ORIGINAL REPORT
EFFECTS OF KINESIO TAPING ON STATIC BALANCE PERFORMANCE AND MUSCLE ACTIVITY IN CHILDREN WITH DEVELOPMENTAL COORDINATION DISORDER: A SINGLE-GROUP PRETEST-POSTTEST STUDY
Li-Liang LI, MSc1 and Fu-Chen CHEN, PhD2
From the 1Physical Education and Health Center, National Kaohsiung University of Hospitality and Tourism and 2Department of Physical Education, National Kaohsiung Normal University, Kaohsiung, Taiwan
Objective: To compare the effects of various kinesio tape applications on static balance and muscle activity in children with developmental coordination disorder.
Methods: Four taping conditions were applied to 48 children with developmental coordination disorder: no taping, gastrocnemius taping, tibialis anterior taping; and peroneus longus taping. Postural sway and electromyographic data were assessed, with eyes closed (30 s), standing still in 2-leg stance, dominant-leg stance, and non-dominant-leg stance.
Results: Kinesio taping significantly reduced postural sway in both anteroposterior and mediolateral directions for dominant-leg stance and non-dominant-leg stance, but not 2-leg stance. During single-leg stances, anteroposterior sway was significantly lower for the gastrocnemius taping condition than for the no taping, tibialis anterior and peroneus longus taping conditions, and significantly lower in the tibialis anterior and peroneus longus taping conditions than in the no taping condition (gastrocnemius < tibialis anterior = peroneus longus < no taping). In addition, mediolateral sway was significantly lower in the tibialis anterior and peroneus longus taping conditions than in the no taping and gastrocnemius taping conditions, and significantly lower in the gastrocnemius taping condition than in the no taping condition (tibialis anterior = peroneus longus < gastrocnemius < no taping). Electromyographic data showed that muscle activity was significantly greater only for muscles where kinesio tape was applied.
Conclusion: Various kinesio tape applications can differentially reduce postural sway and increase muscle activity during single-leg stances in children with developmental coordination disorder.
LAY ABSTRACT
This is the first study to compare the effects of various kinesio tape applications on static balance performance and muscle activity in children with developmental coordination disorder. The following novel results were found: (i) kinesio taping can effectively enhance static balance performance in a single-leg stance; (ii) for anteroposterior sway, kinesio tape applied to the gastrocnemius is the most effective in enhancing balance; (iii) for mediolateral sway, kinesio tape applied to both the tibialis anterior and peroneus longus is the most effective in enhancing balance; and (iv) muscle activity was greater only for muscles where kinesio tape was applied. These findings establish the potential beneficial applications of kinesio tape for improving static balance performance and muscle activity in children with developmental coordination disorder.
Key words: kinesio tape; static balance; electromyography; developmental coordination disorder.
Citation: J Rehabil Med 2023; 55: jrm13403. DOI: https://doi.org/10.2340/jrm.v55.13403.
Copyright: © Published by Medical Journals Sweden, on behalf of the Foundation for Rehabilitation Information. This is an Open Access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/)
Accepted: Aug 1, 2023; Published: Aug 25, 2023
Correspondence address: Fu-Chen Chen, Department of Physical Education, National Kaohsiung Normal University, 116, Heping 1st Rd, Lingya District, Kaohsiung 802, Taiwan. E-mail: fcchen@mail.nknu.edu.tw
Competing interests and funding: The authors have no conflicts of interest to declare.
Developmental coordination disorder (DCD) refers to a neuro-developmental condition characterized by impaired motor coordination skills that negatively affects performance of activities of daily living (e.g. hygiene, dressing, bathing, eating and toileting) and academic achievements (e.g. writing, reading, painting, exercising and sport and leisure activities). The estimated global prevalence of DCD in school-aged children is approximately 5–6% (1). The core feature of children with DCD is difficulties in various motor skills, including manual dexterity, throwing and catching and balance. Among these difficulties, balance deficit is one of the most prevalent problems, affecting 73–87% of children with DCD (2). Therefore, assessing static balance was a key factor in this study.
Numerous studies have demonstrated that children with DCD present deficits in static balance performance during upright stance. Static balance assessment, using a force platform to measure the displacement of the centre of pressure (COP), showed a significantly greater amount of postural sway in children with DCD compared with typically developing children (TDC) under various test conditions, including 2-leg and single-leg standing with eyes either open or closed (3, 4). In addition, postural sway in children with DCD was more pronounced under more challenging conditions, such as standing on 1 leg or standing with the eyes closed. More specifically, children with DCD demonstrate weaker control over the lower limb muscles, such as tibialis anterior and peroneus longus, indicating reduced modulation of postural sway while performing static balance tasks (5). In summary, previous studies suggest that static balance is impaired in children with DCD, and that the deficits in static balance performance may be attributed to altered lower limb muscle activity.
In recent years, kinesio tape (KT) has gained popularity and is commonly utilized in clinical practice. KT is theorized to restore correct muscle function through the stimulation imposed by the tape on the skin (6). The underlying mechanism may be related to afferent inputs from the cutaneous mechanoreceptors to the taped muscles, leading to an increase in the motor unit firing rate (7) and increased muscle activity (8, 9). Hence, KT may have potential as an intervention for mediating balance deficits via modulation of muscle activity. Many studies have evaluated the effects of KT on static balance performance in populations with neuromuscular diseases/disorders. Research demonstrates that applying KT can enhance static balance performance for patients who have experienced a stroke, as measured by the Berg Balance Scale (10), the sensory organization test (11), and force platforms (12, 13). In addition, in a study on patients with multiple sclerosis, KT application reduced postural sway (14). More recently, Tamburella et al. (15) compared postural sway during 2-leg standing to examine the difference between KT and conventional non-elastic taping in patients with spinal cord injuries. The results showed significantly lower postural sway after application of KT compared with conventional taping, suggesting that the effects of KT are superior to conventional taping. Collectively, existing literature has generated consistent findings supporting KT as a viable option for improving static balance performance in special populations.
Finally, to the best of our knowledge, only 1 study has tested the effects of KT on balance for children with DCD. Yam et al. (16) utilized the Lower Quarter Y-balance test (YBT-LQ) and electromyography (EMG) to measure the maximal reaching distance of the raised leg (while maintaining balance on the other leg) and muscle activity in the standing leg. In the experimental group, DCD participants received KT application concurrently on both gastrocnemius and rectus femoris muscles. For the control group, DCD participants received no taping intervention. They found that both performance of the YBT-LQ and muscle activity increased in the KT experimental group relative to the no taping control group. However, YBT-LQ is a tool to assess dynamic rather than static balance. More importantly, Yam et al.’s study was designed to investigate the effect of a single KT application condition vs a no taping control. That is, whether various KT applications can differentially influence static balance performance and muscle activity remains unclear. Therefore, the aims of this study were to determine the effects of various KT applications on static balance performance and muscle activity in the lower extremity muscles for children with DCD.
Previous studies have demonstrated that lower extremity muscle activities are impacted during the execution of static balance tasks in children with DCD (5, 16). In addition, correlations between postural sway during static balancing and EMG activity of the gastrocnemius, tibialis anterior and peroneus longus muscles have been established and reported (e.g. 17, 18). These correlations indicate the importance of these muscles in static balance performance for children with DCD. Accordingly, the gastrocnemius, tibialis and peroneus longus were selected as target muscles for taping and electrography in this study. It was hypothesized that the use of KT can alter static balance performance and muscle activity, and that the various KT applications can differentially influence static balance performance and lower extremity muscle activity in children with DCD.
METHODS
Study design
This was a quasi-experimental single-group pretest-posttest study. A double-blind, within-subject, crossover design was used. The study was approved by the local ethics committee of Antai Tian-Sheng Memorial Hospital (institutional review board number 19-118-A). All participants and their parents provided written informed consent before beginning the study. The experimental procedures used for this study conformed to the Declaration of Helsinki.
Participants
A total of 519 children, age range 10–12 years, were recruited using convenience sampling screened from 6 elementary schools in Kaohsiung, Taiwan. Among them, 48 subjects (26 girls and 22 boys; mean ± standard deviation (SD) height 147.67 ± 9.07 cm; mean ± SD weight 46.79 ± 7.58 kg) took part in this study. Inclusion criteria for children with DCD included scoring below the 5th percentile on total score and balance sub-score as well as scoring above the 95th percentile on checklist score from the 2nd edition of the Movement Assessment Battery for Children (MABC-2) (19). A further inclusion criteria was that participants had to score above the 95th percentile on the MABC-2 checklist score. These criteria ensured that the participants had a sufficiently low level of motor coordination and balance performance, as well as difficulties in their daily living and/or school activities, to qualify for the study. To rule out intellectual and attentional deficits, participants were required to score above 80 on the 2nd edition of Kaufmann Brief Intelligence Test (20), and score below 70 on Conners’ Teacher Rating Scale (21). To exclude placebo effects (psychological beliefs in KT effects) (22), all participants had no knowledge of KT, which was ensured by showing them a roll of KT and asking participants the following questions: “Have you ever seen it?”, “Do you know what it is?”, and “Do you know how it’s used?”. Based on the parents’ reports, all participants had no known genetic diseases (e.g. Down syndrome or muscular dystrophy), neurological disorders (e.g. epilepsy or cerebral palsy), or severe musculoskeletal conditions (e.g. fracture, ligament rupture/sprain, or muscle tear) in the previous 6 months. All participants had normal or corrected-to-normal vision, and all were right foot dominant (defined as preferred foot for kicking a ball) (23). Fig. 1 is the flow diagram for participants’ recruitment. Table I summarizes the baseline data.

Fig. 1. Flow diagram for participants’ recruitment. MABC-2: 2nd edition of The Movement Assessment Battery for Children; KBIT-2: 2nd edition of The Kaufmann Brief Intelligence Test; CTRS: Conners’ Teacher Rating Scale.
Static balance assessment
The static balance assessment consisted of 3 stance conditions: 2-leg stance (TS), dominant-leg stance (DS), and non-dominant-leg stance (NS). Each participant performed 3 30-s trials of the 3 balance conditions (TS, DS and NS) with eyes closed in a counterbalanced order. For all balance conditions, participants stood barefoot on a force platform (Model 0R6-5-1, Advanced Mechanical Technology, Inc., Watertown, MA, USA) with data sampled at 100 Hz low-pass filtered with a second-order Butterworth filter (10 Hz). Toe and heel positions were marked on the platform for each participant, to ensure consistency in foot position and orientation among trials. For the TS condition, participants naturally stood with feet approximately shoulder-width apart and toes pointing forwards and slightly outwards. For DS and NS conditions, participants stood on the centre of the force platform with 1 leg, while the other leg was bent at approximately 90º. During static balance assessment trials, participants wore an eye mask and kept their eyes closed. Participants were asked to stand upright as still as possible for the entire duration of the trial with arms by their sides. Arm movements were not allowed during trials. A researcher always stood near participants to monitor their standing posture and arm movements throughout the trial, and to protect children from falling. Once participants achieved the required standing posture and stabilized their postural sway, the trial was initiated. If obvious arm movements were observed or children lifted or displaced the stance foot from the original position, the trial was discarded and repeated. Prior to the static balance assessment, participants were shown a demonstration by the experimenter and asked to perform a practice trial to familiarize themselves with the test procedures. More practice trials were permitted, as needed. If the participants were incapable of standing on 1 leg for 30 s, the trial was stopped and discarded. The trial was then repeated after a sufficient rest period. Participants were asked to repeat the static balance assessments until 3 successful trials were obtained for each balance condition. The mean ± standard deviation (SD) of the total trial number to obtain 3 successful trials were 3.04 ± 0.20, 3.77 ± 0.86 and 3.83 ± 0.91 for the TS, DS and NS conditions, respectively. The static balance assessment took approximately 10–15 min for each participant.
Muscle activity
During the static balance assessment, surface EMG signals were collected using a wireless EMG acquisition system (Ultium™ Biomechanics System, Noraxon Ltd, Scottsdale AZ, USA). Prior to attaching the electrodes, skin impedance was reduced by shaving leg hair (if necessary) and wiping with alcohol to ensure an impedance less than 15 kOhms. The EMG electrodes were placed on the belly of the gastrocnemius, tibialis anterior, and peroneus longus muscles according to the Surface EMG for Non-Invasive Assessment of Muscles (SENIAS) guidelines (24) on both legs. To normalize muscle activity, participants completed 2 repetitions of 5-s maximal voluntary isometric contractions (MVIC) trials with 1 min relaxation between trials. The MVIC trials were conducted for gastrocnemius, tibialis anterior and peroneus longus muscles based on manual testing methods (25). The raw EMG signal was sampled at 1,000 Hz, amplified by a factor of 1,000, and bandpass filtered from 20 to 460 Hz. Root mean square (RMS) EMG were calculated for every 200-ms epoch during the MVIC test for each muscle. The maximal RMS value was then extracted and averaged across 2 MVIC trials, and used for data normalization.
Kinesio tape applications
KT was applied to participants according to kinesio taping procedure in the paediatrics manual (26). To ensure tape tension remained at 50%, KT length was determined using the following formula (16, 27):
y = {[(x − 4) ÷ 1.5] + 4} × 1.10
where y is the actual tape length and x is the measured distance between origin and insertion of each muscle. For all taping conditions, children were seated on a height-adjustable treatment table and KT was applied to bilateral target muscles in stretched positions. The proximal and distal end of the KT (length 2 cm at each end) was free of tension and the middle portion was applied with 50% tension. For the gastrocnemius (GN) taping condition, 2 I-shaped KT bands were applied starting from the medial and lateral condyles of the femur and ending at the base of the calcaneus (Fig. 2a). For the tibialis anterior (TA) taping condition, an I-shaped KT band was applied starting from the lateral condyle of the tibia, placed along the upper two-thirds of lateral tibia, and ending at the plantar surface of the medial cuneiform and the first metatarsal bone (Fig. 2b). For the peroneus longus (PL) taping condition, an I-shaped KT was applied starting from the head of the fibula, passing behind the lateral malleolus, and ending at the plantar surface of the medial cuneiform and the first metatarsal base (Fig. 2c).
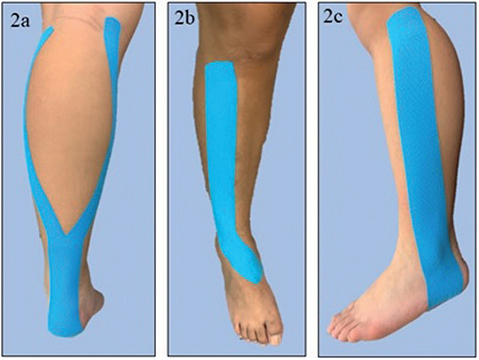
Fig. 2. Kinesio tape application on: (a) gastrocnemius, (b) tibialis anterior, and (c) peroneus longus.
Experimental procedures
Participants performed the static balance assessment under the 4 taping conditions: no taping (NT), GN, TA and PL, during 4 different experimental sessions separated by 7 days. Each taping condition was blocked in an experimental session and the order of experimental sessions was counterbalanced across participants. Prior to commencing experiments, participants received a detailed explanation about the experimental procedures. First, participants’ skin surface was well prepared and EMG electrodes were attached to the target muscles (gastrocnemius, tibialis anterior and peroneus longus). Afterwards, for all experiments except the NT condition, 1 of the target muscles was taped by an experienced athletic trainer. Deception was used to eliminate potential placebo effects, by informing participants that muscle sensors (rather than KT) were applied on the skin to examine circulation. Participants then performed the static balance assessment. This process was repeated until all experimental sessions were completed.
Statistical analysis
Dependent variables were postural sway (quantified as standard deviation of COP displacement) in the body’s anteroposterior (AP) and mediolateral (ML) direction, as well as normalized RMS EMG data for gastrocnemius, tibialis anterior and peroneus longus muscles. Separate 1-way repeated-measures analyses of variance (ANOVAs) with the taping condition (4 levels: NT, GN, TA and PL) as a within-subject factor were used to analyse the effects of KT application on static balance performance and muscle activity. The degrees of freedom were corrected with Greenhouse-Geisser epsilon values, where applicable. Significant effects were further analysed using Bonferroni post-hoc comparisons. Effect sizes in ANOVA were reported as partial eta squared (ηp2). Significance was set at p < 0.05. Analyses were conducted using SPSS 24.0 (IBM Corp., Armonk, NY, USA).
RESULTS
Detailed postural sway and EMG data are shown in Table II.
Postural sway data
Fig. 3 shows postural sway in the AP direction for different stance conditions (TS, DS and NS) under 4 taping conditions (NT, GN, TA and PL). For AP sway, there were significant main effects of taping condition in the DS (F (1.94, 141) = 74.90, p < 0.05, ηp2 = 0.61) and NS (F (3, 141) = 113.47, p < 0.05, ηp2 = 0.70) conditions, but not the TS condition. (F (2.24, 141) = 0.97, p = 0.39). Further analyses by t-test with Bonferroni corrections showed that, for both the DS and NS conditions, AP sway was significantly lower in the GN taping condition compared with NT, TA and PL conditions (ps < 0.05), while AP sway was significantly lower in both the TA and PL taping conditions compared with the NT taping condition (ps < 0.05) (GN < TA = PL < NT).

Fig. 3. Postural sway in the anteroposterior (AP) direction for different stance conditions (2-leg stance (TS), dominant-leg stance (DS) and non-dominant-leg stance (NS)) under 4 taping conditions (NT: no taping; GN: gastrocnemius taping condition; TA: tibialis anterior taping condition; PL: peroneus longus taping condition). Bars: standard errors. *Main effects of taping conditions.
Fig. 4 shows postural sway in the ML direction for different stance conditions (TS, DS and NS) under 4 taping conditions (NT, GN, TA and PL). For ML sway, there were significant main effects of taping condition in the DS (F (1.70, 141) = 21.20, p < 0.05, ηp2 = 0.31) and NS (F (1.84, 141) = 23.78, p < 0.05, ηp2 = 0.34) conditions, but not the TS condition. (F (2.26, 141) = 0.76, p = 0.49). Further analyses by t-test with Bonferroni corrections showed that, for both the DS and NS conditions, ML sway was significantly lower in both TA and PL taping conditions compared with NT and GN taping conditions (ps < 0.01), while ML sway was significantly lower in the GN than NT condition (p < 0.01) (TA = PL < GN < NT).

Fig. 4. Postural sway in the mediolateral (ML) direction for different stance conditions (TS: 2-leg stance; DS: dominant-leg stance; NS: non-dominant-leg stance) under 4 taping conditions (NT: no taping; GN: gastrocnemius taping condition; TA: tibialis anterior taping condition; PL: peroneus longus taping condition). Bars: standard errors. *Main effects of taping conditions.
Electromyographic data
Fig. 5 shows normalized RMS EMG in gastrocnemius muscle for different stance conditions (TS, DS and NS) under 4 taping conditions (NT, GN, TA and PL). For gastrocnemius muscle EMG data, there were significant main effects of taping condition in the DS (F (2.34, 141) = 18.07, p < 0.05, ηp2 = 0.28) and NS (F (1.19, 141) = 5.05, p < 0.05, ηp2 = 0.10) conditions, but not the TS condition. (F (1.10, 141) = 1.10, p = 0.31). Further analyses by t-test with Bonferroni corrections showed that, for both the DS and NS conditions, the normalized RMS EMG of gastrocnemius muscle was significantly greater in the GN taping condition than in the NT, TA and PL conditions (ps < 0.01) (GN > NT = TA = PL).
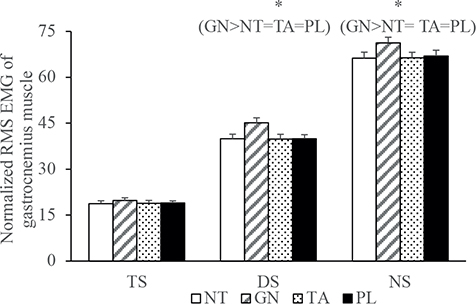
Fig. 5. Normalized root mean squares (RMS) electromyography in gastrocnemius muscle for different stance conditions (TS: 2-leg stance; DS: dominant-leg stance; NS: non-dominant-leg stance) under 4 taping conditions (NT: no taping; GN: gastrocnemius taping condition; TA: tibialis anterior taping condition; PL: peroneus longus taping condition). Bars: standard errors. *Main effects of taping conditions.
Fig. 6 shows normalized RMS EMG in tibialis anterior muscle for different stance conditions (TS, DS and NS) under 4 taping conditions (NT, GN, TA and PL). For tibialis anterior muscle EMG data, there were significant main effects of taping condition in the DS (F (2.23, 141) = 4.27, p < 0.05, ηp2 = 0.08) and NS (F (1.83, 141) = 8.27, p < 0.05, ηp2 = 0.15) conditions, but not the TS condition. (F (1.42, 141) = 1.48, p = 0.24). Further analyses by t-test with Bonferroni corrections showed that, for both the DS and NS conditions, the normalized RMS EMG of tibialis anterior muscles was significantly greater in the TA taping condition than in the NT, GN and PL conditions (ps < 0.01) (TA > NT = GN = PL).

Fig. 6. Normalized root mean squares (RMS) electromyography in tibialis anterior muscle for different stance conditions (TS: 2-leg stance; DS: dominant-leg stance; NS: non-dominant-leg stance) under 4 taping conditions (NT: no taping; GN: gastrocnemius taping condition; TA: tibialis anterior taping condition; PL: peroneus longus taping condition). Bars: standard errors. *Main effects of taping conditions.
Fig. 7 shows normalized RMS EMG in peroneus longus muscle for different stance conditions (TS, DS and NS) under 4 taping conditions (NT, GN, TA and PL). For peroneus longus muscle EMG data, there were significant main effects of taping condition in the DS (F (2.29, 141) = 4.30, p < 0.05,ηp2 = 0.08) and NS (F (1.83, 141) = 7.09, p < 0.05, ηp2 = 0.13) conditions, but not the TS condition. (F (1.68, 141) = 2.37, p = 0.11). Further analyses by t-test with Bonferroni corrections showed that, for both the DS and NS conditions, the normalized RMS EMG of peroneus longus muscle was significantly greater in the PL taping condition than in the NT, GN and TA conditions (ps < 0.01) (PL > NT = GN = TA).

Fig. 7. Normalized root mean squares (RMS) electromyography in peroneus longus muscle for different stance conditions (TS: 2-leg stance; DS: dominant-leg stance; NS: non-dominant-leg stance) under 4 taping conditions (NT: no taping; GN: gastrocnemius taping condition; TA: tibialis anterior taping condition; PL: peroneus longus taping condition). Bars: standard errors. *Main effects of taping conditions.
DISCUSSION
This study examined how KT applied to different lower extremity muscles can alter static balance performance and muscle activity for children with DCD. The results extend previous work by demonstrating the following: (i) KT applications can reduce postural sway in the single-leg stance only (i.e. the DS and NS conditions); (ii) for AP sway, the GN taping condition showed lower sway compared with NT, TA and PL taping conditions, and the TA and PL taping conditions showed lower sway than the NT condition; (iii) for ML sway, both TA and PL taping conditions showed lower sway compared with NT and GN taping conditions and the GN taping condition showed lower ML sway than the NT condition; and (iv) muscle activity was significantly greater only for muscles where KT was applied. Possible explanations underlying the effects of KT application in children with DCD are discussed below.
KT application reduced postural sway in the single-leg stance, but not in the TS, for children with DCD. This may be due to a floor effect of postural sway in the TS condition. Being already low in postural sway amplitude (approximately 2–3 cm in AP and ML sway, separately), the TS condition showed less “space” to decrease postural sway, resulting in interaction effects in which children with DCD showed reduced AP and ML sway with KT applications for both DS and NS conditions that were not observed in the TS condition. It is notable that the current study showed directional specificity of KT effects on postural sway. For AP sway, postural sway was lowest for the GN taping condition, followed by TA and PL taping conditions, and was highest for the NT condition. In contrast, for ML sway, postural stability was lowest for TA and PL taping conditions, followed by GN then NT conditions. These results may reflect the fact that the gastrocnemius is a primary actuator responsible for controlling AP sway and the peroneus longus and tibialis anterior are main actuators involved in controlling ML sway. Previous studies have indicated that the gastrocnemius was the primary contributor to modulating AP sway, particularly for challenging balancing tasks (28, 29). In addition, it has been shown that the peroneus longus plays a key role in adjusting ML sway (30). Finally, research shows that the tibialis anterior is increasingly activated, as lateral tibial displacement is being controlled in closed kinetic chain positions (31). Accordingly, the current study provides robust evidence of clinical applications for KT when addressing static balance deficits in the body’s AP and ML directions.
A possible explanation for why KT can effectively improve static balance performance is related to the acute effects of KT on changing muscle activity in the muscles to which KT is applied. The current study found improvements in static balance performance and increases in normalized RMS EMG following KT application. These results support a previous study showing positive KT effects on dynamic balance performance and muscle activity for children with DCD (16). This effect could be due to KT activating Ia fibres by tactile stimulation through the stretched skin (32), which, in turn, facilitates gamma motor neurone activity (8). Studies demonstrate that EMG amplitude captures information on both motor unit recruitment and firing rate, providing insight into skeletal muscle function (33, 34). Higher EMG amplitudes can indicate that additional motor neurones are being recruited or those recruited are firing at a faster rate. The current results suggest that KT can effectively facilitate and exert greater muscle activity, thereby improving static balance performance.
Another possible explanation is that KT may promote greater muscle force. Previous studies have reported lower muscle force/strength in children with DCD (35). Fong et al. (36) further pointed out that muscle peak force in lower extremities is associated with balance performance, accounting for 5.7% of variance in the MABC-2 balance sub-score in children with DCD. Therefore, muscle force in lower extremities seems crucial for regulating postural sway. Furthermore, extended research shows that increased muscle force resulting from intervention can lead to better balance performance for populations with lower muscular force/strength, such as older adults (37), and people with Parkinson’s disease (38). As EMG amplitude correlated positively with muscle force (39), the observed increase in EMG amplitude immediately following KT application could directly promote muscle force in lower extremities, which can enhance static balance performance in children with DCD. Nevertheless, the current study did not evaluate muscle force and leaves this explanation open to further testing. Further research will determine whether the effects observed for static balance performance are due to the direct influence of KT on muscle force.
Finally, changes in proprioception could influence the positive effects of KT on static balance performance. Proprioceptive feedback is one of the primary sources of information for maintaining balance during an upright stance (40). In a study assessing lower extremity proprioception in children with DCD, children with DCD showed a less accurate joint position sense for knee and ankle joints compared with TDC (41). Thus, proprioception in the lower extremity joints in children with DCD was compromised. Furthermore, studies have shown the beneficial effects of KT on proprioception and suggest that KT can provide mechanical pressure and/or stretching to the skin, which can stimulate cutaneous mechanoreceptors and signal information about joint position or movement (42). This result suggests that improvements in static balance performance for children with DCD could be, at least in part, ascribed to additional proprioceptive afferent feedback supplied by KT application.
Study limitations
This study recruited participants with DCD with a very narrow age band (10–12 years) and, thus caution is needed in the generalization of any positive KT effects on both static balance performance and muscle activity regarding age. Furthermore, static balance performance was evaluated only by maintaining an upright stance on a stable surface. There are many static balance assessments, such as maintaining stability on an unstable surface (e.g. foam or wobble board), as well as in the presence of unpredicted disturbances (e.g. being bumped) and environmental interference (noise or optical flow). Therefore, the current results should not be generalized to other static balance scenarios. Notably, the study did not examine sustained effects after removal of KT. Researchers have suggested that the effects of KT are long-lasting, with effects present from 24 h to 2–3 days after ceasing KT intervention (43). Therefore, the sustainable long-term effectiveness of KT are unknown for children with DCD. Lastly, further research is necessary to determine how long KT should remain in place in order to produce long-term effects.
CONCLUSION
To summarize, KT applications are effective in improving static balance performance during single-leg stance children with DCD. Postural sway in the AP direction was lowest for the GN taping condition, intermediate for the TA and PL conditions, and highest for the NT condition. In addition, postural sway in the ML direction was lowest for the TA and PL taping conditions, intermediate for the GN condition, and highest for the NT condition. Lastly, only EMG activity of the target muscle to which KT was applied increased immediately following KT application. These results establish clinical standard for the use of KT to optimize static balance performance, as well as to facilitate muscle activity for children with DCD.
ACKNOWLEDGEMENT
This work was supported by National Science and Technology Council, Taiwan (109-2410-H-017 -018 -MY2). The author certifies that the submission is original work, and has not been presented as an abstract/poster/oral or as a full article in any national or international conference.
REFERENCES
- American Psychiatric Association Diagnostic and Statistical Manual of Mental Disorders (DSM-5). 5th edn. Arlington, Virginia (VA), USA. American Psychiatric Association; 2013. DOI: 10.1176/appi.books.9780890425596
- Macnab JJ, Miller LT, Polatajko HJ. The search for subtypes of DCD: is cluster analysis the answer? Hum Mov Sci 2001; 20: 49–72. DOI: 10.1016/S0167-9457(01)00028-8
- Mitsiou M, Giagazoglou P, Sidiropoulou M, Kotsikas G, Tsimaras V, Fotiadou E. Static balance ability in children with developmental coordination disorder. Eur J Phys Educ Sport 2016; 11: 17–23.
- Tsai CL, Wu SK, Huang CH. Static balance in children with developmental coordination disorder. Hum Mov Sci 2008; 27: 152–153. DOI: 10.1016/j.humov.2007.08.002
- Geuze RH. Postural control in children with developmental coordination disorder. Neural Plast 2005; 12: 183–196. DOI: 10.1155/NP.2005.183
- Kase K. Until today from birth of kinesio taping method. KMS. Abluquerque, New Mexico (NM), USA. 2001.
- Macgregor K, Gerlach S, Mellor R, Hodges PW. Cutaneous stimulation from patella tape causes a differential increase in vasti muscle activity in people with patellofemoral pain. J Orthop Res 2005; 23: 351–358. DOI: 10.1016/j.orthres.2004.07.006
- Konishi Y. Tactile stimulation with kinesiology tape alleviates muscle weakness attributable to attenuation of Ia afferents. J Sci Med Sport 2013; 16: 45–48. DOI: 10.1016/j.jsams.2012.04.007
- Fratocchi G, Di Mattia F, Rossi R, Mangone M, Santilli V, Paoloni M. Influence of Kinesio Taping applied over biceps brachii on isokinetic elbow peak torque. A placebo controlled study in a population of young healthy subjects. J Sci Med Sport 2013; 16: 245–249. DOI: 10.1016/j.jsams.2012.06.003
- Bae YH, Kim HG, Min KS, Lee SM. Effects of lower-leg kinesiology taping on balance ability in stroke patients with foot drop. Evid Based Complementary Altern Med 2015; 2015: 125629. DOI: 10.1155/2015/125629
- Yazici G, Guclu-Gunduz A, Bayraktar D, Aksoy S, Nazliel B, Kilinc M, et al. Does correcting position and increasing sensorial input of the foot and ankle with Kinesio Taping improve balance in stroke patients? Neurorehabilitation 2015; 36: 345–353. DOI: 10.3233/NRE-151223
- Rojhani-Shirazi Z, Amirian S, Meftahi N. Effects of ankle kinesio taping on postural control in stroke patients. J Stroke Cerebrovasc Dis 2015; 24: 2565–2571. DOI: 10.1016/j.jstrokecerebrovasdis.2015.07.008
- Shin YJ, Kim SM, Kim HS. Immediate effects of ankle eversion taping on dynamic and static balance of chronic stroke patients with foot drop. J Phys Ther Sci 2017; 29: 1029–1031. DOI: 10.1589/jpts.29.1029
- Cortesi M, Cattaneo D, Jonsdottir J. Effect of kinesio taping on standing balance in subjects with multiple sclerosis: a pilot study. NeuroRehabilitation 2011; 28: 365–372. DOI: 10.3233/NRE-2011-0665
- Tamburella F, Scivoletto G, Molinari M. Somatosensory inputs by application of KinesioTaping: effects on spasticity, balance, and gait in chronic spinal cord injury. Front Hum Neurosci 2014; 8: 367. DOI: 10.3389/fnhum.2014.00367
- Yam TTT, Or PPL, Ma AWW, Fong SSM, Wong MS. Effect of Kinesio taping on Y-balance test performance and the associated leg muscle activation patterns in children with developmental coordination disorder: a randomized controlled trial. Gait Posture 2019; 68: 388–396. DOI: 10.1016/j.gaitpost.2018.12.025
- Spiliopoulou SI, Amiridis IG, Tsigganos G, Hatzitaki V. Side-alternating vibration training for balance and ankle muscle strength in untrained women. J Athl Train 2013; 48: 590–600. DOI: 10.4085/1062-6050-48.4.03
- Mulavara AP, Verstraete MC, Simon BN. Correlation between muscle activity and the center of pressure. Conf Proc IEEE Eng Med Biol Soc 1994; 1: 384–385. DOI: 10.1109/IEMBS.1994.411992
- Henderson SE, Sugden DA, Barnett AL. (2007). Movement assessment battery for children. 2nd edn. London, UK. The Psychological Corporation; 2007. DOI: 10.1037/t55281-000
- Kaufman AS, Kaufman NL. KBIT-2 Kaufman Brief Intelligence Test. 2nd edn. Bloomington, Minnesota (MN), USA. NCS Pearson; 1997.
- Conners CK. Conners’ Rating Scale-Revised (CRS-R) technical manual. Toronto, Ontario (ON), CA. Multi-Health Systems; 2001.
- Mak DN, Au IP, Chan M, Chan ZY, An WW, Zhang JH, et al. Placebo effect of facilitatory Kinesio tape on muscle activity and muscle strength. Physiother Theory Pract 2019; 35: 157–162. DOI: 10.1080/09593985.2018.1441936
- van Melick N, Meddeler BM, Hoogeboom TJ, Nijhuis-van der Sanden MWG, van Cingel REH. How to determine leg dominance: the agreement between self-reported and observed performance in healthy adults. PLOS One 2018: 19, e0189876. DOI: 10.1371/journal.pone.0189876
- Hermens HJ, Freriks B, Disselhorst-Klug C, Rau G. Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol 2000; 10: 361–374. DOI: 10.1016/S1050-6411(00)00027-4
- Clarkson HM. Musculoskeletal assessment: joint range of motion and manual muscle strength. 2nd edn. Philadelphia Pennsylvania (PA), USA. Lippincott Williams & Wilkins; 2000.
- Kase K, Martin P, Yasukawa A. Kinesio Taping in pediatrics: fundamentals and whole body taping. Kinesio. Abluquerque, New Mexico (NM), USA. 2006.
- Yam TTT, Wong MS, Fong SSM. Effect of kinesio taping on electromyographic activity of leg muscles during gait in children with developmental coordination disorder: a randomized controlled trial. Medicine 2019; 98: e14423. DOI: 10.1097/MD.0000000000014423
- Borg F, Finell M, Hakala I, Herrala M. Analyzing gastrocnemius EMG-activity and sway data from quiet and perturbed standing. J Electromyogr Kinesiol 2007; 17: 622–634. DOI: 10.1016/j.jelekin.2006.06.004
- Vieira TM, Minetto MA, Hodson-Tole EF, Botter A. How much does the human medial gastrocnemius muscle contribute to ankle torques outside the sagittal plane? Hum Mov Sci 2013; 32: 753–767. DOI: 10.1016/j.humov.2013.03.003
- De Ridder R, Willems T, Vanrenterghem J, Roosen P. Influence of balance surface on ankle stabilizing muscle activity in subjects with chronic ankle instability. J Rehabil Med 2015; 47: 632–638. DOI: 10.2340/16501977-1970
- Pozzi F, Moffat M, Gutierrez G. Neuromuscular control during performance of a dynamic balance task in subjects with and without ankle instability. Int J Sports Phys Ther 2015; 10: 520–529.
- Bravi R, Cohen EJ, Quarta E, Martinelli A, Minciacchi D. Effect of direction and tension of kinesio taping application on sensorimotor coordination. Int J Sports Med 2016; 37: 909–914. DOI: 10.1055/s-0042-109777
- Beck TW, Housh TJ, Cramer JT, Weir JP, Johnson GO, Coburn JW, et al. Mechanomyographic amplitude and frequency responses during dynamic muscle actions: a comprehensive review. BioMed Eng OnLine 2005; 4: 67. DOI: 10.1186/1475-925X-4-67
- Orizio C. Sound myogram and EMG cross-spectrum during exhausting isometric contractions in humans. J Electromyogr Kinesiol 1992; 2: 141–149. DOI: 10.1016/1050-6411(92)90011-7
- Raynor AJ. Strength, power, and coactivation in children with developmental coordination disorder. Dev Med Child Neurol 2001; 43: 676–684. DOI: 10.1017/S0012162201001220
- Fong SSM, Ng SSM, Guo X, Wang Y, Chung RCK, Stat G, et al. Deficits in lower limb muscle reflex contraction latency and peak force are associated with impairments in postural control and gross motor skills of children with developmental coordination disorder: a cross-sectional study. Medicine 2015; 94: e1785. DOI: 10.1097/MD.0000000000001785
- Hess JA, Woollacott M. Effect of high-intensity strength-training on functional measures of balance ability in balance-impaired older adults. J Manip Physiol Ther 2005; 28: 582–590. DOI: 10.1016/j.jmpt.2005.08.013
- Hirsch MA, Toole T, Maitland CG, Rider RA. The effects of balance training and high-intensity resistance training on persons with idiopathic Parkinson’s disease. Arch Phys Med Rehabil 2003; 84: 1109–1117. DOI: 10.1016/S0003-9993(03)00046-7
- Watanabe K, Kouzaki M, Ogawa M, Akima H, Moritani T. Relationships between muscle strength and multi-channel surface EMG parameters in eighty-eight elderly. Eur Rev Aging Phys Act 2018; 15: 3. DOI: 10.1186/s11556-018-0192-z
- Fitzpatrick R, McCloskey DI. Proprioceptive, visual and vestibular thresholds for the perception of sway during standing in humans. J Physiol 1994; 478: 173–186. DOI: 10.1113/jphysiol.1994.sp020240
- Chen FC, Pan CY, Chu CH, Tsai CL, Tseng YT. Joint position sense of lower extremities is impaired and correlated with balance function in children with developmental coordination disorder. J Rehabil Med 2020; 52: jrm00088. DOI: 10.2340/16501977-2720
- Jaraczewska E, Long C. Kinesio taping in stroke: improving functional use of the upper extremity in hemiplegia. Top Stroke Rehabil 2006; 13: 31–42. DOI: 10.1310/33KA-XYE3-QWJB-WGT6
- Słupik A, Dwornik M, Białoszewski D, Zych E. Effect of kinesio taping on bioelectrical activity of vastus medialis muscle: preliminary report. J Orthop Trauma Rehabilitation 2007; 9: 644–651.