ORIGINAL ARTICLE
IMPACT OF EXERCISE TRAINING AT DIFFERENT INTENSITY LEVELS ON CARDIAC FUNCTION AND EXERCISE CAPACITY IN PATIENTS WITH CHRONIC HEART FAILURE: A PROSPECTIVE COHORT STUDY
Zhenying ZHANG, MD1#, Lizhong WANG, MS1#, Jianing XI, MS1, Xiaojing SUN, MS1 and Xingguo SUN, MD2
From the 1Cardiac Rehabilitation Center, Beijing Rehabilitation Hospital, Capital Medical University and 2National Center for Cardiovascular Disease, Fuwai Hospital, Chinese Academy of Medical Sciences, Beijing, China
Objective: To investigate training at different intensity levels on cardiac function, exercise capacity, and health-related quality of life in patients with chronic heart failure.
Methods: This prospective cohort study enrolled patients with chronic heart failure at Beijing Rehabilitaion Hospital, Beijing, China from January 2018 to January 2020. Participants received conventional therapy (non-exercise group) or therapy plus cycle ergometer exercises at an intensity of 80% anaerobic threshold (EA group) and Δ50% power above anaerobic threshold (EB group) for 12 weeks. The primary outcome was peak oxygen uptake.
Results: Forty-five patients (15/group) completed the study without serious complications. Exercise training at an intensity of Δ50% power above anaerobic threshold had better effects on exercise capacity than exercise at an intensity of 80% anaerobic threshold, as shown by a greater improvement in peak oxygen uptake (20.3 ± 4.1 vs 16.8 ± 3.2 mL/min/kg), peak O2 pulse (12.5 ± 2.3 vs 10.1 ± 2.1 mL/beat), and peak workload (123.1 ± 26.9 vs 102.8 ± 29.5 W) in patients with chronic heart failure (all p < 0.001). Exercise improved the 6-min walk test distance (control: 394.0 ± 74.1; EA: 481.4 ± 89.4; EB: 508.9 ± 92.5 m; p < 0.001) and health-related quality of life (control: 40.7 ± 12.3; EA: 16.2 ± 8.6; EB: 11.5 ± 6.4; p < 0.001).
Conclusion: Compared with an intensity of 80% anaerobic threshold, exercise training at an intensity of Δ 50% power above anaerobic threshold was safe and had better effects on cardiac function, exercise capacity, and health-related quality of life.
LAY ABSTRACT
The aim of this study was to investigate training at different intensity levels on cardiac function, exercise capacity, and health-related quality of life in patients with chronic heart failure. Exercise training at an intensity of Δ 50% power above anaerobic threshold (mild anaerobic exercise) had better effects on exercise capacity than non-exercise and intensity of 80% anaerobic threshold (aerobic exercise). In addition, exercise resulted in improved health-related quality of life compared with non-exercise. Therefore, the benefit may be greater if the exercise intensity is increased appropriately under safe conditions.
Key words: heart failure; cardiac rehabilitation; exercise test; exercise tolerance; peak oxygen uptake; left ventricular function.
Citation: J Rehabil Med 2022; 54: jrm00347. DOI: https://dx.doi.org/10.2340/jrm.v54.1023
Copyright: © Published by Medical Journals Sweden, on behalf of the Foundation for Rehabilitation Information. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Oct 3, 2022; Epub ahead of print: Oct 20, 2022; Published: Nov 04, 2022
Correspondence address: Jianing Xi, Cardiac Rehabilitation Center, Beijing Rehabilitation Hospital, Capital Medical University, Beijing, China. E-mail: 13331012530@189.cn
Competing interests and funding: The authors have no conflicts of interest to declare.
#These authors contributed equally to this work.
Chronic heart failure (CHF) is a complex clinical syndrome characterized by typical symptoms, such as breathlessness or fatigue. CHF often results in a marked reduction in physical activity and health-related quality of life (HRQoL). CHF develops in the late stages of coronary artery disease and cardiomyopathies, with an estimated prevalence of 65 million patients worldwide (1), including approximately 12.6 million in the Chinese population (2), and in-hospital mortality of 5.3% (3). In the USA, the lifetime risk of developing CHF is as high as 20% for individuals ≥40 years of age (4). CHF is associated with high morbidity and mortality, making CHF a major public health issue (4). Exercise training is strongly recommended in patients with CHF by various guidelines, including those from the European Society of Cardiology (ESC) (5) and American Heart Association (AHA) (4), and is an important component of cardiac rehabilitation programmes offered to patients with CHF.
Several trials have confirmed that exercise training could improve exercise capacity and HRQoL and may reduce mortality and hospitalization in patients with CHF (6–11). Individualized exercise prescription should be implemented under the guidance of professionals to achieve effective functional improvement, determine the patient’s acceptability of exercising, and ensure safety. Cardiopulmonary exercise testing (CPET) is a non-invasive method for assessing cardiopulmonary performance during exercises and is an effective asset in managing patients with CHF (10).
Exercise intensity is the key component in exercise training programmes, and it is crucial not only for obtaining the desired benefits, but also for ensuring patient safety (6, 12). However, there is no consensus to date as to what level of exercise intensity will lead to better cardiac rehabilitation efficacy among patients with CHF (13–15). Low-to-moderate intensity exercises have been investigated in several studies with encouraging results (16, 17), but whether higher intensity exercise has added benefits in improving exercise capacity remains unclear.
A previous study by the authors verified the efficacy and safety of exercise training at an intensity of Δ 50% above anaerobic threshold (AT) power in patients with stable CHF (18). Based on the previously obtained results, the current study aimed to investigate the effects of exercise training at different intensity levels on cardiac function, exercise capacity, plasma B-type natriuretic peptide (BNP), and HRQoL in patients with CHF. The results could provide a basis for developing clinical cardiac rehabilitation programmes.
METHODS
Study design and participants
This prospective cohort study enrolled patients with CHF, Class II–III New York Heart Association (NYHA), and left ventricular ejection fraction (LVEF) < 45%, who underwent treatment at Beijing Rehabilitation Hospital of Capital Medical University, China, from January 2018 to January 2020. The diagnosis and treatment of CHF were performed according to the 2018 Chinese Guidelines for diagnosing and treating heart failure (19). Patients with acute coronary syndrome, acute heart failure, active myocarditis, acute pericarditis, malignant arrhythmia, severe valvular heart disease, uncontrolled hypertension, or non-cardiovascular causes of exercise limitation were excluded because these patients were unable to exercise at the intensity required in this study. All patients had to remain clinically stable for at least 1 month, without any clinical symptoms or physical signs, before participation.
This study was approved by the Institutional Ethics Committee at Beijing Rehabilitation Hospital (approval #2015bkkf-002; November 2, 2015). Each patient signed the informed consent form before performing the CPET and entering the exercise training programme.
Grouping and rehabilitation programme
The patients were prescribed exercise after a comprehensive discussion between the patient and the physician and based on the patient’s condition. The participants were grouped into a non-exercise group, an exercise at an intensity of 80% AT group (EA group), and a Δ50% power above AT group (EB group) as shown in Fig.1.
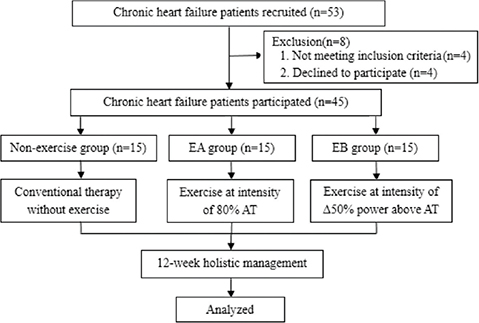
Fig. 1. Flow diagram of the study design. Exercise group A (EA group): exercised at an intensity of 80% anaerobic threshold (AT); exercise group B (EB group): exercised at an intensity of Δ50% power above AT.
All patients were on managed medicine prescriptions for secondary prevention, including renin-angiotensin system inhibition, beta-blockers, mineralocorticoid receptor antagonists, and other drugs (diuretics, digoxin, etc.) according to their conditions and the 2018 Chinese Guidelines for the diagnosis and treatment of heart failure (19). Lifestyle management, emotional and sleep management, and risk factor management were also suggested based on the guidelines. Before cardiac rehabilitation, the patient’s general state was evaluated, including cardiac diseases, co-morbidities, symptoms, medications, and risk factors. All patients were provided with individualized lifestyle recommendations and self-management strategies, including stopping smoking, avoiding excessive alcohol or fluid intake, alleviating psychological stress, and improving sleep quality. In addition, all patients received education about the causes of CHF, the symptoms, disease course, therapy, and how to control the risk factors. The participants in the non-exercise group received conventional therapy according to the 2018 Chinese Guidelines for diagnosing and treating heart failure (19), excluding exercises. The participants in the EA group and EB group underwent therapy plus cycle ergometer exercises.
A precise and individualized exercise intensity prescription was tailored according to the grouping and quantitative function evaluation by CPET. The participants in the EA group exercised at an intensity of 80% AT, and the participants in the EB group exercised at an intensity of Δ50% above AT. Δ 50% power (W) = [(power at AT – incremental rate × 0.75) + (power at peak work – incremental rate × 0.75)] / 2. The participants in the EA and EB groups underwent 5 training sessions per week for 12 weeks under multiparameter monitoring in Beijing rehabilitation hospital. After 5 min of warm-up, the participants exercised on a cycle ergometer (Custo-med, Germany) for 30 min at permanent individual power, followed by 5 min cool-down period. At the beginning of the rehabilitation course, the participants were allowed to exercise for 5–10 min and rest for 3–5 min, according to the individual states, until a total of 30 min of exercise (excluding rest) was reached. After 5–7 training sessions, the participants in the EA and EB groups were able to finish the entire exercise training without rest.
Outcomes and data collection
The primary outcome was peak oxygen uptake (VO2), measured by symptom-limited CPET according to the continuous increment power programme from Habor-UCLA Medical Center in Los Angeles, California, USA (20, 21) and expressed as a value adjusted to body weight (mL/kg/min). All CPET values were reported in absolute terms and normalized to the percentage of predicted (%pred) (22).
Secondary outcomes included AT, peak O2 pulse, peak workload, left ventricular end-diastolic diameter (LVEDD) and LVEF using echocardiography, BNP, HRQoL score, and 6-min walk test distance (6MWD). Both LVEDD and LVEF of all participants were evaluated by Doppler echocardiography in the ultrasound department. Plasma BNP concentrations were detected using a human BNP ELISA kit (Huanzhong Bioengineering Institute, Shijiazhuang, China) according to the manufacturer’s instructions. HRQoL was measured using the Minnesota Living with Heart Failure (MLWHF) questionnaire. The 6-min walk test was conducted in a standardized format, as previously reported (23). All parameters were evaluated at the baseline and the end of the 12-week rehabilitation programme.
Demographic characteristics and other clinical data were collected at baseline, including age, sex, body mass index (BMI), smoking history, co-morbidities, and medication history.
Statistical analysis
SPSS 26.0 (IBM Corp., Armonk, NY, USA) was used for statistical analysis. The Kolmogorov-Smirnov test was used to test the continuous variables for normal distribution. The continuous variables were presented as means ± standard deviation (SD). The differences among the 3 groups were assessed by 1-way analysis of variance (ANOVA) with the Student-Newman-Keuls post hoc test. The paired sample t-test was used to analyse the differences before and after the intervention. The categorical variables were presented as n (%) and analysed using the Fisher exact probability test. p-values < 0.05 were considered statistically significant.
RESULTS
Patients’ characteristics
A total of 45 participants with CHF were enrolled, 15 in each group. The mean age was 66 ± 8 years, and 87% were male. All groups were comparable in age, BMI, and pre-treatment CPET parameters. The aetiology of CHF was ischaemic in 89% of participants (Table I).
Effects of exercise training on CPET parameters
The participants in the EA group and EB group completed the 12-week exercise programme safely without serious complications. No serious cardiac events occurred during CPET and the exercise programme. The participants achieved maximal or near-maximal CPET at baseline and after the 12-week training, as reflected by the respiratory exchange ratio of >1.15.
Peak VO2, AT, and peak workload in the EA and EB groups were significantly improved by the 12-week exercise programme. After 12 weeks of exercise, AT, peak VO2, and peak workload were higher in the EA group compared with the non-exercise group (p < 0.001). In the EB group, AT, peak VO2, peak O2 pulse, and peak workload were higher than in the non-exercise group (p < 0.001). Notably, peak VO2, peak O2 pulse, and peak workload of the participants in the EB group were significantly increased by the 12-week exercise compared with the EA group (Table II).
| Parameters | Non-exercise group (n = 15) | EA group (n = 15) | EB group (n = 15) | p-value |
| Resting HR, beats/min, mean ± SD | 71.9 ± 14.3 | 74.5 ± 12.4 | 74.0 ± 12.7 | 0.847 |
| Peak HR, beats/min, mean ± SD | 113.7 ± 21.9 | 121.1 ± 20.8 | 128.2 ± 20.2 | 0.189 |
| Resting SBP, mmHg, mean ± SD | 117.7 ± 19.2 | 107.9 ± 12.8 | 119.2 ± 15.0 | 0.125 |
| Peak SBP, mmHg, mean ± SD | 151.8 ± 31.1 | 155.7 ± 25.0 | 173.7 ± 16.0# | 0.051 |
| Resting DBP, mmHg, mean ± SD | 65.9 ± 8.6 | 66.3 ± 9.7 | 70.7 ± 5.8 | 0.225 |
| Peak DBP, mmHg, mean ± SD | 74.3 ± 8.1 | 74.5 ± 14.6 | 77.7 ± 5.2 | 0.617 |
| AT | ||||
| mL/min, mean ± SD | 693.7 ± 159.6 | 901.5 ± 237.8* | 1032.2 ± 285.4# | 0.001 |
| mL/min/kg, mean ± SD | 9.8 ± 2.1 | 12.1 ± 2.3* | 13.8 ± 3.7# | 0.001 |
| % predicted, mean ± SD | 64.4 ± 17.2 | 72.1 ± 14.7 | 82.7 ± 14.0# | 0.009 |
| Peak VO2 | ||||
| mL/min, mean ± SD | 895.5 ± 200.6 | 1219.6 ± 362.5* | 1561.2 ± 437.3† | < 0.001 |
| mL/min/kg, mean ± SD | 13.1 ± 2.4 | 16.8 ± 3.2* | 20.3 ± 4.1† | < 0.001 |
| % predicted, mean ± SD | 58.6 ± 16.4 | 70.4 ± 15.8* | 80.2 ± 13.9# | 0.002 |
| Peak O2 pulse | ||||
| mL/beat, mean ± SD | 8.9 ± 2.2 | 10.1 ± 2.1 | 12.5 ± 2.3† | < 0.001 |
| % predicted, mean ± SD | 87.7 ± 28.7 | 89.9 ± 17.1 | 99.3 ± 12.0 | 0.286 |
| Peak workload | ||||
| W, mean ± SD | 76.6 ± 22.3 | 102.8 ± 29.5* | 123.1 ± 26.9† | < 0.001 |
| % predicted, mean ± SD | 72.3 ± 20.1 | 94.6 ± 26.6* | 92.4 ± 23.9# | 0.025 |
| LVEF, %, mean ± SD | 38.7 ± 4.5 | 45.0 ± 9.8* | 47.4 ± 8.0# | 0.011 |
| LVEDD, mm, mean ± SD | 58.0 ± 5.9 | 57.5 ± 5.3 | 56.8 ± 5.8 | 0.844 |
| BNP, pg/ml, mean ± SD | 293.3 ± 178.2 | 231.4 ± 222.7 | 159.9 ± 92.5# | 0.120 |
| 6MWD, m, mean ± SD | 394.0 ± 74.1 | 481.4 ± 89.4* | 508.9 ± 57.0# | < 0.001 |
| HRQoL, mean ± SD | 40.7 ± 12.3 | 16.2 ± 8.6* | 11.5 ± 6.4# | < 0.001 |
| CHF: chronic heart failure; HR: heart rate; SBP: systolic blood pressure; DBP: diastolic blood pressure; LVEF: left ventricular ejection fraction; LVEDD: left ventricular end-diastolic diameter. P: analysis of variance (ANOVA) test among 3 groups after 12 weeks. the multiple comparisons among 3 groups were assessed by ANOVA with the Student-Newman-Keuls post hoc test *Compared with non-exercise group, p < 0.05. #Compared with non-exercise group, p < 0.05. †Compared with EA group, p < 0.05. |
||||
Effects of exercise training on echocardiography, BNP, 6MWD and HRQoL
After the exercise programme, the LVEF in the EA and EB groups was increased compared with baseline. The plasma BNP levels of the EB group were decreased significantly compared with baseline. After the 12-week exercise programme, the LVEF of the EA group (45.0 ± 9.8) and the LVEF and plasma BNP of the EB group (47.4 ± 8.0; 159.9 ± 92.5) were different from those of the non-exercise group (LVEF: 38.7 ± 4.5; BNP: 293.3 ± 178.2; all p < 0.001), but there were no significant differences between the EA and EB groups (Table II).
6MWD (EA: 481.4 ± 89.4; EB: 508.9 ± 57.0; non-exercise: 394.0 ± 74.1) and HRQoL (EA: 16.2 ± 8.6; EB: 11.5 ± 6.4; non-exercise: 40.7 ± 12.3) improved significantly in the EA and EB groups compared with non-exercise group (all p < 0.001), although there were no significant differences between the EA and EB groups (Table II).
DISCUSSION
This study reported that exercise training intensity of 80% AT and Δ50% above AT, as measured by CPET, both improved exercise capacity, cardiopulmonary function, and HRQoL in patients with CHF, as demonstrated through peak VO2, peak workload, LVEF, BNP, and MLWHF questionnaire, respectively. Exercises at an intensity of Δ50% power above AT had better effects on exercise capacity than exercise at 80% AT, as shown by more obviously improved peak oxygen uptake, peak O2 pulse, and peak workload. No serious cardiac event, such as malignant arrhythmia, unstable angina, myocardial infarction, syncope, or sudden death occurred during the 12-week training programme.
Previous studies have already noted that exercise capacity and HRQoL of patients with CHF improve after exercising at an intensity level of AT (7, 16, 24–26), and this intensity level is recommended by a Chinese specialist consensus for Chinese patients with CHF (27). Giuliano et al. (25) showed that peripheral remodelling through intermittent muscular exercise (PRIME) improved the aerobic capacity of patients with CHF compared with traditional exercises, as supported by previous studies (28, 29). A previous study by the authors also obtained comparable results after 12 weeks of exercise at an intensity of Δ50% above AT (10, 18). To the best of our knowledge, the current study is the first to report that the exercise capacity of patients with CHF improves exercise capacity parameters more efficiently at an intensity of Δ 50% power above AT than at 80% AT, as indicated by peak VO2 (mL/min, mL/min/kg), peak O2 pulse (mL/beat), and peak workload, suggesting that higher intensity exercise training had a better effect on cardiac improvement. The ongoing HE-EI Trial will compare no exercise vs high-intensity exercise (70% peak VO2) vs moderate-intensity exercise (at the AT intensity) in patients with CHF (30). Although it uses different exercise intensities than in the current study, it will provide additional insights about the optimal exercise intensity in patients with CHF.
In addition, this study showed that plasma BNP levels decreased and peak SBP increased significantly in the EB group, but plasma BNP levels and peak SBP showed no obvious changes in the EA group after 12-week exercise, indicating that cardiac function was markedly improved only by exercise at an intensity of Δ50% power above AT. The precise mechanism by which exercise-based rehabilitation programme benefits patients with CHF is not completely understood, but it was reported previously that exercise training improves myocardial perfusion by alleviating endothelial dysfunction and dilating coronary vessels (31, 32), attenuates ventricular remodelling (33), and improves myocardial contractility and diastolic filling (31, 34). In addition, exercise training can improve skeletal muscle O2 transport and utilization (35), modify autonomic nervous system function (36), and attenuate the production of pro-inflammatory cytokines and natriuretic peptides (37, 38).
Core elements of an exercise programme include frequency, intensity, duration, and type, of which exercise intensity is the most important part. Despite that, there are controversies in exercise intensity evaluation and prescription formulations (13–15). Moreover, some evaluation methods have their limitations in CHF. Specifically, the HR index model, used to evaluate exercise intensity in traditional medicine, is limited due to the influence of beta-blockers (39, 40). The rating of perceived exertion (RPE) has also been used as an indicator of exercise intensity; however, it is not quantitative and is easily affected by subjective factors. Thus, symptom-limited CPET can be used to evaluate exercise capacity objectively and quantitatively and was the most suitable for the present study. Among many variables from CPET, peak VO2, which increase is related to better clinical outcomes, is one of the most sensitive independent prognostic factors in patients with CHF (41–43), as was confirmed by the current study.
The current study also showed that LVEF and 6MWD of patients with CHF increased, and the HRQoL scores of patients with CHF were decreased significantly in the EA and EB groups after the 12-week exercise intervention, but were not significantly different between the EA and EB groups after exercise training, indicating that the assessment methods, such as echocardiography and 6-min walk test, are not sensitive in evaluating exercise capacity of trained patients with CHF compared with peak VO2. Thus, the current study supports the statement that CPET parameters, especially peak VO2, are recommended to evaluate exercise training efficiency in patients with CHF.
This study has some limitations. It was a single-centre study with a small sample size and a lower ratio of female distribution (13%). Only the patients with CHF who were able to complete the 12-week exercise training programme were included. In addition, many patients treated in our rehabilitation centre less than 1 month after the last cardiovascular acute event were excluded, leading to a selection bias.
In conclusion, in stable patients with CHF and NYHA Class II–III, an exercise-based cardiac rehabilitation programme resulted in clinically important improvements in exercise capacity, cardiopulmonary function, and HRQoL. Compared with exercise training at an intensity of 80% AT, an intensity of Δ50% power above AT was safe and had better effects on cardiac function and exercise capacity.
ACKNOWLEDGEMENTS
The authors would like to thank all of the study participants.
This work was supported by the Capital Clinical Application Research (Z141107002514084), the Capital Clinical Application Research and Promotion Projects (Z161100000516127), and the Special Scientific Research Fund of Beijing Rehabilitation Hospital (2019-003, 2020-002).
REFERENCES
- Groenewegen A, Rutten FH, Mosterd A, Hoes AW. Epidemiology of heart failure. Eur J Heart Fail 2020; 22: 1342–1356. DOI:10.1002/ejhf.1858.
- Gu DF, Huang GY, He J,Wu XG, Duan XF, Stephen M, et al. Investigation of prevalence and distributing feature of chronic heart failure in Chinese adult population. Chin J Cardiol 2003; 31: 3–6. DOI: 10.3760/j:issn:0253-3758.2003.01.002.
- Zhang J, Zhang YH. China Heart Failure Registry Study – a multicenter, prospective investigation for preliminary analysis on etiology,clinical features and treatment in heart failure patients. Chin Circul J 2015; 30: 413–416. DOI:10.3969/j.issn.1000-3614.2015.05.002.
- Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Drazner MH, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2013; 62: e147–e239. DOI:10.1016/j.jacc.2013.05.019.
- Pelliccia A, Sharma S, Gati S, Bäck M, Börjesson M, Caselli S, et al. 2020 ESC Guidelines on sports cardiology and exercise in patients with cardiovascular disease. Eur Heart J 2021; 42: 17–96. DOI: 10.1093/eurheartj/ehaa605.
- Piepoli MF, Conraads V, Corrà U, Dickstein K, Francis DP, Jaarsma T, et al. Exercise training in heart failure: from theory to practice. A consensus document of the Heart Failure Association and the European Association for Cardiovascular Prevention and Rehabilitation. Eur J Heart Fail 2011; 13: 347–357. DOI:10.1093/eurjhf/hfr017.
- Keteyian SJ, Piña IL, Hibner BA, Fleg JL. Clinical role of exercise training in the management of patients with chronic heart failure. J Cardiopulm Rehabil Prev 2010; 30: 67–76. DOI:10.1097/HCR.0b013e3181d0c1c1.
- Laoutaris ID, Adamopoulos S, Manginas A, Panagiotakos DB, Kallistratos MS, Doulaptsis C, et al. Benefits of combined aerobic/resistance/inspiratory training in patients with chronic heart failure. A complete exercise model? A prospective randomised study. Int J Cardiol 2013; 167: 1967–1972. DOI:10.1016/j.ijcard.2012.05.019.
- Davies EJ, Moxham T, Rees K, Singh S, Coats AJ, Ebrahim S, et al. Exercise training for systolic heart failure: Cochrane systematic review and meta-analysis. Eur J Heart Fail 2010; 12: 706–715. DOI:10.1093/eurjhf/hfq056.
- Zhang ZY, Sun XG, Xi JN, Liu YL, Feng J, Jiang LQ, et al. Effects of exercise intensity deduced from cardiopulmonary exercise testing on exercise rehabilitation efficacy among patients with chronic heart failure: a clinical study. Chin Gen Prac 2016; 19: 4302–4309. DOI: 10.3969/j.issn.1007-9572.2016.35.004.
- Whellan DJ, O’Connor CM, Lee KL, Keteyian SJ, Cooper LS, Ellis SJ, et al. Heart failure and a controlled trial investigating outcomes of exercise training (HF-ACTION): design and rationale. Am Heart J 2007; 153: 201–211. DOI: 10.1016/j.ahj.2006.11.007.
- Selig SE, Levinger I, Williams AD, Smart N, Holland DJ, Maiorana A, et al. Exercise & Sports Science Australia Position Statement on exercise training and chronic heart failure. J Sci Med Sport 2010; 13: 288–294. DOI: 10.1016/j.jsams.2010.01.004.
- Taylor JL, Bonikowske AR, Olson TP. Optimizing outcomes in cardiac rehabilitation: the importance of exercise intensity. Front Cardiovasc Med 2021; 8: 734278. DOI:10.3389/fcvm.2021.734278.
- Goncalves C, Raimundo A, Abreu A, Bravo J. Exercise intensity in patients with cardiovascular diseases: systematic review with meta-analysis. Int J Environ Res Public Health 2021; 18: 3574. DOI:10.3390/ijerph18073574.
- Bai Y, Hua B, Zhang F, Zhou W, Deng B. Effect of different intensity exercises intervention on cardiovascular functions and quality of life on patients with chronic heart failure: a protocol for systematic review and meta-analysis. Medicine (Baltimore) 2022; 101: e28554. DOI: 10.1097/MD.0000000000028554.
- Tabet JY, Meurin P, Benzidi Y, Beauvais F, Ben Driss A, Weber H, et al. Greater prognostic value of peak VO2 after exercise training program completion in heart failure patients. Int J Cardiol 2013; 168: 4139–4144. DOI: 10.1016/j.ijcard.2013.07.076.
- Xueyu L, Hao Y, Shunlin X, Rongbin L, Yuan G. Effects of low-intensity exercise in older adults with chronic heart failure during the transitional period from hospital to home in China: a randomized controlled trial. Res Gerontol Nurs 2017; 10: 121–128. DOI: 10.3928/19404921-20170411-02.
- Zhang ZY, Sun XG, Xi JN,Ge WG, Li H, Liu YL et al. The role of cardiopulmonary exercise testing in the formulation of high-intensity individualized rehabilitation exercise prescription and exercise rehabilitation effects evaluation among patients with chronic heart failure. Chin Gen Prac 2016; 19: 2061–2067. DOI:10.3969/j.issn.1007-9572.2016.17.015.
- Cardiovascular Branch of Chinese Medical Assosiation. Chinese Guidelines for the diagnosis and treatment of heart Failure. Chin J Cardiol 2018; 46: 760–789. DOI: 10.3760/cma.j.issn.0253-3758.2018.10.004.
- Wasserman K, Hansen JE, Sue DY, Striger WW, Sietsema KE, Sun XG, et al. Principles of exercise testing and interpretation – including pathophysiology and clinical applications. 5th edition. Philadelphia Lippincott Williams & Wilkins; 2012.
- Sun XG, Hansen JE, Stringer WW. Oxygen uptake efficiency plateau best predicts early death in heart failure. Chest 2012; 141: 1284–1294. DOI: 10.1378/chest.11-1270.
- Sun XG. The new 9 panels display of data from cardiopulmonary exercise test, emphasizing holistic integrative multi-systemic functions. Chin J Appl Phys 2015; 31: 369–373. DOI: 10.13459/j.cnki.cjap.2015.04.018.
- Forman DE, Fleg JL, Kitzman DW, Brawner CA, Swank AM, McKelvie RS, et al. 6-min walk test provides prognostic utility comparable to cardiopulmonary exercise testing in ambulatory outpatients with systolic heart failure. J Am Coll Cardiol 2012; 60: 2653–2661. DOI: 10.1016/j.jacc.2012.08.1010.
- Belardinelli R, Georgiou D, Cianci G, Purcaro A. Randomized, controlled trial of long-term moderate exercise training in chronic heart failure: effects on functional capacity, quality of life, and clinical outcome. Circulation 1999; 99: 1173–1182. DOI: 10.1161/01.cir.99.9.1173.
- Giuliano C, Levinger I, Vogrin S, Neil CJ, Allen JD. PRIME-HF: novel exercise for older patients with heart failure. a pilot randomized controlled study. J Am Geriatr Soc 2020; 68: 1954–1961. DOI: 10.1111/jgs.16428.
- Guo Y, Xiao C, Zhao K, He Z, Liu S, Wu X, et al. Physical exercise modalities for the management of heart failure with preserved ejection fraction: a systematic review and meta-analysis. J Cardiovasc Pharmacol 2022; 79: 698–710. DOI: 10.1097/FJC.0000000000001254.
- Professional Committee of Cardiovascular Prevention and Rehabilitation of Chinese Rehabilitation Medical Association. Expert consensus on cardiac rehabilitation for chronic heart failure in China. Chin J Intern Med 2020; 59: 942–952. DOI: 10.3760/cma.j.cn112138-20200309-00210
- Allen JD, Vanbruggen MD, Johannsen NM, Robbins JL, Credeur DP, Pieper CF, et al. PRIME: A novel low-mass, high-repetition approach to improve function in older adults. Med Sci Sports Exerc 2018; 50: 1005–1014. DOI: 10.1249/MSS.0000000000001518.
- Allen JD, Robbins JL, Vanbruggen MD, Credeur DP, Johannsen NM, Earnest CP, et al. Unlocking the barriers to improved functional capacity in the elderly: rationale and design for the “Fit for Life trial”. Contemp Clin Trials 2013; 36: 266–275. DOI: 10.1016/j.cct.2013.07.007.
- Shen T, Liu X, Zhuang B, Luo Q, Jin Y, Li G, et al. Efficacy and safety of different aerobic exercise intensities in patients with heart failure with reduced ejection fraction: design of a multicenter randomized controlled trial (HF-EI Trial). Front Cardiovasc Med 2021; 8: 705972. DOI: 10.3389/fcvm.2021.705972.
- Atchley AE, Kitzman DW, Whellan DJ, Iskandrian AE, Ellis SJ, Pagnanelli RA, et al. Myocardial perfusion, function, and dyssynchrony in patients with heart failure: baseline results from the single-photon emission computed tomography imaging ancillary study of the Heart Failure and A Controlled Trial Investigating Outcomes of Exercise TraiNing (HF-ACTION) Trial. Am Heart J 2009; 158: S53–S63. DOI: 10.1016/j.ahj.2009.07.009.
- Pearson MJ, Smart NA. Effect of exercise training on endothelial function in heart failure patients: a systematic review meta-analysis. Int J Cardiol 2017; 231: 234–243. DOI: 10.1016/j.ijcard.2016.12.145.
- Chen YM, Li ZB, Zhu M, Cao YM. Effects of exercise training on left ventricular remodelling in heart failure patients: an updated meta-analysis of randomised controlled trials. Int J Clin Pract 2012; 66: 782–791. DOI: 10.1111/j.1742-1241.2012.02942.x.
- Belardinelli R, Georgiou D, Ginzton L, Cianci G, Purcaro A. Effects of moderate exercise training on thallium uptake and contractile response to low-dose dobutamine of dysfunctional myocardium in patients with ischemic cardiomyopathy. Circulation 1998; 97: 553–561. DOI: 10.1161/01.cir.97.6.553.
- Hirai DM, Musch TI, Poole DC. Exercise training in chronic heart failure: improving skeletal muscle O2 transport and utilization. Am J Physiol Heart Circ Physiol 2015; 309: H1419–H1439. DOI: 10.1152/ajpheart.00469.2015.
- Besnier F, Labrunée M, Pathak A, Pavy-Le Traon A, Galès C, Sénard JM, et al. Exercise training-induced modification in autonomic nervous system: an update for cardiac patients. Ann Phys Rehabil Med 2017; 60: 27–35. DOI: 10.1016/j.rehab.2016.07.002.
- Tabet JY, Meurin P, Driss AB, Weber H, Renaud N, Grosdemouge A, et al. Benefits of exercise training in chronic heart failure. Arch Cardiovasc Dis 2009; 102: 721–730. DOI: 10.1016/j.acvd.2009.05.011.
- Nakanishi M, Nakao K, Kumasaka L, Arakawa T, Fukui S, Ohara T, et al. Improvement in exercise capacity by exercise training associated with favorable clinical outcomes in advanced heart failure with high B-type natriuretic peptide level. Circ J 2017; 81: 1307–1314. DOI: 10.1253/circj.CJ-16-1268.
- Guimaraes GV, d’Avila VM, Silva MS, Ferreira SA, Ciolac EG, Carvalho VO, et al. A cutoff point for peak oxygen consumption in the prognosis of heart failure patients with β-blocker therapy. Int J Cardiol 2010; 145: 75–77. DOI: 10.1016/j.ijcard.2009.05.001.
- Yabe H, Kono K, Onoyama A, Kiyota A, Moriyama Y, Okada K, et al. Predicting a target exercise heart rate that reflects the anaerobic threshold in nonbeta-blocked hemodialysis patients: the Karvonen and heart rate reserve formulas. Ther Apher Dial 2021; 25: 884–889. DOI: 10.1111/1744-9987.13628.
- Sun X. Prospective and value of cardiopulmonary exercise testing in clinical cardiovascular medicine. Chin J Cardiol 2014; 42: 347–351. DOI: 10.3760/cma.j.issn.0253-3758.2014.04.018
- Shafiq A, Brawner CA, Aldred HA, Lewis B, Williams CT, Tita C, et al. Prognostic value of cardiopulmonary exercise testing in heart failure with preserved ejection fraction. The Henry Ford HospITal CardioPulmonary EXercise Testing (FIT-CPX) project. Am Heart J 2016; 174: 167–172. DOI: 10.1016/j.ahj.2015.12.020.
- Lala A, Shah KB, Lanfear DE, Thibodeau JT, Palardy M, Ambardekar AV, et al. Predictive value of cardiopulmonary exercise testing parameters in ambulatory advanced heart failure. JACC Heart Fail 2021; 9: 226–236. DOI: 10.1016/j.jchf.2020.11.008.