Health Authorities (grant 2019110), The Cancer Society (212426) and AstraZeneca for research support.
The authors would like to thank all participating patients and their families, study nurses, engineers and lab personnel in all hospitals for their valuable contribution to the trial.
NPCM 2025 was financially supported by the Acta Oncologica Foundation.
Data availability statement
Data are available upon request.
Ethics declarations & trial registry information
All research reported here have been conducted in an ethical and responsible manner and is in full compliance with all relevant codes of experimentation and legislation. All patients gave signed an informed consent, and the regional ethics committee has approved the study (#48655). The study is now transferred to CTIS. It is registered in the Clinical.trials.gov: NCT04392505.
Authors’ contributions
CRediT authorship contribution
FÅS: Data Curation, Analyses, Visualization, Writing original draft, revising manuscript.
HH: Data Curation, Investigation, Visualization, review editing.
DES: Analyses and visualization.
JK: Investigation, Writing – review editing.
TOH: Investigation Writing – review editing.
BHG: Investigation, Writing – review editing.
MA: Investigation, Writing – review editing.
SC: Investigation, Writing – review editing.
NH: Investigation, Writing – review editing.
JA: Investigation, Writing – review editing.
MS: Investigation, Writing – review editing.
GB: Investigation, Writing – review editing.
MMB: Investigation, Writing – review and editing.
VDH: Data Curation, Methodology, review editing.
ÅKÖ: Investigation, Writing – review editing.
KO: Investigation, Writing – review editing.
ÅH: Conceptualization, Data Curation, Funding Acquisition, Investigation, Methodology, Project Administration, Supervision, Writing – Review editing.
TM: Investigation, Methodology, Supervision, Writing – review editing.
References
[1] Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Abate D, Abbasi N, Abbastabar H, Abd-Allah F, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the Global Burden of Disease Study. JAMA Oncol. 2019;5(12):1749–68.
[2] Casal-Mourino A, Ruano-Ravina A, Lorenzo-Gonzalez M, Rodriguez-Martinez A, Giraldo-Osorio A, Varela-Lema L, et al. Epidemiology of stage III lung cancer: frequency, diagnostic characteristics, and survival. Transl Lung Cancer Res. 2021;10(1):506–18. https://doi.org/10.21037/tlcr.2020.03.40
[3] Zer A, Ahn MJ, Barlesi F, Bubendorf L, De Ruysscher D, Garrido P, et al. Early and locally advanced non-small-cell lung cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2025;36(11):1245–62. https://doi.org/10.1016/j.annonc.2025.08.003
[4] Spigel DR, Faivre-Finn C, Gray JE, Vicente D, Planchard D, Paz-Ares L, et al. Five-year survival outcomes from the PACIFIC trial: durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. J Clin Oncol. 2022;40(12):1301–11. https://doi.org/10.1200/JCO.21.01308
[5] Antonia SJ. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. reply. N Engl J Med. 2019;380(10):990. https://doi.org/10.1056/NEJMc1900407
[6] Loganathan RS, Stover DE, Shi W, Venkatraman E. Prevalence of COPD in women compared to men around the time of diagnosis of primary lung cancer. Chest. 2006;129(5):1305–12. https://doi.org/10.1378/chest.129.5.1305
[7] Young RP, Hopkins RJ, Christmas T, Black PN, Metcalf P, Gamble GD. COPD prevalence is increased in lung cancer, independent of age, sex and smoking history. Eur Respir J. 2009;34(2):380–6. https://doi.org/10.1183/09031936.00144208
[8] Wasswa-Kintu S, Gan WQ, Man SF, Pare PD, Sin DD. Relationship between reduced forced expiratory volume in one second and the risk of lung cancer: a systematic review and meta-analysis. Thorax. 2005;60(7):570–5. https://doi.org/10.1136/thx.2004.037135
[9] Durham AL, Adcock IM. The relationship between COPD and lung cancer. Lung Cancer. 2015;90(2):121–7. https://doi.org/10.1016/j.lungcan.2015.08.017
[10] Chiu KC, Lin WC, Chang CL, Wu SY. Impact of chronic obstruction pulmonary disease on survival in patients with advanced stage lung squamous cell carcinoma undergoing concurrent chemoradiotherapy. Cancers (Basel). 2021;13(13):3231. https://doi.org/10.3390/cancers13133231
[11] Zhai R, Yu X, Shafer A, Wain JC, Christiani DC. The impact of coexisting COPD on survival of patients with early-stage non-small cell lung cancer undergoing surgical resection. Chest. 2014;145(2):346–53. https://doi.org/10.1378/chest.13-1176
[12] Tan LE, Razak AM, Lim CS. Association of chronic obstructive pulmonary disease and postresection lung cancer survival: a systematic review and meta-analysis. J Investig Med. 2017;65(2):342–52. https://doi.org/10.1136/jim-2016-000059
[13] Lee SJ, Lee J, Park YS, Lee CH, Lee SM, Yim JJ, et al. Impact of chronic obstructive pulmonary disease on the mortality of patients with non-small-cell lung cancer. J Thorac Oncol. 2014;9(6):812–17. https://doi.org/10.1097/JTO.0000000000000158
[14] Jassem J. Tobacco smoking after diagnosis of cancer: clinical aspects. Transl Lung Cancer Res. 2019;8(Suppl 1):S50–8. https://doi.org/10.21037/tlcr.2019.04.01
[15] Bjerager M, Palshof T, Dahl R, Vedsted P, Olesen F. Delay in diagnosis of lung cancer in general practice. Br J Gen Pract. 2006;56(532):863–8.
[16] Ellis PM, Vandermeer R. Delays in the diagnosis of lung cancer. J Thorac Dis. 2011;3(3):183–8.
[17] Smith SM, Campbell NC, MacLeod U, Lee AJ, Raja A, Wyke S, et al. Factors contributing to the time taken to consult with symptoms of lung cancer: a cross-sectional study. Thorax. 2009;64(6):523–31. https://doi.org/10.1136/thx.2008.096560
[18] Hassanzadeh C, Sita T, Savoor R, Samson PP, Bradley J, Gentile M, et al. Implications of pneumonitis after chemoradiation and durvalumab for locally advanced non-small cell lung cancer. J Thorac Dis. 2020;12(11):6690–700. https://doi.org/10.21037/jtd-20-1792
[19] Stoffers RH, Niezink AGH, Chouvalova O, Ubbels JF, Woltman-van Iersel M, Hiltermann TJN, et al. Rising incidence of radiation pneumonitis after adjuvant durvalumab in NSCLC patients treated with concurrent chemoradiotherapy. Acta Oncol. 2025;64:267–75. https://doi.org/10.2340/1651-226X.2025.42384
[20] Zhou Y, Yan T, Zhou X, Cao P, Luo C, Zhou L, et al. Acute severe radiation pneumonitis among non-small cell lung cancer (NSCLC) patients with moderate pulmonary dysfunction receiving definitive concurrent chemoradiotherapy: impact of pre-treatment pulmonary function parameters. Strahlenther Onkol. 2020;196(6):505–14. https://doi.org/10.1007/s00066-019-01552-4
[21] Marandino L, La Salvia A, Sonetto C, De Luca E, Pignataro D, Zichi C, et al. Deficiencies in health-related quality-of-life assessment and reporting: a systematic review of oncology randomized phase III trials published between 2012 and 2016. Ann Oncol. 2018;29(12):2288–95. https://doi.org/10.1093/annonc/mdy449
[22] Reale ML, De Luca E, Lombardi P, Marandino L, Zichi C, Pignataro D, et al. Quality of life analysis in lung cancer: a systematic review of phase III trials published between 2012 and 2018. Lung Cancer. 2020;139:47–54. https://doi.org/10.1016/j.lungcan.2019.10.022
[23] Waisberg F, Lopez C, Enrico D, Rodriguez A, Hirsch I, Burton J, et al. Assessing the methodological quality of quality-of-life analyses in first-line non-small cell lung cancer trials: a systematic review. Crit Rev Oncol Hematol. 2022;176:103747. https://doi.org/10.1016/j.critrevonc.2022.103747
[24] Kostikas K, Hillas G, Gogali A. 2025 GOLD report: what is new and what is noteworthy for the practicing clinician. COPD. 2025;22(1):2451613. https://doi.org/10.1080/15412555.2025.2451613
[25] Fayers PM, Bjordal K, Groenvold M, Curran D, Bottomley A, on behalf of the EORTC Quality of Life Group. The EORTC QLQ-C30 scoring manual. 3rd ed. Brussels: Organisation for Research and Treatment of Cancer; 2001.
[26] Osoba D, Rodrigues G, Myles J, Zee B, Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998;16(1):139–44. https://doi.org/10.1200/JCO.1998.16.1.139
[27] Hui R, Ozguroglu M, Villegas A, Daniel D, Vicente D, Murakami S, et al. Patient-reported outcomes with durvalumab after chemoradiotherapy in stage III, unresectable non-small-cell lung cancer (PACIFIC): a randomised, controlled, phase 3 study. Lancet Oncol. 2019;20(12):1670–80. https://doi.org/10.1016/S1470-2045(19)30519-4
