ORIGINAL REPORT
PREVALENCE AND BIOPSYCHOSOCIAL PREDICTORS OF POST-STROKE FATIGUE IN PATIENTS WITH MINOR STROKES
Anita KJEVERUD, PhD1, Stein ANDERSSON, PhD2,3, Anners LERDAL, PhD4,5, Anne-Kristine SCHANKE, PhD2,6 and Kristin ØSTLIE, MD, PhD1
From the 1Department of Physical Medicine and Rehabilitation, Innlandet Hospital Trust, Ottesta, 2Department of Psychology, University of Oslo, Oslo, 3Psychosomatic and CL Psychiatry, Division of Mental Health and Addiction, Oslo University Hospital, Oslo, 4Research Department, Lovisenberg Diaconal Hospital, Oslo, 5Department of Public Health and Interdisciplinary Health Sciences, Institute of Health and Society, Faculty of Medicine, University of Oslo, Oslo, and 6Research Department, Sunnaas Rehabilitation Hospital, Nesodden, Norway
Background: The majority of stroke patients suffer minor stroke. Little is known about the prevalence of post-stroke fatigue (PSF) and which factors are associated with fatigue in minor stroke patients.
Objective: To investigate the prevalence of PSF in a sample of minor stroke patients and to explore associations between biopsychosocial factors and fatigue 12 months post stroke.
Methods: In this observational study of 72 minor stroke patients fatigue symptoms were measured in the acute phase and 12 months post stroke using the Fatigue Severity Scale (FSS). At 12 months, data on psychological distress symptoms, coping strategies, balance, cognitive function, and self- report of stroke-related symptoms were collected using standardized questionnaires and tests. To explore the possible associations between fatigue and the different variables the Mann–Whitney U test and Spearman’s correlations were used. A multiple regression was conducted to identify which of the factors had the strongest association with fatigue
Results: Almost 20% of the sample were fatigue cases 1 year post stroke using a cut off of 5 on the FSS. PSF was strongly associated with having psychological distress symptoms. In univariate analyses, PSF was also associated with fatigue in the acute phase, stroke-related physical impairments, and self-report of symptoms of cognitive impairments.
Conclusions: PSF is present in a subgroup of patients with minor stroke. To identify patients at risk of developing chronic PSF, minor stroke patients should be screened for fatigue early after stroke. A short screening of factors associated with fatigue, such as psychological distress and impaired motor, visual, or cognitive function is also advisable. There is a need for further research in larger samples on PSF and associated factors in minor stroke.
LAY ABSTRACT
Post-stroke fatigue is a common consequence of stroke and is rated among the worst symptoms to cope with by stroke patients. Little is known about the prevalence of post-stroke fatigue and which factors are associated with fatigue in minor stroke patients, although the majority of stroke patients suffer minor stroke. In this observational study of 72 minor stroke patients, we investigated the prevalence of post-stroke fatigue and explored how biopsychosocial factors such as medical, psychological, physiological, and cognitive factors were associated with fatigue 1 year post stroke. This study indicates that post-stroke fatigue is present in a subgroup of patients with minor stroke. Post-stroke fatigue was associated with psychological distress symptoms, stroke-related impairments, and fatigue in the acute phase. For clinicians, it is important to identify fatigue when present as it impacts on life quality and ability to return to work. Based on our results we suggest a short screening in the early stages after stroke assessing fatigue, psychological distress, stroke-related impairments, and self-report of cognitive and visual dysfunction.
Key words: fatigue; minor stroke; psychological distress; post-stroke fatigue; physical impairments; cognitive impairments.
Citation: J Rehabil Med 2026; 58: jrm44763. DOI: https://doi.org/10.2340/jrm.v58.44763.
Copyright: © 2026 The Author(s). Published by MJS Publishing, on behalf of the Foundation for Rehabilitation Information. This is an Open Access article distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/).
Submitted: Sept 16, 2025; Accepted after revision: Mar 26, 2026; Published: Apr 21, 2026.
Correspondence address: Anita Kjeverud, Department of Physical Medicine and Rehabilitation, Innlandet Hospital Trust, Ottestad, Norway. E-mail: anita.kjeverud@sykehuset-innlandet.no
Stroke is one of the leading causes of death and disability worldwide (1). According to a meta study by Alghamdi et al. (2), post-stroke fatigue (PSF) is a severe condition affecting about 48% (95% CI 42–53%) of stroke survivors (2), associated with reduced quality of life, rehabilitation outcome, and ability to work (3–6). Fatigue has been described as a lack of physical and/or mental energy, comprising complex interactions of biological, motor-perceptive, cognitive, emotional, psychosocial, and behavioural factors (7).
Although there is a lack of consensus on the definition of minor stroke (8), current literature suggests that about 60–75% (6, 8–10) of strokes can be classified as minor. Minor stroke patients frequently have few visible disabilities and are often discharged home with little follow-up (11–13). However, research indicates that people with minor strokes also experience lasting impairments such as fatigue (8, 12, 14, 15). A recent study on patients with minor stroke reported fatigue in 53% using a cut-off of ≥ 4 on the Fatigue Severity Scale (FSS) at 3 months post stroke (10). Another study found fatigue in 31% using a cut off of ≥ 5 on the FSS 12 months post stroke (8). Substantial fatigue was found in 65.3% using the Fatigue Assessment Scale in a sample of patients with minor stroke 1 year after stroke onset (6). Nevertheless, a recent review describes a significant knowledge gap on the prevalence of fatigue following minor stroke (14).
Sequalae such as subtle motoric dysfunction, cognitive, or visual impairments, may be associated with PSF in minor stroke (4, 16, 17). A recent cross-sectional study of patients with predominantly minor stroke found impairments in either cognitive function or motor function in about one-third of the patients (9). Cognitive sequalae such as reduced information-processing speed, attention, visual memory, or executive dysfunction may be more subtle in patients with minor compared with major stroke and may therefore go unnoticed (18). However, they can still impact on functional recovery, quality of life, and return to work (19).
It has also been found that the ability to work following minor stroke is negatively affected by depression or fatigue (20, 21). Fatigue and depression are often concomitant (22), and depression may also be a consequence of fatigue if ability to return to work and other aspects of life are hampered by PSF.
In this exploratory study we have 2 aims:
- First, we aim to investigate the prevalence of PSF in a sample of stroke patients suffering minor stroke in the acute phase and at 1e year post stroke.
- Second, we aim to explore how biopsychosocial factors such as severity of physical disability, cognitive function, pain, sleep quality, coping strategies, and psychological distress are associated with fatigue in patients with minor stroke at 12 months post stroke.
MATERIALS AND METHODS
Study sample and procedures
We performed an observational study recruiting stroke patients from 1 hospital in Innlandet Hospital Trust, Norway, between February 2017 and October 2019.
Patients were eligible for the study if they were 18 years or older and had ischaemic stroke according to the International Classification of Disease 10 (ICD-10 I60-I63.9, I64) confirmed by trained physicians in the acute hospital. Exclusion criteria for participation were intracerebral haemorrhagic stroke, other somatic or psychiatric debilitating diseases, or severe language or cognitive dysfunctions causing potential problems with data collection. Patients were included if the National Institutes of Health Stroke Scale (NIHSS) score was < 5 at discharge, indicating a minor stroke and few stroke-related impairments. 28% of the original sample (n = 78) had thrombolytic treatment, in some cases leading to a significant drop in NHISS score from admission to discharge. The NIHSS score on discharge from hospital was thus chosen as this reflected their perceived level of impairment upon return to everyday life or rehabilitation.
A total of 72 patients participated in the data collection in the acute phase, defined as within the first 2 weeks after stroke onset, and 12 months post stroke. The recruitment and inclusion process is described in Fig. 1.
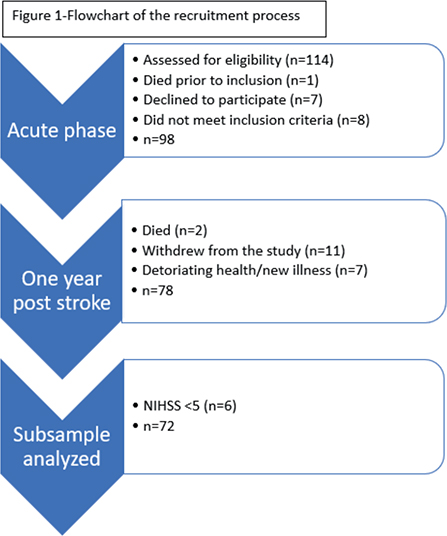
Fig. 1. Flowchart of the recruitment process.
The patients met at the clinic 12 months post-stroke. Data were collected using standardized questionnaires, neuropsychological assessment of cognitive function, and physical tests, as detailed in Table SI. Furthermore, data from the acute phase were collected from the patient’s medical records and included stroke classification, NIHSS score, comorbidities, demographic, and medical variables. Data on fatigue severity were collected at both time-points through administration of the Fatigue Severity questionnaire-7 (FSS-7). Data on pre-stroke fatigue, cohabitation, work status, and possible new comorbidities were obtained through a short interview, and the BMI was calculated at the clinic. The main outcome measures are also detailed below.
Outcome measures
Fatigue. Fatigue PSF was assessed using the Fatigue Severity Scale (FSS). In accordance with English et al. (23), the 7-item version of the instrument was chosen. The FSS is the most frequently used self-report instrument for measuring fatigue in stroke populations (24). The scale includes items such as “Fatigue is among my three most disabling symptoms” and “Fatigue interferes with carrying out certain duties and responsibilities”. Responses reflect the degree to which one agrees with each statement on a Likert scale of 1–7 based on experience within the last 7 days (25). A mean score is calculated, with higher scores indicating more severe fatigue. The FSS has shown acceptable validity and reliability across different clinical populations (26).
In accordance with Lerdal et al. (27) we used a cut-off of 5 for identifying clinically significant fatigue (17, 28). For lack of a consensus on a clinically meaningful cut-off to identify fatigue cases (29), we used the mean FSS-7 scores as a continuous variable when exploring associations between fatigue severity and predictor variables.
Demographic and medical variables. We collected information on age, employment status, and educational level from medical records and by personal interview.
National Institute of Health Stroke Scale (NIHSS) is an 11-item scale used to objectively quantify stroke impairment. It is scored during admission to hospital in the acute phase and on discharge. The scores range from 0–42; higher scores equal more impairment. Scores 0–4 indicate minor stroke, scores 5–15 a moderate stroke, scores 16–20 moderate to severe stroke, and 21–42 severe stroke (30). In this study, in accordance with Lyden et al. (30, 31), minor stroke is defined as a NIHSS score of < 5.
Questionnaires and methods to assess physical and cognitive function. The Barthel Index for ADL (activities of daily living) and Berg test of balance were used to measure level of disability and physical function. Subtests from the Delis–Kaplan Executive Function System, the WAIS IV, and the Halstead–Reitan neuropsychological test battery were used to assess cognitive function. Standardized questionnaires were used to collect data on psychological distress, pain, sleep quality, and symptoms of cognitive impairments. For details on questionnaires and methods to assess physical and cognitive function, see Table SI.
Data analyses
Statistical analyses were performed using SPSS software version 23 (IBM Corp, Armonk, NY, USA).
The Mann–Whitney U test was used to analyse how categorical variables such as pre-stroke fatigue or cohabitation were related to fatigue severity. Spearman’s correlations were conducted to explore the relationships between scores on the FSS-7 and the continuous variables. Spearman was chosen because the scores on the FSS-7 at 12 months post stroke were not normally distributed.
To reduce the risk of Type I error due to multiple testing, we set the level for statistically significant differences to p ≤ 0.01. Associations between fatigue scores and predictor variables significant at the 0.05 level are also reported but should be regarded as tendencies.
A multiple regression was conducted to identify which of the factors had the strongest association with fatigue. The screening criterion for the multivariate analyses was set to p ≤ 0.05 to avoid excluding clinically and theoretically important variables. The categorical variable “cohabitation” was transformed into a dummy variable before analyses.
Values from a Durbin–Watson analysis were acceptable within the range of 1.5 to 2.5 when controlling for possible autocorrelations.
Ethics
Before inclusion, all participants signed informed consent. The study was approved by the Regional Ethics Committee for Medical and Health Research Ethics in South-Eastern Norway (Ref. 2016/589/REK Sør-Øst C).
RESULTS
Demographic and medical characteristics
Of the 72 patients included in the analyses, a slight majority were male and the mean age on admission was 66.4 (SD 9.7). One year post stroke, the most common comorbidities were hypertension and diabetes mellitus; no association was found between comorbidities and fatigue. However, those who reported higher fatigue severity tended to have a higher NIHSS score on discharge from the acute hospital (rs.28; p = 0.02) (for details on demographic and medical characteristics, see Table I). The majority of the patients were living with a partner 1 year post stroke; living alone was associated with fatigue; z = (–2.13); p = 0.03. None of the other demographic or medical factors were associated with fatigue severity.
| Variable | ||
| Demographic | ||
| Age on admission (years), mean (SD1) | 66.4 (9.7) | |
| Sex (male/female) | 48 / 24 | |
| Cohabitation (partner/alone) | 57 / 15 | |
| Working at stroke onset (yes/no) | 34 / 38 | |
| Clinical | ||
| NIHSS2 on admission, mean (SD) | 2.8 (1.3) | |
| NIHSS on discharge, mean (SD) | 0.8 (1.4) | |
| Thrombolysis, n (%) | 20 (27.8%) | |
| BMI3, mean (SD) | 26.6 (4.3) | |
| Comorbidities, mean (SD) | 1.8 (1.4) | |
| Pre-stroke fatigue (yes), n (%) | 17 (23.6%) | |
| Physical | ||
| Barthel Index (ADL4), mean (SD) | 19.6 (1.8) | |
| Berg Balance Test, mean (SD) | 53.2 (8.1) | |
| Cognitive | ||
| Visuomotor function, mean (SD) | 47.2 (8.5) | |
| Processing speed, mean (SD) | 47.9 (8.4) | |
| Attention, mean (SD) | 49.8 (10.4) | |
| Executive function, mean (SD) | 8.1 (8.5) | |
| Self-report | ||
| Self-reported stroke-related symptoms (RPQ5), mean (SD) | 13.7 (10.3) | |
| Psychological distress (HSCL- 256), mean (SD) | 1.3 (0.3) | |
| Avoidant coping | 16.3 (3.5) | |
| Approach coping | 26.0 (6.0) | |
| Sleep quality (PSQI7), mean (SD) | 4.2 (3.4) | |
| Pain (Numeric Rating Scale), mean (SD) | 2.0 (2.4) | |
| 1Standard deviation, 2National Institute of Health Stroke Scale, 3Body Mass Index, 4Activities of Daily Living, 5Rivermead Post Concussion Symptoms Questionnaire, 6Hopkins Symptom Checklist-25, 7Pittsburgh Sleep Quality Index | ||
Fatigue symptoms, fatigue-caseness, and pre-stroke fatigue
At one year post stroke, about a fifth of the patients were fatigue cases based on a cut off of 5 at the FSS-7; of these patients, about 40% reported pre-stroke fatigue as well. Patients who reported pre-stroke fatigue also reported more fatigue 1 year post stroke; however, the association was not significant (p = 0.06). In general, the patient’s FSS scores were stable through the first year post stroke, although there were individual differences. More severe fatigue in the acute phase predicted fatigue 1 year post stroke (rs = 41; p < 0.001) (for details see Table II).
| Variable | Spearman’s Rho /z (r1) | p-value2 |
| Demographic | ||
| Age on admission (years), Spearman’s Rho | –0.01 | 0.91 |
| Sex (male/female), z (r) | –1.25 (–0.15) | 0.21 |
| Cohabitation, z (r) | –2.13 (–0.25) | 0.03* |
| Working at stroke onset (yes/no), z (r) | –2.00 (–0.24) | 0.84 |
| Clinical | ||
| NIHSS3 on admission, Spearman’s Rho | 0.016 | 0.17 |
| NIHSS3 on discharge, Spearman’s Rho | 0.28 | 0.02* |
| Thrombolysis, n (%), z (r) | –0.03 (–0.00) | 0.98 |
| BMI4, Spearman’s Rho | –0.04 | 0.78 |
| Comorbidities, Spearman’s Rho | 0.20 | 0.10 |
| Pre-stroke fatigue (yes), z (r) | –1.9 (–0.22) | 0.06 |
| Physical | ||
| Barthel Index (ADL5), Spearman’s Rho | –0.19 | 0.11 |
| Berg Balance Test, Spearman’s Rho | –0.48 | < 0.001** |
| Cognitive | ||
| Visuomotor function, Spearman’s Rho | –0.02 | 0.84 |
| Processing speed, Spearman’s Rho | 0.06 | 0.63 |
| Attention, Spearman’s Rho | 0.10 | 0.40 |
| Executive function, Spearman’s Rho | 0.04 | 0.73 |
| Self-report | ||
| FSS-scores in the acute phase, Spearman’s Rho | 0.41 | < 0.001** |
| Self-reported stroke-related symptoms (RPQ6), Spearman’s Rho | 0.42 | < 0.001** |
| Psychological distress (HSCL-257), Spearman’s Rho | 0.52 | < 0.001** |
| Avoidant coping, Spearman’s Rho | 0.37 | 0.01* |
| Approach coping, Spearman’s Rho | 0.33 | 0.01* |
| Sleep quality (PSQI8), Spearman’s Rho | 0.25 | 0.03* |
| Pain (Numeric Rating, Spearman’s Rho | 0.16 | 0.17 |
| 1Magnitude of the difference between the 2 independent groups (r), 2significance level, 3National Institute of Health Stroke Scale, 4Body Mass index, 5Activities of Daily Living, 6Rivermead Post Concussion Scale, 7Hopkins Symptom Checklist-25, 8Pittsburgh Sleep Quality Index. | ||
Associations between self-reported fatigue and clinical characteristics in the sample; physical and cognitive characteristics and self-report of symptoms
At 1 year post stroke, most of the patients in the sample were independent in activities of daily living. However, having somewhat impaired balance was significantly associated with fatigue symptoms (r2 = –0.48; p < 0.001).
There was no association between the scores on cognitive measures and fatigue severity. There was, however, a strong association between reporting more stroke-related cognitive and sensory symptoms on the RPQ (r = 0.50, p < 0.001) and fatigue severity. On the RPQ, the following items had the strongest correlation with fatigue symptoms: feelings of dizziness (rs = 0.38; p < 0.001), forgetfulness, poor memory (rs = 0.51, p < 0.001), and taking longer to think (rs = 0.39, p < 0.001). There was also an association between fatigue symptoms and poor concentration (rs = 0.35, p = 0.00, noise sensitivity (rs = 0.31, p = 0.01) and blurred vision (rs = 0.30, p = 0.01).
Patients who reported more psychological distress experienced more fatigue than those with fewer symptoms of psychological distress (rs = 0.52, p < 0.001). Severe fatigue symptoms were associated with using more avoidant coping (rs = 37; p = 0.002) and also associated with using more approach-coping strategies (rs = 0.33; p = 0.008).
Those with poorer sleep quality tended to experience more fatigue (rs = 25; p = 0.03). Most patients in this study did not report severe pain, and no association between pain and fatigue was found.
A multivariate analysis that included variables that were associated with the scores on the FSS at 12 months post stroke with a p-value of ≤ 0.05 was conducted, using linear regression. The following variables were entered into the model; FSS scores in the acute phase, NHISS score on discharge, cohabitation, Berg test of balance, psychological distress symptoms, avoidant coping, approach coping, and sleep quality. The present model suggests that 33% of the variance in fatigue could be attributed to having psychological distress symptoms. For details, see Table IV).
| Predictor | B1 | 95% CI | β2 | p-value3 |
| Cohabitation | 1.39 | –89–3.87 | 0.14 | 0.23 |
| NIHSS on discharge | 0.18 | –12–0.47 | 0.15 | 0.23 |
| Berg Balance Test | –0.01 | –0.06–0.03 | –0.08 | 0.53 |
| FSS scores in the acute phase | 0.17 | –0.12–0.46 | 0.16 | 0.46 |
| Self-reported stroke-related symptoms (RPQ44) | 0.00 | –0.04–04 | 0.03 | 0.85 |
| Psychological distress (HSCL-255) | 1.34 | 0.02–2.68 | 0.31 | 0.04* |
| Avoidant coping | 0.03 | –0.10–0.16 | 0.06 | 0.69 |
| Approach coping | 0.02 | 0.09–67 | 0.09 | 0.50 |
| Sleep quality (PSQI6) | 0.02 | –0.09–0.14 | 0.05 | 0.68 |
| Cohabitation | 1.39 | –89–3.87 | 0.14 | 0.23 |
| 1Unstardardized coefficient, 2Standardized coefficient, 3Significance level. The following variables were used in the analyses based on a p-value of <=0.05: FSS score in the acute phase, NHISS on discharge, Berg Balance Test, Self Reported Stroke Related Symptoms, 4Rivermead Post Concussion Symptoms questionnaire, Psychological distress, 5Hopkins Symptom Checklist-25, Avoidant coping, Approach coping, Sleep quality, 6Pittsburgh sleep Quality Index), Cohabitation. | ||||
DISCUSSION
Prevalence of PSF
In this study, we explored the prevalence of PSF and associates of fatigue in patients with minor stroke. We found fewer fatigue cases 1 year post stroke (19.4%) in our study than has been found in other studies, such as in the studies by Vitturi et al. (10), Morsund et al. (8) or Einstad et al. (9), who reported fatigue in 53%, 31%, and 65% in their samples. This variance may be due to differences in how many patients received thrombolytic treatment in the different studies; thrombotic treatment led to a significant reduction in stroke-related impairments in our sample. Furthermore, there is a challenge in the comparison of data on prevalence of PSF across studies, due to lack of consensus on methodology used for identifying substantial fatigue. While Vitturi et al. used a cut-off of 4 on the FSS, we used a cut off of 5 in accordance with Lerdal et al. (32). If we had used a cut-off of 4, 29.4% would have been fatigue cases 12 months post stroke, making the results more similar regarding prevalence. However, using a cut-off of 4 to identify cases may lead to an overestimation of severe fatigue as the mean score is reported to be 3.67 (SD 1.17) in the general Norwegian population (27).
Different definitions of minor stroke present another challenge in comparing results across studies. (14). Some use a NIHSS score of < 4 (10) while others use a NHISS score of <5, yet another an NIHSS score = < 5 (30, 31). Some studies use a score of ≤ 2 on the modified Rankin Scale (mRS) to define minor stroke (6, 10). We chose a cut-off of < 5 on the NIHSS in accordance with Lyden et al. (33). A similar approach is also used in comparable studies (9, 34).
A majority of stroke survivors experience minor stroke (10, 11). A prevalence of PSF of 20% in minor stroke survivors thus accounts for a large number of people.
Association between PSF and biopsychosocial factors
We found that experiencing fatigue post stroke was associated with both physical and psychological factors, and with self-reported cognitive impairment.
The strong association between symptoms of psychological distress and fatigue severity is in line with previous studies showing an association between psychological distress and PSF (35–37). For some patients, depression might be a risk or maintaining factor for fatigue, or a consequence of fatigue impacting on the capacity for participation in various aspects of life. Further, in univariate analyses there was also a strong association between living alone and experiencing fatigue. Living alone may imply less support from significant others, and thereby possibly lead to more psychological distress and fatigue.
The association of impaired balance being associated with fatigue is in line with other studies reporting that severity of PSF is associated with severity of disability (5) and functional decline (26). PSF can in some patients be attributed to the increased effort associated with the use of new movement patterns and increased effort in coping with impairments in everyday life (38). It is likely that a stronger association between fatigue and severity of disability would have been found in a sample that included patients with moderate to severe stroke, i.e., in a sample of patients with more residual impairments because of their stroke.
Scores within the normal range on cognitive tests in the majority of the patients in this sample are to be expected; however, the finding that self-report of symptoms of cognitive impairments such as using longer time to think, difficulty concentrating, and memory dysfunction was associated with experiencing more fatigue symptoms is a highly relevant finding for clinical practice. This indicates the importance of being aware of these more subtle signs of cognitive impairment to identify fatigue, even though the patients' neuropsychological tests are inconspicuous. Small deviations from population norms might represent a within-person decline. Unfortunately, we do not have any measures of the participants’ cognitive function before the stroke. The subjective cognitive complaint may be an expression of how they experience cognitive decline after their stroke. Further, subtle cognitive impairments contribute to fatigue, especially in situations that demand sustained cognitive effort. It may also be that these symptoms overlap with mental fatigue, and that mental fatigability is expressed as difficulties with concentration and forgetfulness.
Inspecting which items on the RPQ showed the highest correlation with fatigue, we found a tendency for self-reported visual problems to be associated with PSF. This is in line with the study by Pedersen et al. (17). Slow visual tracking and impaired fine-motor control may make both mental and physical tasks more effortful and thus cause fatigue, and visual rehabilitation may reduce fatigue in some stroke survivors.
Most likely is that the lack of strong association between pain or having comorbid conditions and fatigue in our study is due to a low frequency of these symptoms in our sample of minor stroke, compared with the stroke population in general.
Finally, several studies have found a strong association between pre-stroke fatigue and post-stroke fatigue (32, 39). In this study, although we found that those who reported pre-stroke fatigue tended to have higher scores on the FSS 1 year post stroke, the association was not significant. Fatigue might thus be a new experience for some minor stroke patients; in our sample 58% of the fatigue cases did not report pre-stroke fatigue. There was, however, a strong association between fatigue in the acute phase and fatigue 1 year post stroke. Thus, in line with previous studies, early fatigue predicts more long-lasting fatigue (39, 40). This implies that, in many cases, long-lasting fatigue may be predicted from experiencing fatigue symptoms shortly after a stroke. Thus, early assessment of fatigue severity is warranted.
Implications for clinical practice and further research
The results in this study support earlier findings that some patients suffer from PSF after minor stroke. Clinically, it is crucial to identify those who are vulnerable. Fatigue in the early phase post stroke predicts fatigue in later stages post stroke. Thus, an early screening of fatigue and associated factors such as stroke-related impairments is advisable to prevent the development of long-lasting, or even chronic, PSF. Primary healthcare workers may be a resource in both identifying possible unmet needs for follow-up, and in the management of the symptoms.
To date, there is relatively sparse research on treatment strategies aimed at alleviating PSF (41). Graded exercise combined with cognitive therapy (42) or psychoeducation (43) has been suggested.
Perceiving the future as manageable and using adaptive coping strategies has been suggested as a protective buffer against fatigue (44, 45). Hence, coping strategies and self-efficacy beliefs may also be possible targets for intervention. It may be that we had too few participants to capture a clear association between the use of different coping styles and fatigue, or that the unclarity results from response bias such as generally agreeing with the statements on the scale used. The possible associations between coping styles, self-efficacy beliefs, and PSF should be studied further in future research with larger samples. Qualitative research and semi-structured interviews may expand our understanding of the reasons for fatigue, and as such contribute to choices for instruments and hypotheses in future research.
In further research, a focus should also be on achieving consensus on how to measure fatigue and how to define minor stroke, yielding comparable data from different samples. Clinically, definitions of minor stroke might impact treatment decisions (46).
Strengths and weaknesses
A key strength of this study is having a multidimensional protocol consisting of demographic, medical, physical, cognitive, and psychological variables. Assessment of fatigue in the acute phase is another strength, including specific measures of coping strategies and cognitive function.
The study also has limitations. The sample is small, limiting the statistical strength of the analyses. The group of fatigue cases is small, further limiting the statistical power, and statistical outliers might affect the result. The sample being small, the findings from this exploratory study must be regarded as hypothesis-generating. There is a need for further research in larger samples, preferably multicentre studies. Further studies may include more measures in the acute phase to investigate which factors may predict long-lasting fatigue. For instance, if depression in the acute phase predicts fatigue 1 year post stroke, an early assessment and treatment of symptoms of depression and/or psychological distress is warranted.
In general, when conducting research on patients with minor stroke, definitions of minor stroke might lead to potential misclassification of the study population. It has been found that patients with left hemisphere strokes have higher NIHSS scores than patients with right hemisphere strokes due to the weight on language deficits (aphasia) in the scoring system (46).
Associations between fatigue and predictor variables at a significance level of p < 0.05 are described. However, there is a risk of type I error due to multiple testing and these findings should be regarded as tendencies. In order to minimize this risk, we reduced the level of significance from p < 0.05 to p < 0.01.
Conclusions
This exploratory study that measures fatigue in the acute phase and at 1 year post stroke indicates that PSF is present in a subgroup of patients with minor stroke. To identify patients at risk of developing chronic PSF, minor stroke patients should be screened for fatigue early after stroke. A short screening of factors associated with fatigue, such as psychological distress, and impaired motor, visual, or cognitive function, is also advisable.
It is important to identify fatigue when present as it impacts on the ability to return to work, on capacity for those who do return to work, and on everyday life and quality of life (21). Given the diversity of the factors associated with PSF, fatigue assessment and interventions should be multidisciplinary. Further research on larger samples is needed for in-depth exploration of how different biopsychosocial factors interact and impact on the development and severity of post-stroke fatigue following minor stroke.
REFERENCES
- Saini V, Guada L, Yavagal DR. Global epidemiology of stroke and access to acute ischemic stroke interventions. Neurology 2021; 97: S6–s16. https://doi.org/10.1212/WNL.0000000000012781
- Alghamdi I, Ariti C, Williams A, Wood E, Hewitt J. Prevalence of fatigue after stroke: aA systematic review and meta-analysis. Eur Stroke J 2021; 6: 319–332. https://doi.org/10.1177/23969873211047681
- Ogwumike OO, Omoregie AA, Dada OO, Badaru UM. Quality of life of stroke survivors: a cross-sectional study of association with functional independence, self-reported fatigue and exercise self-efficacy. Chronic Illn 2022; 18: 599–607. https://doi.org/10.1177/17423953211023960
- Samuelsson H, Viken J, Redfors P, Holmegaard L, Blomstrand C, Jern C, et al. Cognitive function is an important determinant of employment amongst young ischaemic stroke survivors with good physical recovery. Eur J Neurol 2021; 28: 3692–3701. https://doi.org/10.1111/ene.15014
- Staub F, Bogousslavsky J. Fatigue after stroke: a major but neglected issue. Cerebrovasc Dis 2001; 12: 75–81. https://doi.org/10.1159/000047685
- Ramírez-Moreno JM, Muñoz-Vega P, Alberca SB, Peral-Pacheco D. Health-related quality of life and fatigue after transient ischemic attack and minor stroke. J Stroke Cerebrovasc Dis 2019; 28: 276–284. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.09.046
- Acciarresi M, Bogousslavsky J, Paciaroni M. Post-stroke fatigue: epidemiology, clinical characteristics and treatment. Eur Neurology 2014; 72: 255–261. https://doi.org/10.1159/000363763
- Morsund Å H, Ellekjær H, Gramstad A, Reiestad MT, Midgard R, Sando SB, et al. Cognitive and emotional impairment after minor stroke and non-ST-elevation myocardial infarction (NSTEMI): a prevalence study. Stroke Res Treat 2019; 2019: 2527384. https://doi.org/10.1155/2019/2527384
- Einstad MS, Saltvedt I, Lydersen S, Ursin MH, Munthe-Kaas R, Ihle-Hansen H, et al. Associations between post-stroke motor and cognitive function: a cross-sectional study. BMC Geriatr 2021; 21: 103. https://doi.org/10.1186/s12877-021-02055-7
- Vitturi BK, Mitre LP, Kim AIH, Gagliardi RJ. Prevalence and predictors of fatigue and neuropsychiatric symptoms in patients with minor ischemic stroke. J Stroke Cerebrovasc Dis 2021; 30: 105964. https://doi.org/10.1016/j.jstrokecerebrovasdis.2021.105964
- Liljehult J, Molsted S, Christensen T, Møller T, Overgaard D. Mastering health following minor stroke: a qualitative explorative study. J Stroke Cerebrovasc Dis 2022; 31: 106607. https://doi.org/10.1016/j.jstrokecerebrovasdis.2022.106607
- Turner GM, Aquino M, Atkins L, Foy R, Mant J, Calvert M. Factors influencing follow-up care post-TIA and minor stroke: a qualitative study using the theoretical domains framework. BMC Health Serv Res 2022; 22: 235. https://doi.org/10.1186/s12913-022-07607-0
- Turner GM, Jones R, Collis P, Patel S, Jowett S, Tearne S, et al. Structured follow-up pathway to support people after transient ischaemic attack and minor stroke (SUPPORT TIA): protocol for a feasibility study and process evaluation. BMJ Open 2022; 12e060280. https://doi.org/10.1136/bmjopen-2021-060280
- Hede Ebbesen B, Modrau B, Kontou E, Finch E, Crowfoot G, Crow J, et al. Lasting impairments following transient ischemic attack and minor stroke: a systematic review protocol. Front Neurol 2023; 14: 1177309. https://doi.org/10.3389/fneur.2023.1177309
- Morsund ÅH, Ellekjaer H, Gramstad A, Reiestad MT, Midgard R, Sando SB, et al. The development of cognitive and emotional impairment after a minor stroke: a longitudinal study. Acta Neurol Scand 2019; 140: 281–289. https://doi.org/10.1111/ane.13143
- Vlachos G, Ihle-Hansen H, Bruun Wyller T, Brækhus A, Mangset M, Hamre C, et al. Cognitive and emotional symptoms in patients with first-ever mild stroke: the syndrome of hidden impairments. J Rehabil Med 2021; 53: jrm00135. https://doi.org/10.2340/16501977-2764
- Pedersen SG, Løkholm M, Friborg O, Halvorsen MB, Kirkevold M, Heiberg G, et al. Visual problems are associated with long-term fatigue after stroke. J Rehabil Med 2023; 55: jrm00374. https://doi.org/10.2340/jrm.v55.4813
- Divya KP, Menon RN, Varma RP, Sylaja PN, Thomas B, Kesavadas C, et al. Post-stroke cognitive impairment: a cross-sectional comparison study between mild cognitive impairment of vascular and non-vascular etiology. J Neurol Sci 2017; 372: 356–362. https://doi.org/10.1016/j.jns.2016.10.031
- Fride Y, Adamit T, Maeir A, Ben Assayag E, Bornstein NM, Korczyn AD, et al. What are the correlates of cognition and participation to return to work after first ever mild stroke? Top Stroke Rehabil 2015; 22: 317–325. https://doi.org/10.1179/1074935714Z.0000000013
- Wicht CA, Chavan CF, Annoni JM, Balmer P, Aellen J, Humm AM, et al. Predictors for returning to paid work after transient ischemic attack and minor ischemic stroke. J Pers Med 2022; 12. https://doi.org/10.3390/jpm12071109
- Norlander A, Lindgren I, Pessah-Rasmussen H, Gard G, Brogårdh C. Fatigue in men and women who have returned to work after stroke: assessed with the Fatigue Severity Scale and Mental Fatigue Scale. J Rehabil Med 2021; 53: jrm00227. https://doi.org/10.2340/16501977-2863
- Ponchel A, Bombois S, Bordet R, Henon H. Factors associated with poststroke fatigue: a systematic review. Stroke Res Treat 2015: 347920. https://doi.org/10.1155/2015/347920
- English C, Simpson DB, Billinger SA, Churilov L, Coupland KG, Drummond A, et al. A roadmap for research in post-stroke fatigue: consensus-based core recommendations from the third Stroke Recovery and Rehabilitation Roundtable. Int J Stroke 2023: 17474930231189135. https://doi.org/10.1177/17474930231189135
- Skogestad IJ, Kirkevold M, Indredavik B, Gay CL, Lerdal A. Lack of content overlap and essential dimensions: a review of measures used for post-stroke fatigue. J Psychosom Res 2019; 124: 109759. https://doi.org/10.1016/j.jpsychores.2019.109759
- Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD. The fatigue severity scale: application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol 1989; 46: 1121–1123. https://doi.org/10.1001/archneur.1989.00520460115022
- Nadarajah M, Mazlan M, Abdul-Latif L, Goh HT. Test–retest reliability, internal consistency and concurrent validity of Fatigue Severity Scale in measuring post-stroke fatigue. Eur J Phys Rehabil Med 2017; 53: 703–709. https://doi.org/10.23736/S1973-9087.16.04388-4
- Lerdal A, Wahl A, Rustoen T, Hanestad BR, Moum T. Fatigue in the general population: a translation and test of the psychometric properties of the Norwegian version of the fatigue severity scale. Scand J Public Health 2005; 33: 123–130. https://doi.org/10.1080/14034940410028406
- Lerdal A, Gay CL. Acute-phase fatigue predicts limitations with activities of daily living 18 months after first-ever stroke. J Stroke Cerebrovasc Dis 2017; 26: 523–531. https://doi.org/10.1016/j.jstrokecerebrovasdis.2016.11.130
- Kjeverud A, Andersson S, Lerdal A, Schanke AK, Østlie K. A cross-sectional study exploring overlap in post-stroke fatigue caseness using three fatigue instruments: Fatigue Severity Scale, Fatigue Questionnaire and the Lynch’s Clinical Interview. J Psychosom Res 2021: 110605. https://doi.org/10.1016/j.jpsychores.2021.110605
- Lyden P. Using the National Institutes of Health Stroke Scale: a cautionary tale. Stroke 2017; 48: 513–519. https://doi.org/10.1161/STROKEAHA.116.015434
- Yakhkind A, McTaggart RA, Jayaraman MV, Siket MS, Silver B, Yaghi S. Minor stroke and transient ischemic attack: research and practice. Front Neurol 2016; 7: 86. https://doi.org/10.3389/fneur.2016.00086
- Lerdal A, Bakken LN, Rasmussen EF, Beiermann C, Ryen S, Pynten S, et al. Physical impairment, depressive symptoms and pre-stroke fatigue are related to fatigue in the acute phase after stroke. Disabil Rehabil 2011; 33: 334–342. https://doi.org/10.3109/09638288.2010.490867
- Lyden PD, Lu M, Levine SR, Brott TG, Broderick J. A modified National Institutes of Health Stroke Scale for use in stroke clinical trials: preliminary reliability and validity. Stroke 2001; 32: 1310–1317. https://doi.org/10.1161/01.STR.32.6.1310
- Marsh EB, Lawrence E, Hillis AE, Chen K, Gottesman RF, Llinas RH. Pre-stroke employment results in better patient-reported outcomes after minor stroke. Clin Neurol Neurosurg 2018: 38–42. https://doi.org/10.1016/j.clineuro.2017.12.020
- Pedersen A, Almkvist E, Holmegaard L, Lagging C, Redfors P, Blomstrand C, et al. Fatigue 7 years post-stroke: predictors and correlated features. Acta Neurol Scand 2022; 146: 295–303. https://doi.org/10.1111/ane.13665
- Rahamatali M, De Bont N, Valet M, Halkin V, Hanson P, Deltombe T, et al. Post-stroke fatigue: how it relates to motor fatigability and other modifiable factors in people with chronic stroke. Acta Neurol Belg 2021; 121: 181–189. https://doi.org/10.1007/s13760-020-01453-9
- Sibbritt D, Bayes J, Peng W, Maguire J, Adams J. Associations between fatigue and disability, depression, health-related hardiness and quality of life in people with stroke. J Stroke Cerebrovasc Dis 2022; 31: 106543. https://doi.org/10.1016/j.jstrokecerebrovasdis.2022.106543
- Goh HT, Stewart JC. Poststroke fatigue is related to motor and cognitive performance: a secondary analysis. J Neurol Phys Ther 2019; 43: 233–239. https://doi.org/10.1097/NPT.0000000000000290
- Kjeverud A, Østlie K, Schanke A-K, Gay C, Thoresen M, Lerdal A. Trajectories of fatigue among stroke patients from the acute phase to 18 months post-injury: a latent class analysis. PloS One 2020; 15: e0231709. https://doi.org/10.1371/journal.pone.0231709
- Lerdal A, Gay CL. Fatigue in the acute phase after first stroke predicts poorer physical health 18 months later. Neurology 2013; 81: 1581–1587. https://doi.org/10.1212/WNL.0b013e3182a9f471
- Aali G, Drummond A, das Nair R, Shokraneh F. Post-stroke fatigue: a scoping review. F1000Res 2020; 9: 242. https://doi.org/10.12688/f1000research.22880.2
- Zedlitz AM, Rietveld TC, Geurts AC, Fasotti L. Cognitive and graded activity training can alleviate persistent fatigue after stroke: a randomized, controlled trial. Stroke 2012; 43: 1046–1051. https://doi.org/10.1161/STROKEAHA.111.632117
- Su Y, Yuki M, Otsuki M. Non-pharmacological interventions for post-stroke fatigue: systematic review and network meta-analysis. J Clin Med 2020; 9: 621. https://doi.org/10.3390/jcm9030621
- Løke D, Andelic N, Helseth E, Vassend O, Andersson S, Ponsford JL, et al. Impact of somatic vulnerability, psychosocial robustness and injury-related factors on fatigue following traumatic brain injury: a cross-sectional study. J Clin Med 2022; 11: 1733. https://doi.org/10.3390/jcm11061733
- Ezekiel L, Field L, Collett J, Dawes H, Boulton M. Experiences of fatigue in daily life of people with acquired brain injury: a qualitative study. Disabil Rehabil 2021; 43: 2866–2874. https://doi.org/10.1080/09638288.2020.1720318
- Vitti E, Kim G, Stockbridge MD, Hillis AE, Faria AV. Left hemisphere bias of NIH Stroke scale is most severe for middle cerebral artery strokes. Front Neurol 2022; 13: 912782. https://doi.org/10.3389/fneur.2022.912782
- Mahoney FI, Barthel DW. Functional evaluation: the Barthel Index. Md State Med J 1965: 61–65. https://doi.org/10.1037/t02366-000
- Berg KO, Wood-Dauphinee SL, Williams JI, Maki B. Measuring balance in the elderly: validation of an instrument. Can J Public Health 1992; 83: S7–11.
- Sangha O, Stucki G, Liang MH, Fossel AH, Katz JN. The Self-Administered Comorbidity Questionnaire: a new method to assess comorbidity for clinical and health services research. Arthritis Rheum 2003; 49: 156–163. https://doi.org/10.1002/art.10993
- Reitan RM, Wolfson D. The Halstead–Reitan neuropsychological test battery: theory and clinical interpretation. Tucson, AZ: Neuropsychology Press; 1985.
- Delis DC, Kaplan E, Kramer JH. Delis–Kaplan executive function system. 2001. Available from: https://doi.org/10.1037/t15082-000
- Delis D, Kramer J, Kaplan E, Ober B. California verbal learning test, adult version (CVLT-II). Cleveland, OH: Psychological Corporation; 2000.
- Wechsler D. Wechsler adult intelligence scale. Arch Clin Neuropsychol 1955.
- King NS, Crawford S, Wenden FJ, Moss NE, Wade DT. The Rivermead Post Concussion Symptoms Questionnaire: a measure of symptoms commonly experienced after head injury and its reliability. J Neurol 1995; 242: 587–592. https://doi.org/10.1007/BF00868811
- Derogatis LR, Lipman RS, Rickels K, Uhlenhuth EH, Covi L. The Hopkins Symptom Checklist (HSCL): a measure of primary symptom dimensions. Mod Probl Pharmacopsychiatry 1974: 79–110. https://doi.org/10.1159/000395070
- Eisenberg SA, Shen BJ, Schwarz ER, Mallon S. Avoidant coping moderates the association between anxiety and patient-rated physical functioning in heart failure patients. J Behav Med 2012; 35: 253–261. https://doi.org/10.1007/s10865-011-9358-0
- Carver CS. You want to measure coping but your protocol’s too long: consider the brief COPE. Int J Behav Med 1997; 4: 92–100. https://doi.org/10.1207/s15327558ijbm0401_6
- Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatr Res 1989; 28: 193–213. https://doi.org/10.1016/0165-1781(89)90047-4
- Williamson A, Hoggart B. Pain: a review of three commonly used pain rating scales. J Clin Nurs 2005; 14: 798–804. https://doi.org/10.1111/j.1365-2702.2005.01121.x